Who Buys Carfilzomib from India — 105 Importers Behind a $1.2M Market
India's carfilzomib import market is served by 105 active buyers who collectively imported $1.2M across 203 shipments. ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDA (ECUADOR) leads with a 4.4% market share, followed by VLADIMIR GASPARYAN and EVEREST BANK LTD. The top 5 buyers together control 10.4% of total import value, reflecting a moderately competitive buyer landscape.
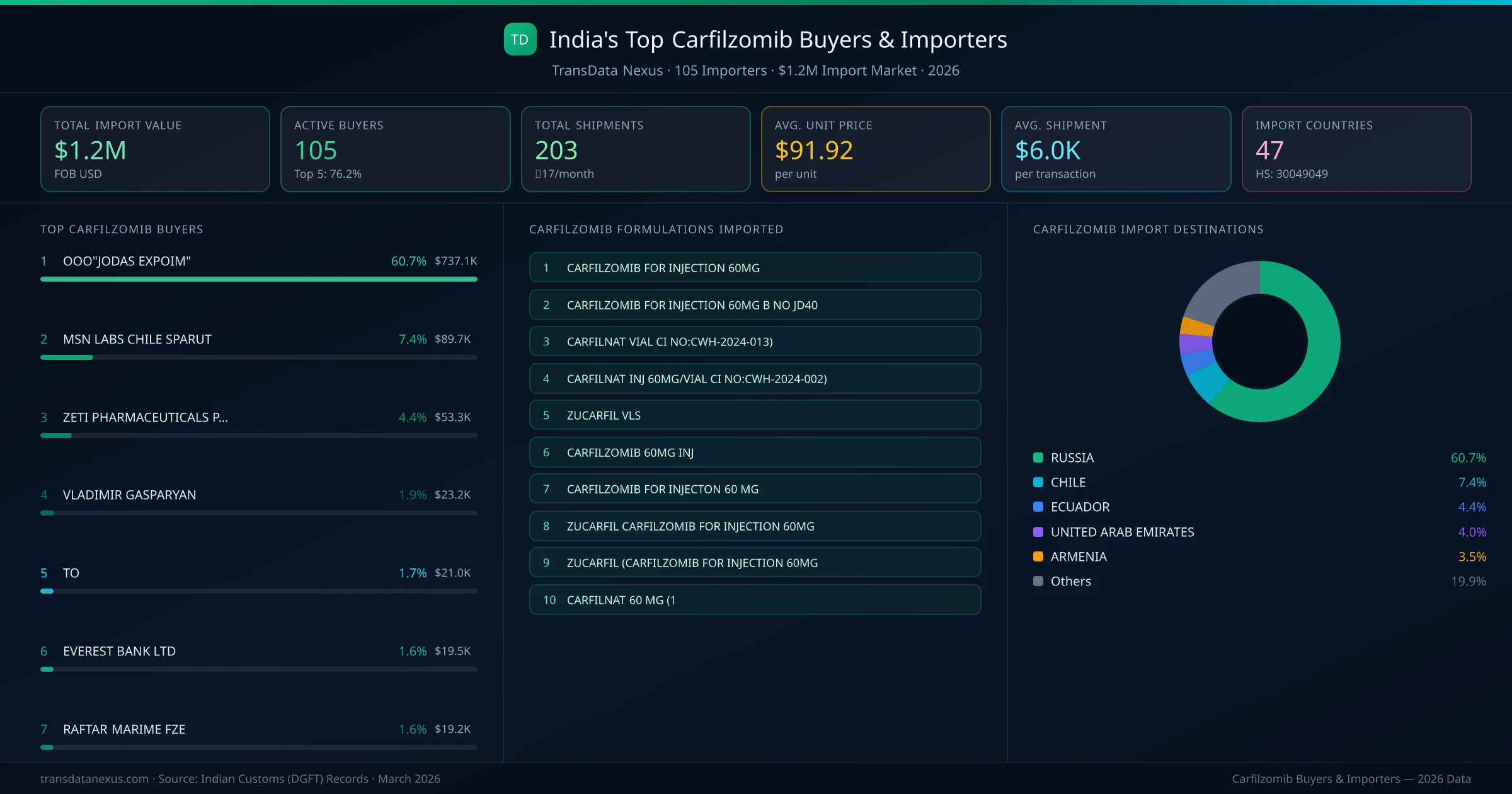
Top Carfilzomib Importers — Ranked by Import Value
ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDA (ECUADOR) is the leading carfilzomib importer from India, holding a 4.4% share of the $1.2M market across 203 shipments from 105 buyers. The top 5 buyers — ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDA (ECUADOR), VLADIMIR GASPARYAN (ARMENIA), EVEREST BANK LTD (NEPAL), RAFTAR MARIME FZE (UNITED ARAB EMIRATES), VLADIMIR GASPARYAN APP (ARMENIA) — collectively control 10.4% of total import value.
