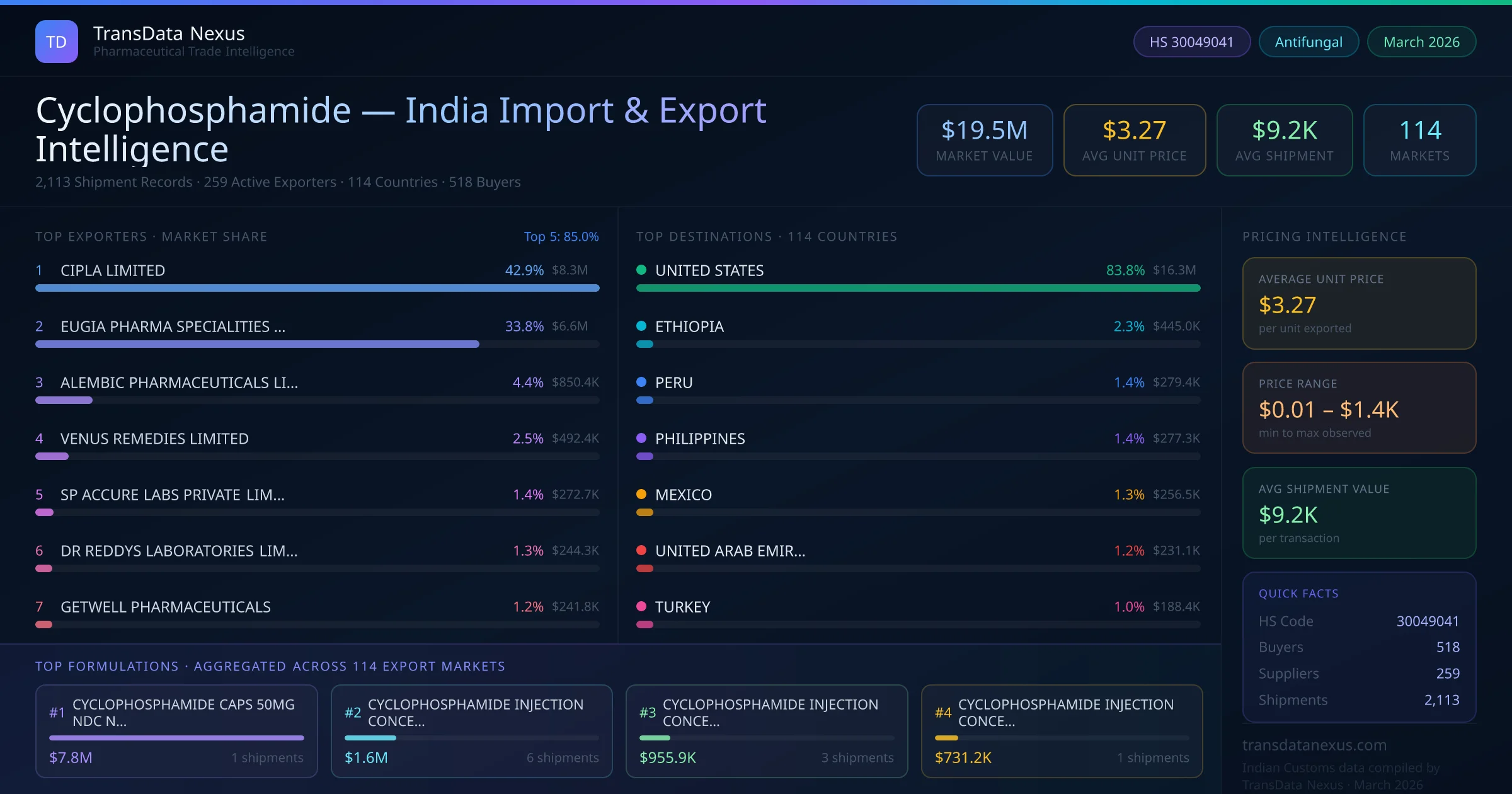
To import finished pharmaceutical formulations containing Cyclophosphamide (HS Code 30049041) into India, the following regulatory steps are mandatory:
1. Import Registration and License: Obtain an import license from the Central Drugs Standard Control Organization (CDSCO) as per the Drugs and Cosmetics Act, 1940, and associated rules. This license is essential for the legal import and sale of pharmaceutical products in India.
2. CDSCO Registration: The product must be registered with CDSCO, ensuring compliance with Indian standards for safety, efficacy, and quality.
3. Form CT-20/40/41: Depending on the product's classification, submit the appropriate form (CT-20 for new drugs, CT-40 for clinical trial imports, or CT-41 for restricted drugs) to CDSCO for approval.
4. DCGI Approval: The Drug Controller General of India (DCGI) must approve the product, confirming its compliance with Indian regulations.
5. No Objection Certificate (NOC): If the product is already marketed in another country, an NOC from the regulatory authority of that country may be required.
Timeline: The import drug registration process can take several months, depending on the completeness of the application and the regulatory workload.
For Cyclophosphamide formulations under HS Code 30049041, ensure all documentation, including manufacturing licenses, stability data, and quality control certificates, are in order to facilitate a smooth approval process.