Cote D Ivoire to India Pharmaceutical Import
Bilateral Trade Intelligence · $0 Total Trade · 1 Foreign Suppliers · 1 Indian Buyers · DGFT Verified · Updated March 2026
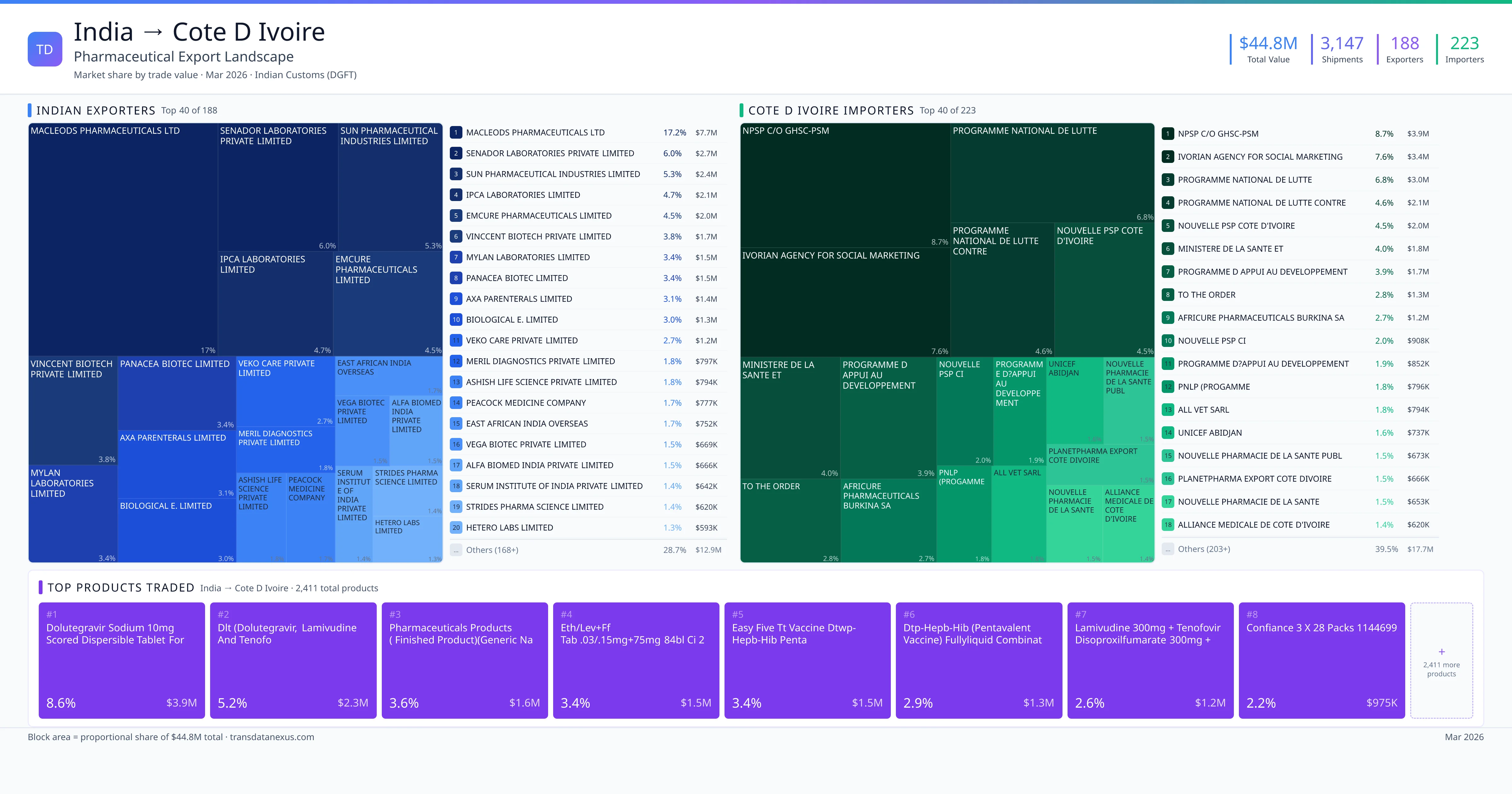
India imported $0 worth of pharmaceutical formulations from Cote D Ivoire across 1 verified shipments, from 1 foreign suppliers to 1 Indian buyers. The top suppliers are CENTRE NATIONAL DE TRANSFUSION SANGUINE ( CNTS ) ($0). The leading products are Plasma ($0). Average shipment value: $0.