Top Carfilzomib Buyers & Importers
Ranked by import value · 105 active buyers · Indian Customs (DGFT) data
| # | Buyer & Formulations | Value | Suppliers | Share |
|---|---|---|---|---|
| 1 | ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDAECUADOR | $53.3K | 5 | 4.4% |
| 2 | VLADIMIR GASPARYANARMENIA CARFILZOMIB FOR INJECTION 60MGCARFILZOMIB FOR INJECTION 60MG B NO JD40 | $23.2K | 5 | 1.9% |
| 3 | EVEREST BANK LTDNEPAL CARFILNAT VIAL CI NO:CWH-2024-013)CARFILNAT INJ 60MG/VIAL CI NO:CWH-2024-002)CARFILZOMIB FOR INJECTION 60MG | $19.5K | 5 | 1.6% |
| 4 | RAFTAR MARIME FZEUNITED ARAB EMIRATES | $19.2K | 5 | 1.6% |
| 5 | VLADIMIR GASPARYAN APPARMENIA CARFILZOMIB FOR INJECTION 60MGCARFILZOMIB FOR INJECTION 60MG B NO JD40 | $11.6K | 5 | 1.0% |
| 6 | SIMONE TAMARA WILLIAMSJAMAICA ZUCARFIL VLSZUCARFIL CARFILZOMIB FOR INJECTION 60MGZUCARFIL (CARFILZOMIB FOR INJECTION 60MG | $7.6K | 5 | 0.6% |
| 7 | LAMAR INTERNATIONALSOUTH AFRICA | $6.8K | 5 | 0.6% |
| 8 | SKIN LINE LOGISTICIRAQ | $5.1K | 5 | 0.4% |
Related Analysis
Market Analysis: Top Importing Countries & Importers
Comprehensive geographic market intelligence dashboard
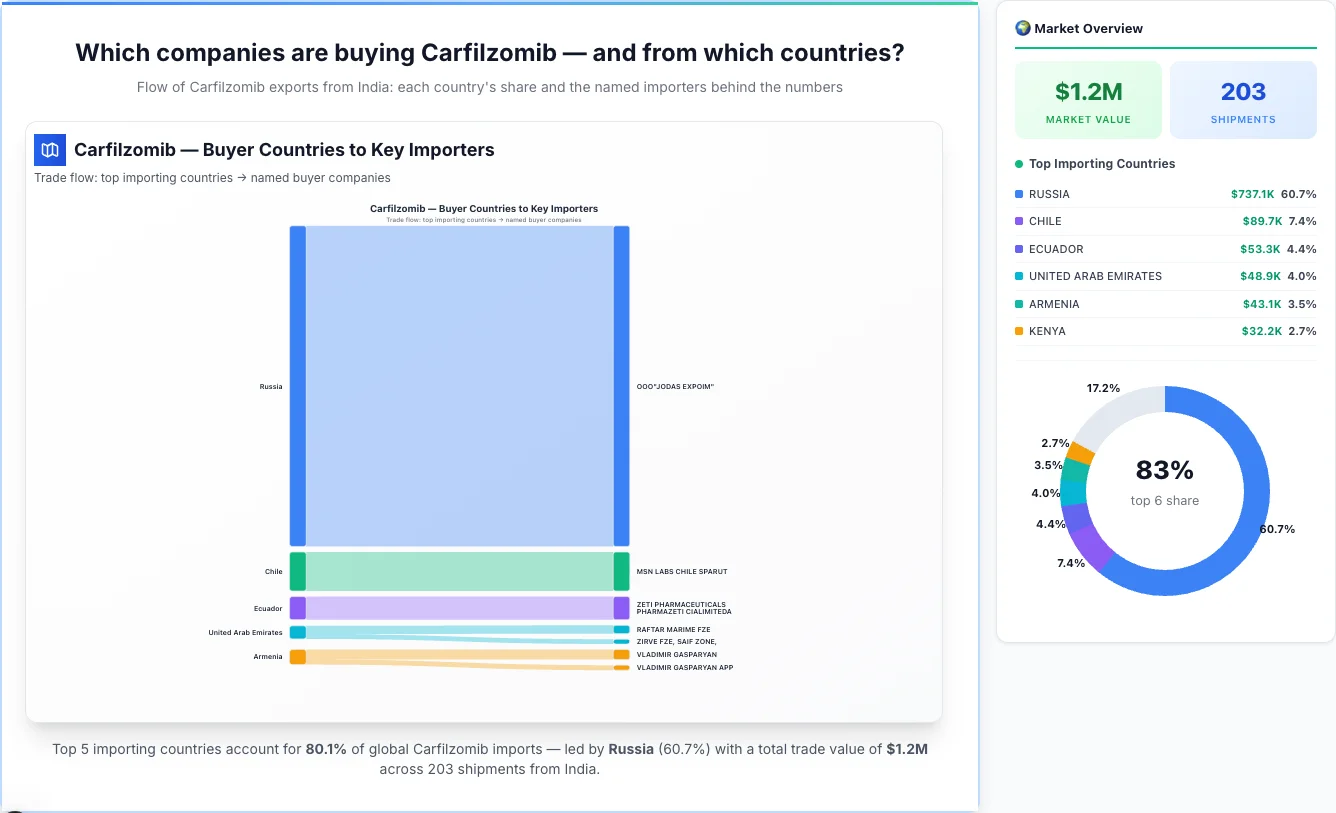
Which companies are buying Carfilzomib — and from which countries?
Flow of Carfilzomib exports from India: each country's share and the named importers behind the numbers
Carfilzomib — Buyer Countries to Key Importers
Trade flow: top importing countries → named buyer companies
Market Analysis: Top Importing Countries & Their Importers
1Russia
Russia emerges as the first largest importing country, achieving a total trade value of $737.1K through 0 shipments. This represents a market share of 60.7% of the total market activity, demonstrating their significant presence in the industry.
Key Insight: Russia demonstrates strong market positioning with an average shipment value of $737.1K, indicating premium pricing strategy compared to the market average of $6.0K.
Russia is a key market for Carfilzomib imports, representing 60.7% of total trade value.
2Chile
Chile emerges as the second largest importing country, achieving a total trade value of $89.7K through 0 shipments. This represents a market share of 7.4% of the total market activity, demonstrating their significant presence in the industry.
Key Insight: Chile demonstrates strong market positioning with an average shipment value of $89.7K, indicating premium pricing strategy compared to the market average of $6.0K.
Chile is a key market for Carfilzomib imports, representing 7.4% of total trade value.
3Ecuador
Ecuador emerges as the third largest importing country, achieving a total trade value of $53.3K through 2 shipments. This represents a market share of 4.4% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Ecuador has a diverse importer base, with their top importers including:
• ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDA - $53.3K (100.0% of country's total business)
Key Insight: Ecuador demonstrates strong market positioning with an average shipment value of $26.6K, indicating premium pricing strategy compared to the market average of $6.0K.
Ecuador is a key market for Carfilzomib imports, representing 4.4% of total trade value.
4United Arab Emirates
United Arab Emirates emerges as the fourth largest importing country, achieving a total trade value of $48.9K through 4 shipments. This represents a market share of 4.0% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: United Arab Emirates has a diverse importer base, with their top importers including:
• RAFTAR MARIME FZE - $19.2K (39.2% of country's total business)
Key Insight: United Arab Emirates demonstrates strong market positioning with an average shipment value of $12.2K, indicating premium pricing strategy compared to the market average of $6.0K.
United Arab Emirates is a key market for Carfilzomib imports, representing 4.0% of total trade value.
5Armenia
Armenia emerges as the fifth largest importing country, achieving a total trade value of $43.1K through 8 shipments. This represents a market share of 3.5% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Armenia has a diverse importer base, with their top importers including:
• VLADIMIR GASPARYAN - $23.2K (53.8% of country's total business)
• VLADIMIR GASPARYAN APP - $11.6K (27.0% of country's total business)
Key Insight: Armenia demonstrates strong market positioning with an average shipment value of $5.4K, indicating competitive pricing strategy compared to the market average of $6.0K.
Armenia is a key market for Carfilzomib imports, representing 3.5% of total trade value.
Buyer Segment Analysis
TransData Nexus Carfilzomib buyer market intelligence
1Buyer Segment Analysis
Carfilzomib, a proteasome inhibitor used in the treatment of multiple myeloma, has been exported from India to 47 countries, involving 105 buyers across 203 shipments totaling $1.2 million. The repeat buyer rate stands at 36.2%, indicating a moderate level of recurring demand.
Buyer Segments:
1. Distributors: Entities that purchase Carfilzomib for resale to healthcare providers. For instance, ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDA in Ecuador, the top buyer with purchases totaling $0.05 million, likely operates as a distributor, supplying the drug to local hospitals and clinics.
2. Hospital Groups: Large healthcare institutions procuring directly for patient treatment. While specific hospital groups are not identified in the top buyers, it's common for such entities to source medications directly to ensure supply chain reliability.
3. Retail Chains: Pharmacy chains that stock Carfilzomib for outpatient dispensing. The data does not specify retail chains among the top buyers, suggesting that purchases are predominantly made by larger entities.
4. Government Procurement: National health agencies or international organizations like UNICEF and the Global Fund that acquire medications for public health programs. The absence of such buyers in the top 10 suggests that Carfilzomib procurement may be more decentralized or handled through other channels in these regions.
5. Re-exporters: Companies that import Carfilzomib for the purpose of exporting to other markets. RAFTAR MARIME FZE in the United Arab Emirates, with purchases totaling $0.02 million, may function as a re-exporter, leveraging the UAE's strategic position as a trade hub.
6. Contract Manufacturers: Firms that produce Carfilzomib under contract for other companies. The provided data does not indicate contract manufacturers among the top buyers, implying that Indian exporters are supplying finished products directly to end-users or distributors.
Buying Behavior and Order Patterns:
- Distributors and Re-exporters: These buyers typically place bulk orders to achieve economies of scale, ensuring a steady supply to meet market demand. Their purchasing patterns are influenced by market demand forecasts and inventory turnover rates.
- Hospital Groups: When involved, they procure based on patient needs and treatment protocols, often engaging in long-term contracts to secure consistent supply.
- Retail Chains: If present, they would order smaller quantities more frequently, aligning with prescription trends and shelf-life considerations.
- Government Procurement: Such entities usually engage in large-scale tenders, focusing on cost-effectiveness and broad population coverage.
Country-Specific Import Regulations
TransData Nexus Carfilzomib buyer market intelligence
1Country-Specific Import Regulations
Understanding the regulatory landscape is crucial for market entry and compliance. Below is an overview of the import regulations for Carfilzomib in the top five importing countries:
1. Russia:
- Regulatory Authority: Ministry of Health of the Russian Federation.
- Import Registration Process: Requires submission of a registration dossier, including clinical trial data, quality documentation, and labeling information.
- Estimated Timeline: Approximately 12-18 months, depending on the completeness of the submission and any additional requirements.
- Import Tariff/Duty: Pharmaceutical products under HS Code 30049049 are subject to a standard import duty; specific rates should be confirmed with the Federal Customs Service.
2. Chile:
- Regulatory Authority: Instituto de Salud Pública de Chile (ISP).
- Import Registration Process: Submission of a technical dossier, including product specifications, manufacturing details, and clinical data.
- Estimated Timeline: Approximately 6-12 months, subject to the review process and any additional information requests.
- Import Tariff/Duty: Medicines classified under HS Code 30049049 may benefit from preferential tariffs under trade agreements; verification with Chilean Customs is recommended.
3. Ecuador:
- Regulatory Authority: Agencia Nacional de Regulación, Control y Vigilancia Sanitaria (ARCSA).
- Import Registration Process: Requires a comprehensive dossier submission, including quality, safety, and efficacy data.
- Estimated Timeline: Approximately 8-14 months, depending on the evaluation process and any additional requirements.
- Import Tariff/Duty: Pharmaceutical imports under HS Code 30049049 are subject to import duties; specific rates should be confirmed with Ecuadorian Customs.
4. United Arab Emirates:
- Regulatory Authority: Ministry of Health and Prevention (MOHAP).
- Import Registration Process: Submission of a registration application, including product details, manufacturing information, and clinical data.
- Estimated Timeline: Approximately 6-9 months, subject to the review process and any additional information requests.
- Import Tariff/Duty: Pharmaceutical products under HS Code 30049049 are generally exempt from import duties; confirmation with UAE Customs is advisable.
5. Armenia:
- Regulatory Authority: Ministry of Health of the Republic of Armenia.
- Import Registration Process: Requires submission of a registration dossier, including quality, safety, and efficacy data.
- Estimated Timeline: Approximately 9-12 months, depending on the completeness of the submission and any additional requirements.
- Import Tariff/Duty: Pharmaceutical imports under HS Code 30049049 are subject to import duties; specific rates should be confirmed with Armenian Customs.
Demand Drivers & Market Opportunity
TransData Nexus Carfilzomib buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Carfilzomib in importing countries is influenced by several factors:
1. Disease Prevalence: Multiple myeloma, the primary indication for Carfilzomib, has varying prevalence rates globally. Countries with higher incidence rates exhibit greater demand for effective treatments.
2. Government Healthcare Programs: National health initiatives aimed at improving cancer care accessibility contribute to increased procurement of oncology drugs. For example, countries expanding universal health coverage are more likely to include advanced therapies like Carfilzomib in their formularies.
3. WHO Essential Medicines List (EML): Inclusion of a drug in the WHO EML can influence national procurement decisions. While Carfilzomib is not currently listed, its therapeutic significance may prompt consideration for inclusion, thereby affecting demand.
4. Tender-Based Purchasing: Many countries utilize tender systems for pharmaceutical procurement, focusing on cost-effectiveness and supply reliability. Participation in such tenders can significantly impact market opportunities for Carfilzomib suppliers.
Given the presence of 105 buyers across 47 countries, the market for Carfilzomib demonstrates a broad international reach. Targeted engagement with healthcare providers, alignment with national health priorities, and compliance with regulatory requirements are essential strategies for capitalizing on these market opportunities.
Common Questions — Carfilzomib Buyers & Importers
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
QWho is the largest carfilzomib buyer importing from India?
Based on import volume and value, ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDA (ECUADOR) leads with $53.3K in imports and a 4.4% market share — the highest of any single carfilzomib importer. VLADIMIR GASPARYAN (ARMENIA) and EVEREST BANK LTD (NEPAL) are the next largest buyers.
QHow many companies buy carfilzomib from India?
There are 105 active carfilzomib buyers importing from India, with a combined market of $1.2M across 203 shipments to 47 countries. The top 5 buyers hold 10.4% of total import value, while the remaining 100 buyers handle the other 89.6%.
QWhich countries import the most carfilzomib from India?
The top importing countries for carfilzomib from India are Russia (60.7%), Chile (7.4%), Ecuador (4.4%), United Arab Emirates (4.0%), Armenia (3.5%). These markets represent the largest demand centres for Indian pharmaceutical exports of carfilzomib, with buyers ranging from government procurement agencies to private pharmaceutical distributors.
QWhat is the average import order value for carfilzomib from India?
The average import transaction value for carfilzomib from India is $6.0K, with an average unit price of $91.92 per unit. Order sizes vary significantly based on buyer type — government tenders tend to be larger while private distributors place more frequent smaller orders.
Buyer Segment Analysis
TransData Nexus Carfilzomib buyer market intelligence
1Buyer Segment Analysis
Carfilzomib, a proteasome inhibitor used in the treatment of multiple myeloma, has been exported from India to 47 countries, involving 105 buyers across 203 shipments totaling $1.2 million. The repeat buyer rate stands at 36.2%, indicating a moderate level of recurring demand.
Buyer Segments:
1. Distributors: Entities that purchase Carfilzomib for resale to healthcare providers. For instance, ZETI PHARMACEUTICALS PHARMAZETI CIALIMITEDA in Ecuador, the top buyer with purchases totaling $0.05 million, likely operates as a distributor, supplying the drug to local hospitals and clinics.
2. Hospital Groups: Large healthcare institutions procuring directly for patient treatment. While specific hospital groups are not identified in the top buyers, it's common for such entities to source medications directly to ensure supply chain reliability.
3. Retail Chains: Pharmacy chains that stock Carfilzomib for outpatient dispensing. The data does not specify retail chains among the top buyers, suggesting that purchases are predominantly made by larger entities.
4. Government Procurement: National health agencies or international organizations like UNICEF and the Global Fund that acquire medications for public health programs. The absence of such buyers in the top 10 suggests that Carfilzomib procurement may be more decentralized or handled through other channels in these regions.
5. Re-exporters: Companies that import Carfilzomib for the purpose of exporting to other markets. RAFTAR MARIME FZE in the United Arab Emirates, with purchases totaling $0.02 million, may function as a re-exporter, leveraging the UAE's strategic position as a trade hub.
6. Contract Manufacturers: Firms that produce Carfilzomib under contract for other companies. The provided data does not indicate contract manufacturers among the top buyers, implying that Indian exporters are supplying finished products directly to end-users or distributors.
Buying Behavior and Order Patterns:
- Distributors and Re-exporters: These buyers typically place bulk orders to achieve economies of scale, ensuring a steady supply to meet market demand. Their purchasing patterns are influenced by market demand forecasts and inventory turnover rates.
- Hospital Groups: When involved, they procure based on patient needs and treatment protocols, often engaging in long-term contracts to secure consistent supply.
- Retail Chains: If present, they would order smaller quantities more frequently, aligning with prescription trends and shelf-life considerations.
- Government Procurement: Such entities usually engage in large-scale tenders, focusing on cost-effectiveness and broad population coverage.
Country-Specific Import Regulations
TransData Nexus Carfilzomib buyer market intelligence
1Country-Specific Import Regulations
Understanding the regulatory landscape is crucial for market entry and compliance. Below is an overview of the import regulations for Carfilzomib in the top five importing countries:
1. Russia:
- Regulatory Authority: Ministry of Health of the Russian Federation.
- Import Registration Process: Requires submission of a registration dossier, including clinical trial data, quality documentation, and labeling information.
- Estimated Timeline: Approximately 12-18 months, depending on the completeness of the submission and any additional requirements.
- Import Tariff/Duty: Pharmaceutical products under HS Code 30049049 are subject to a standard import duty; specific rates should be confirmed with the Federal Customs Service.
2. Chile:
- Regulatory Authority: Instituto de Salud Pública de Chile (ISP).
- Import Registration Process: Submission of a technical dossier, including product specifications, manufacturing details, and clinical data.
- Estimated Timeline: Approximately 6-12 months, subject to the review process and any additional information requests.
- Import Tariff/Duty: Medicines classified under HS Code 30049049 may benefit from preferential tariffs under trade agreements; verification with Chilean Customs is recommended.
3. Ecuador:
- Regulatory Authority: Agencia Nacional de Regulación, Control y Vigilancia Sanitaria (ARCSA).
- Import Registration Process: Requires a comprehensive dossier submission, including quality, safety, and efficacy data.
- Estimated Timeline: Approximately 8-14 months, depending on the evaluation process and any additional requirements.
- Import Tariff/Duty: Pharmaceutical imports under HS Code 30049049 are subject to import duties; specific rates should be confirmed with Ecuadorian Customs.
4. United Arab Emirates:
- Regulatory Authority: Ministry of Health and Prevention (MOHAP).
- Import Registration Process: Submission of a registration application, including product details, manufacturing information, and clinical data.
- Estimated Timeline: Approximately 6-9 months, subject to the review process and any additional information requests.
- Import Tariff/Duty: Pharmaceutical products under HS Code 30049049 are generally exempt from import duties; confirmation with UAE Customs is advisable.
5. Armenia:
- Regulatory Authority: Ministry of Health of the Republic of Armenia.
- Import Registration Process: Requires submission of a registration dossier, including quality, safety, and efficacy data.
- Estimated Timeline: Approximately 9-12 months, depending on the completeness of the submission and any additional requirements.
- Import Tariff/Duty: Pharmaceutical imports under HS Code 30049049 are subject to import duties; specific rates should be confirmed with Armenian Customs.
Demand Drivers & Market Opportunity
TransData Nexus Carfilzomib buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Carfilzomib in importing countries is influenced by several factors:
1. Disease Prevalence: Multiple myeloma, the primary indication for Carfilzomib, has varying prevalence rates globally. Countries with higher incidence rates exhibit greater demand for effective treatments.
2. Government Healthcare Programs: National health initiatives aimed at improving cancer care accessibility contribute to increased procurement of oncology drugs. For example, countries expanding universal health coverage are more likely to include advanced therapies like Carfilzomib in their formularies.
3. WHO Essential Medicines List (EML): Inclusion of a drug in the WHO EML can influence national procurement decisions. While Carfilzomib is not currently listed, its therapeutic significance may prompt consideration for inclusion, thereby affecting demand.
4. Tender-Based Purchasing: Many countries utilize tender systems for pharmaceutical procurement, focusing on cost-effectiveness and supply reliability. Participation in such tenders can significantly impact market opportunities for Carfilzomib suppliers.
Given the presence of 105 buyers across 47 countries, the market for Carfilzomib demonstrates a broad international reach. Targeted engagement with healthcare providers, alignment with national health priorities, and compliance with regulatory requirements are essential strategies for capitalizing on these market opportunities.
Official References & Regulatory Resources
- European Medicines Agency
- WHO Essential Medicines
- Invest India — Pharma Sector
- India Trade Statistics (DGFT)
Verify import regulations and drug registration requirements with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Identification: 105 global importers of Carfilzomib identified from consignee fields in DGFT shipping bill records.
- 2.Import Value Analysis: Total import value aggregated from 203 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Geographic Distribution: Buyer imports distributed across 47 destination countries with market share per buyer calculated from capped values.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
203 Verified Shipments
105 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists