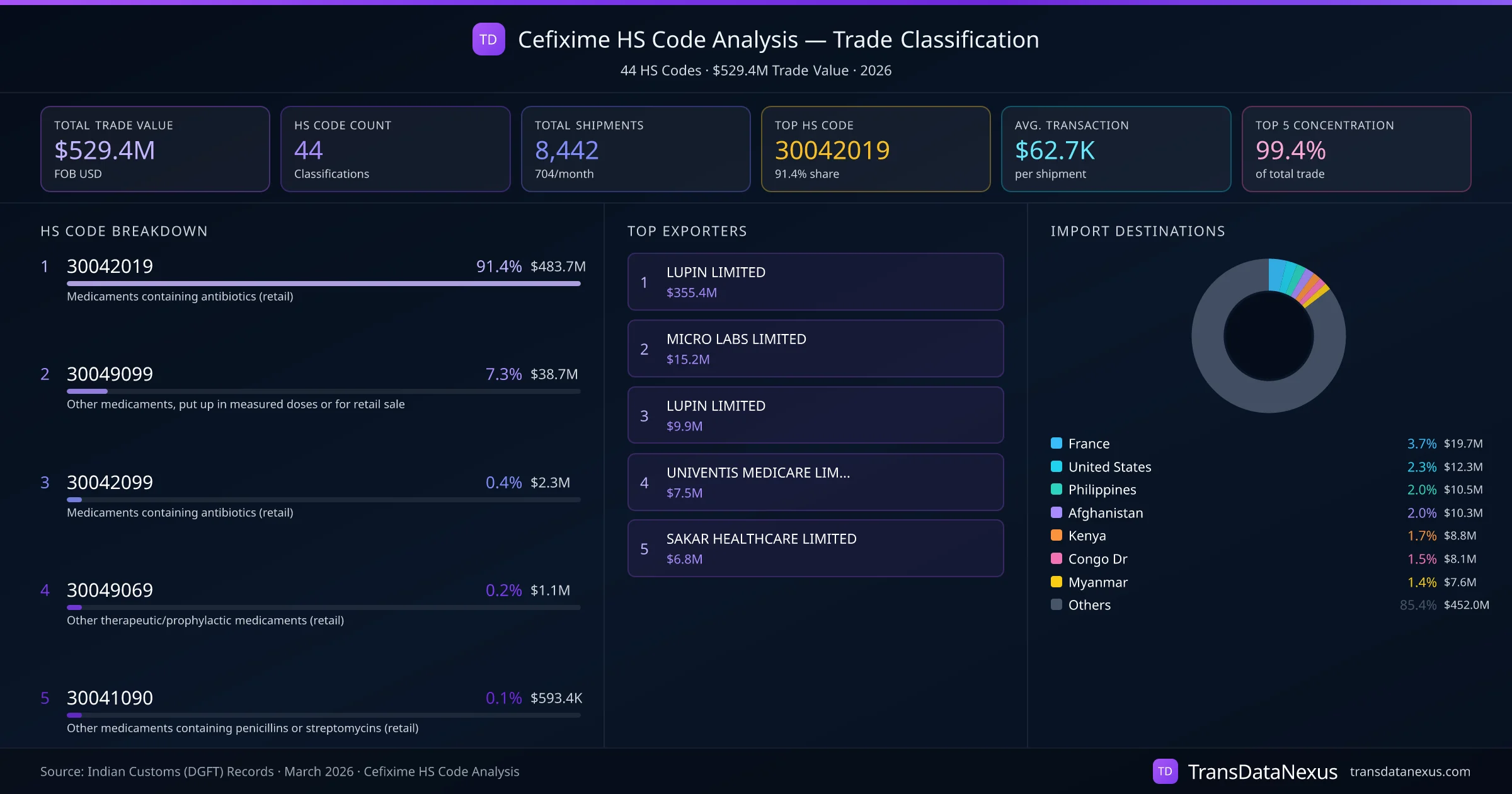
Cefixime, an antibiotic used to treat various bacterial infections, is primarily classified under HS code 30042019, which pertains to "Other, containing antibiotics." However, classification disputes can arise, particularly concerning whether a product should be classified under HS code 3003 or 3004.
- HS Code 3003: This code covers "Medicaments... not put up in measured doses or in forms or packings for retail sale." Products classified under this code are typically bulk pharmaceuticals intended for further processing or repackaging.
- HS Code 3004: This code includes "Medicaments... put up in measured doses or in forms or packings for retail sale." Finished pharmaceutical products ready for consumer use fall under this classification.
Common misclassification errors occur when the packaging and intended use of Cefixime are not accurately considered. For instance, bulk Cefixime intended for pharmaceutical manufacturing should be classified under HS code 3003, while finished products in retail packaging should be under HS code 3004.
Misclassification can lead to customs penalties, including fines and delays in shipment clearance. The World Customs Organization (WCO) emphasizes the importance of accurate classification to ensure compliance and facilitate international trade. Recent amendments to the Harmonized System, effective from January 1, 2028, aim to address classification challenges and reduce litigation risks associated with pharmaceutical products.