Top Pharmaceutical Formulations — Cote D Ivoire to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Plasma | $0 | 0.0% |
India imports 1+ pharmaceutical formulations from Cote D Ivoire with a combined trade value of $0. Key products include Plasma ($0). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Cote D Ivoire to India Trade Routes
These are the top pharmaceutical products exported from India to Cote D Ivoire, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Cote D Ivoire buyers, regulatory requirements, and logistics for that specific product corridor. Products include Plasma ($0) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Cote D Ivoire Suppliers to India
1 Indian pharmaceutical companies export finished formulations to Cote D Ivoire. Leading exporters include Centre National De Transfusion Sanguine ( Cnts ), . The top exporter accounts for 0.0% of total India–Cote D Ivoire pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Cote D Ivoire
1 companies in Cote D Ivoire import pharmaceutical formulations from India. Top buyers include Promea Therapeutics Private Limited, . The largest buyer accounts for 0.0% of India–Cote D Ivoire pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–Cote D Ivoire Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between Côte d'Ivoire and India has remained minimal from 2022 to 2026. In 2022, India imported pharmaceutical products worth $25 from Côte d'Ivoire, according to the United Nations COMTRADE database. This figure remained unchanged in subsequent years, indicating a stagnant trade relationship in this sector. The limited volume suggests that the pharmaceutical trade between the two nations has not experienced significant growth or development during this period.
2Cote D Ivoire's Role in India's Pharma Imports
Côte d'Ivoire's contribution to India's pharmaceutical imports is negligible, accounting for a minimal fraction of India's total pharmaceutical import value. The primary product imported from Côte d'Ivoire is plasma, with a total import value of $0 and a share of 0.0%. This indicates that Côte d'Ivoire does not play a significant role in supplying pharmaceutical products to India.
3Recent Developments
Between 2024 and 2026, there have been no notable developments in the pharmaceutical trade between Côte d'Ivoire and India. No new trade agreements, policy changes by the Central Drugs Standard Control Organisation (CDSCO), or regulatory harmonization efforts have been reported. The stagnant trade volume suggests a lack of significant policy or regulatory changes affecting this trade corridor during this period.
Cote D Ivoire Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
For pharmaceutical products to be imported into India, they must be registered with the CDSCO under Form 41. This registration serves as authorization for the importation of specific pharmaceutical products. Additionally, companies engaged in pharmaceutical import activities must obtain an Importer Exporter Code (IEC) from the Directorate General of Foreign Trade (DGFT). The IEC is a prerequisite for customs clearance and facilitates international trade transactions. Importers are also required to submit an authorization letter from the manufacturer or marketing authorization holder (MAH) authorizing them to import specific pharmaceutical products on their behalf.
2GMP & Manufacturing Standards
India requires that manufacturing sites comply with Good Manufacturing Practices (GMP) standards to ensure the quality and safety of pharmaceutical products. The CDSCO conducts inspections to verify compliance with these standards. Manufacturing sites must also obtain WHO prequalification, which is a prerequisite for exporting pharmaceutical products to India. This prequalification ensures that the manufacturing site meets international quality standards recognized by the World Health Organization.
3Import Documentation
Importers must obtain an import license (Form 10) issued by the CDSCO, which is mandatory for importing pharmaceutical products into India. The registration certificate under Form 41 serves as authorization for the importation of specific pharmaceutical products. A No Objection Certificate (NOC) from the manufacturer or marketing authorization holder (MAH) is required, authorizing the importer to import specific pharmaceutical products on their behalf. A test license may be required for certain products to ensure they meet Indian standards. Customs procedures involve submitting the necessary documentation to Indian customs authorities for clearance.
Product Categories & Therapeutic Trends — India to Cote D Ivoire
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
The pharmaceutical imports from Côte d'Ivoire to India are limited, with plasma being the only product imported, accounting for 100% of the import value. This suggests that there are no significant therapeutic areas or product types dominating India's pharmaceutical imports from Côte d'Ivoire.
2Innovation & Specialty Imports
Given the minimal volume of pharmaceutical imports from Côte d'Ivoire to India, there are no notable innovator drugs, novel formulations, or advanced therapies sourced from Côte d'Ivoire that are not manufactured domestically in India.
3Import Demand Drivers
The negligible pharmaceutical imports from Côte d'Ivoire to India indicate that factors such as patent protection, technology gaps, quality requirements, or disease burden are not significant drivers for importing pharmaceutical products from Côte d'Ivoire.
Trade Policy & Tariff Intelligence — India and Cote D Ivoire
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes the Most Favored Nation (MFN) tariff rates, basic customs duty, Integrated Goods and Services Tax (IGST), and health cess. However, specific tariff rates and duties applicable to pharmaceutical imports from Côte d'Ivoire are not detailed in the available data. The minimal import volume suggests that these tariffs and duties have a limited impact on the trade between the two countries.
2Trade Agreements & Preferences
There is no evidence of a Free Trade Agreement (FTA) between India and Côte d'Ivoire, nor are there preferential duty rates or bilateral negotiations specifically affecting pharmaceutical imports. The stagnant trade volume indicates a lack of significant trade agreements or preferences in this sector.
3IP, Patents & Price Control
India's patent regime, including Section 3(d), compulsory licensing, and price control measures by the National Pharmaceutical Pricing Authority (NPPA), applies to imported drugs. However, the minimal pharmaceutical imports from Côte d'Ivoire suggest that these intellectual property and price control mechanisms have limited relevance in this trade corridor.
Supply Chain & Logistics — India to Cote D Ivoire Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
The pharmaceutical trade between Côte d'Ivoire and India is minimal, with only one recorded shipment. Therefore, specific details regarding major sea and air routes, transit times, and route reliability are not available.
2Port Infrastructure
The primary export ports in Côte d'Ivoire include the Autonomous Port of Abidjan and the port of San-Pedro. In India, major import ports include Jawaharlal Nehru Port (Nhava Sheva), Mumbai Port, and Chennai Port. However, given the minimal pharmaceutical trade between the two countries, specific data on port infrastructure handling these shipments is limited.
3Indian Customs & Cold Chain
For pharmaceutical imports, Indian customs procedures involve clearance by customs authorities, which includes verification of import documentation and compliance with regulatory requirements. The Central Drugs Standard Control Organisation (CDSCO) conducts testing to ensure that imported pharmaceutical products meet Indian standards. Cold chain management and Good Distribution Practice (GDP) compliance are critical for maintaining the quality of pharmaceutical products during transit. However, due to the minimal volume of pharmaceutical imports from Côte d'Ivoire, specific details on these logistics aspects are not extensively documented.
Market Opportunity Assessment — Cote D Ivoire for Indian Pharma
Market size, healthcare system, growth outlook
1India's Import Dependency
Despite being a major pharmaceutical manufacturer, India imports certain pharmaceutical products to meet specific needs, such as patented drugs, biologics, or products not manufactured domestically. However, the negligible pharmaceutical imports from Côte d'Ivoire suggest that India does not rely on Côte d'Ivoire for these products.
2Cote D Ivoire Companies in India
There is limited information available regarding major Côte d'Ivoire pharmaceutical companies supplying India. The minimal trade volume indicates that Côte d'Ivoire companies have limited presence or partnerships in the Indian pharmaceutical market.
3Outlook & Self-Reliance
India's Atmanirbhar Bharat initiative and the Production-Linked Incentive (PLI) scheme aim to boost domestic pharmaceutical manufacturing and reduce import dependency. Given the minimal pharmaceutical imports from Côte d'Ivoire, it is unlikely that these imports will grow significantly or be substituted by domestic production in the near future.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Cote D Ivoire
Competing origins, India's edge, challenges and threats
1Competing Import Origins
India imports pharmaceutical products from various countries, including the USA, EU, China, Switzerland, and Japan. Côte d'Ivoire's share in this import landscape is negligible compared to these other origins. The minimal trade volume suggests that Côte d'Ivoire does not compete significantly in supplying pharmaceutical products to India.
2Cote D Ivoire's Competitive Advantages
The limited pharmaceutical trade between Côte d'Ivoire and India indicates that Côte d'Ivoire's products do not possess significant competitive advantages in the Indian market, such as innovation, quality, patent protection, or brand reputation.
3Domestic Competition & Threats
Indian manufacturers are continually developing alternatives to imported pharmaceutical products, including biosimilars, process patents, and expanding capacity through initiatives like the PLI scheme. The minimal pharmaceutical imports from Côte d'Ivoire suggest that domestic competition and threats in this sector are not significantly influenced by Côte d'Ivoire's pharmaceutical products.
FAQ — India to Cote D Ivoire Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Cote D Ivoire?
India exported pharmaceuticals worth $0 to Cote D Ivoire across 1 verified shipments.
Who are the top Indian pharmaceutical exporters to Cote D Ivoire?
1. CENTRE NATIONAL DE TRANSFUSION SANGUINE ( CNTS ) — $0. Total: 1 suppliers.
Which companies in Cote D Ivoire import pharmaceuticals from India?
1. PROMEA THERAPEUTICS PRIVATE LIMITED — $0. 1 buyers total.
What pharmaceutical products does India export most to Cote D Ivoire?
1. Plasma ($0, 0.0%)
Which ports handle pharmaceutical shipments from India to Cote D Ivoire?
Export: . Import: .
Why does Cote D Ivoire import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $0 corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Cote D Ivoire?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Cote D Ivoire pharmaceutical trade?
$0 per consignment across 1 shipments.
How many Indian pharmaceutical companies export to Cote D Ivoire?
1 Indian companies. Largest: CENTRE NATIONAL DE TRANSFUSION SANGUINE ( CNTS ) with $0.
How can I find verified Indian pharmaceutical suppliers for Cote D Ivoire?
TransData Nexus covers 1 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Cote D Ivoire Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Cote D Ivoire identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1 verified shipments from 1 Indian exporters to 1 Cote D Ivoire buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
1 Exporters
1 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists