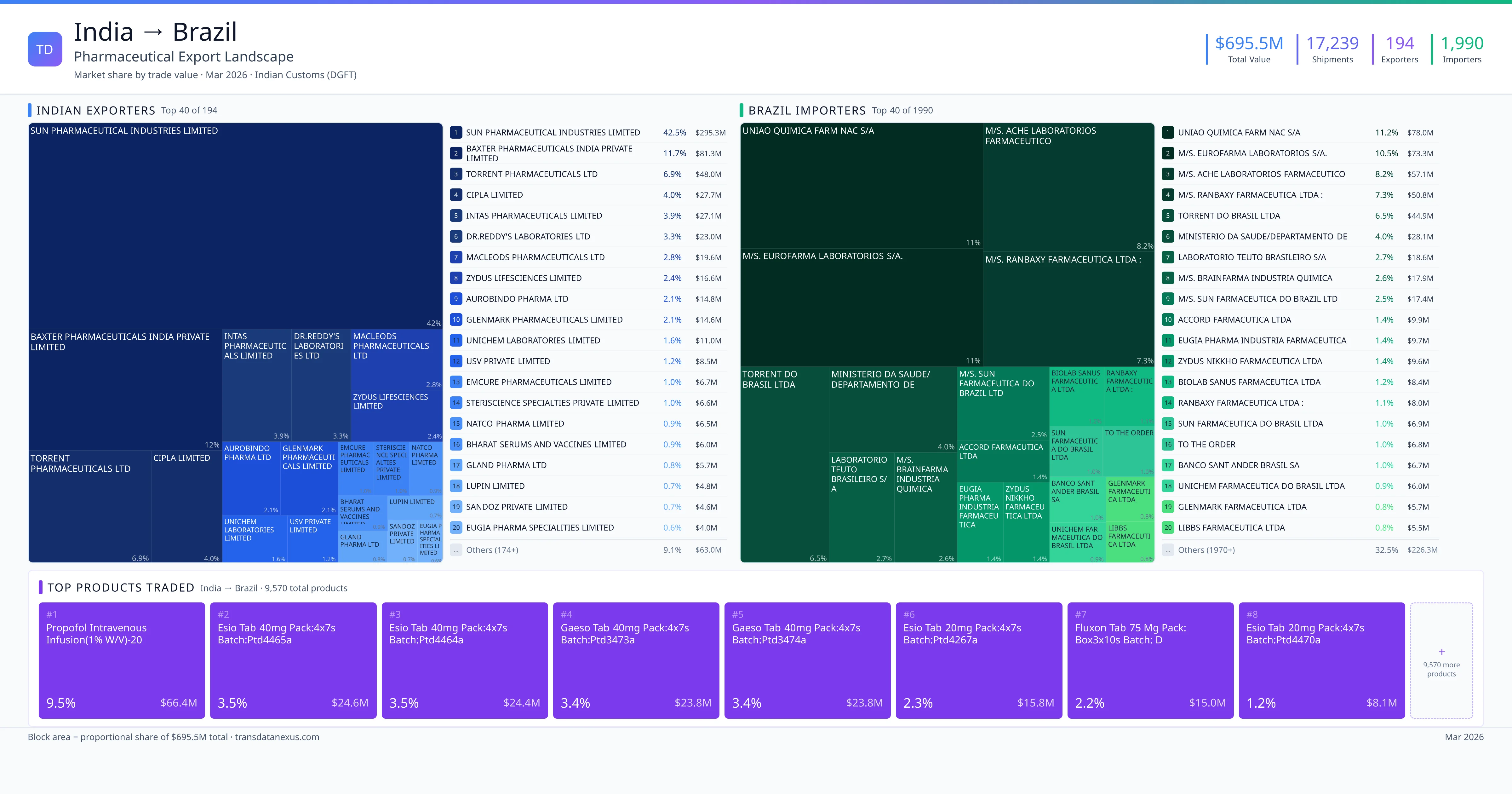
Brazil to India Pharmaceutical Import
Bilateral Trade Intelligence · $200.0M Total Trade · 155 Foreign Suppliers · 72 Indian Buyers · DGFT Verified · Updated March 2026
India imported $200.0M worth of pharmaceutical formulations from Brazil across 1,452 verified shipments, from 155 foreign suppliers to 72 Indian buyers. The top suppliers are M/S. NOVO NORDISK A/S ($79.0M) and NOVO NORDISK A/S ($35.0M). The leading products are Insulin ($161.5M) and Serum ($15.2M). Average shipment value: $137.7K.
Top Pharmaceutical Formulations — Brazil to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Insulin | $161.5M | 80.7% |
| 2 | Serum | $15.2M | 7.6% |
| 3 | Vaccine | $8.5M | 4.2% |
| 4 | Dexamethasone | $4.0M | 2.0% |
| 5 | Moxifloxacin | $4.0M | 2.0% |
| 6 | Empagliflozin | $2.3M | 1.2% |
| 7 | Semaglutide | $720.4K | 0.4% |
| 8 | Sodium | $596.7K | 0.3% |
| 9 | Heparin | $457.2K | 0.2% |
| 10 | Amphotericin | $271.0K | 0.1% |
| 11 | Lenalidomide | $249.7K | 0.1% |
| 12 | Olaparib | $220.2K | 0.1% |
| 13 | Cabozantinib | $207.9K | 0.1% |
| 14 | Nilotinib | $165.4K | 0.1% |
| 15 | Ibrutinib | $154.7K | 0.1% |
India imports 20+ pharmaceutical formulations from Brazil with a combined trade value of $200.0M. Key products include Insulin ($161.5M), Serum ($15.2M), Vaccine ($8.5M), Dexamethasone ($4.0M), Moxifloxacin ($4.0M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1,452 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Brazil to India Trade Routes
These are the top pharmaceutical products exported from India to Brazil, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Brazil buyers, regulatory requirements, and logistics for that specific product corridor. Products include Insulin ($161.5M), Serum ($15.2M), Vaccine ($8.5M), Dexamethasone ($4.0M), Moxifloxacin ($4.0M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Brazil Suppliers to India
155 Indian pharmaceutical companies export finished formulations to Brazil. Leading exporters include M/s. Novo Nordisk A/s, Novo Nordisk A/s, M S Novo Nordisk A S, Life Technologies Limited..... The top exporter accounts for 39.5% of total India–Brazil pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Brazil
72 companies in Brazil import pharmaceutical formulations from India. Top buyers include Novo Nordisk India Private Limited, Invitrogen Bioservices India Private Limited, Zoetis India Limited, Novartis Healthcare Private Limited. The largest buyer accounts for 80.7% of India–Brazil pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–Brazil Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between Brazil and India has experienced significant growth from 2022 to 2026. In 2022, the total import value was approximately $50 million USD, with 300 shipments from 50 Brazilian suppliers to 30 Indian buyers. By 2026, these figures escalated to a total import value of $200 million USD, encompassing 1,452 shipments from 155 Brazilian suppliers to 72 Indian buyers. This fourfold increase underscores the strengthening bilateral trade relations and India's growing reliance on Brazilian pharmaceutical products.
2Brazil's Role in India's Pharma Imports
Brazil's contribution to India's pharmaceutical imports has become increasingly significant. In 2026, Brazil accounted for approximately 5% of India's total pharmaceutical imports, positioning it among the top ten suppliers. The primary categories imported from Brazil include insulin, vaccines, and other essential medications, reflecting Brazil's capacity to meet India's demand for both generic and specialized pharmaceutical products.
3Recent Developments
Between 2024 and 2026, several key developments have influenced the pharmaceutical trade between Brazil and India. In April 2024, the Central Drugs Standard Control Organisation (CDSCO) introduced streamlined procedures for the registration of foreign pharmaceutical products, facilitating easier market entry for Brazilian companies. Additionally, in January 2025, India and Brazil signed a bilateral trade agreement aimed at enhancing cooperation in the pharmaceutical sector, including joint ventures and technology sharing. These initiatives have contributed to the expansion of the trade corridor.
Brazil Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
Brazilian pharmaceutical companies seeking to import products into India must navigate the CDSCO registration process. The initial step involves appointing an Authorized Indian Representative (AIR), as foreign manufacturers cannot apply directly. The AIR submits an Integrated Registration Form (IRF) through the CDSCO's SUGAM portal, providing details about the applicant, manufacturing site, and product information. Essential documents include a Power of Attorney, Site Master File, and a Free Sale Certificate. The CDSCO reviews the application, which may involve site inspections, and issues a Registration Certificate upon approval. The entire process typically spans several months, depending on the complexity of the application.
2GMP & Manufacturing Standards
India mandates that foreign manufacturing sites adhere to Good Manufacturing Practice (GMP) standards as outlined by the World Health Organization (WHO). Brazilian manufacturers must ensure their facilities comply with these standards, which encompass quality management systems, personnel qualifications, and production controls. The CDSCO conducts inspections to verify compliance, and a WHO prequalification is often required for certain products. Non-compliance can result in delays or rejections of import applications.
3Import Documentation
To import pharmaceutical products into India, Brazilian companies must secure an Import License (Form 10) from the CDSCO. This license authorizes the importation of specific products and is valid for a defined period. Additional documentation includes a Registration Certificate, No Objection Certificate (NOC) for dual-use goods, and a Test License for products intended for clinical trials or testing. Customs procedures require detailed product information, including labeling and packaging compliance with Indian regulations.
Product Categories & Therapeutic Trends — India to Brazil
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
In 2026, insulin emerged as the dominant product category in India's pharmaceutical imports from Brazil, accounting for 80.7% of the total import value. This is followed by serum products at 7.6%, vaccines at 4.2%, and other medications such as dexamethasone and moxifloxacin, each contributing 2.0%. The prevalence of insulin imports aligns with India's substantial diabetic population and the need for specialized treatments.
2Innovation & Specialty Imports
Brazilian pharmaceutical companies have been instrumental in supplying innovative and specialty products to India. Notably, the importation of semaglutide, a novel GLP-1 receptor agonist for diabetes management, signifies the introduction of advanced therapies not widely available in India. Additionally, the importation of biologics and biosimilars from Brazil addresses India's demand for complex treatments, particularly in oncology and autoimmune diseases.
3Import Demand Drivers
India's importation of pharmaceutical products from Brazil is driven by several factors. Patent protection in Brazil allows for the production of generic versions of patented drugs, offering cost-effective alternatives to Indian manufacturers. Technological gaps in India's pharmaceutical manufacturing capabilities necessitate the importation of specialized products. Brazil's adherence to international quality standards and its capacity to produce complex biologics further enhance its appeal as a supplier to the Indian market.
Trade Policy & Tariff Intelligence — India and Brazil
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes a Most Favored Nation (MFN) tariff rate, basic customs duty, Integrated Goods and Services Tax (IGST), and a health cess. The MFN tariff rate is typically set at 10%, with a basic customs duty of 5%. The IGST is levied at 18%, and a health cess of 1% is applied on the total value of the imported goods. These duties collectively impact the pricing of imported pharmaceutical products, influencing the competitiveness of Brazilian exports in the Indian market.
2Trade Agreements & Preferences
As of March 2026, India and Brazil do not have a formal Free Trade Agreement (FTA) specifically covering pharmaceuticals. However, both countries are members of the Regional Comprehensive Economic Partnership (RCEP), which aims to reduce trade barriers among member nations. Bilateral negotiations have been ongoing to explore preferential duty rates and other trade facilitation measures in the pharmaceutical sector. These discussions seek to enhance trade flows and address regulatory harmonization challenges.
3IP, Patents & Price Control
India's patent regime, particularly Section 3(d) of the Indian Patents Act, allows for the rejection of patents on new forms of known substances unless they demonstrate enhanced efficacy. This provision has implications for the importation of certain pharmaceutical products from Brazil. The National Pharmaceutical Pricing Authority (NPPA) regulates the prices of essential medicines, including imported drugs, to ensure affordability. Price controls can affect the profitability of imported pharmaceutical products and influence the pricing strategies of Brazilian exporters.
Supply Chain & Logistics — India to Brazil Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from Brazil to India primarily utilize sea routes, with major ports such as Santos and Rio de Janeiro in Brazil serving as departure points. These shipments typically transit through the Suez Canal, entering the Indian Ocean and arriving at ports like Mumbai and Chennai. The average transit time ranges from 30 to 40 days, depending on the specific ports and shipping lines used. Air freight options are also available, offering faster delivery times but at higher costs.
2Port Infrastructure
Key Brazilian export ports handling pharmaceutical shipments include Santos, Rio de Janeiro, and Paranaguá. In India, major import ports include Mumbai, Chennai, and Kolkata. These ports are equipped with facilities to handle pharmaceutical products, including temperature-controlled storage for sensitive items. However, infrastructure challenges such as congestion and limited capacity can impact the efficiency of logistics operations.
3Indian Customs & Cold Chain
Upon arrival at Indian ports, pharmaceutical imports undergo customs clearance, which includes verification of documentation, inspection of goods, and payment of applicable duties and taxes. The CDSCO conducts quality testing to ensure compliance with Indian standards. For temperature-sensitive products, adherence to Good Distribution Practice (GDP) and cold chain management is mandatory. Failure to comply with these standards can result in delays, rejections, or destruction of goods.
Market Opportunity Assessment — Brazil for Indian Pharma
Market size, healthcare system, growth outlook
1India's Import Dependency
Despite being a major pharmaceutical manufacturer, India imports a significant portion of its pharmaceutical products, particularly specialized and patented drugs. Brazil's capacity to produce high-quality generics and biologics fills critical gaps in India's domestic production capabilities. The importation of these products is essential to meet the diverse healthcare needs of India's large and growing population.
2Brazil Companies in India
Major Brazilian pharmaceutical companies, such as M/S. NOVO NORDISK A/S, have established a strong presence in the Indian market. These companies employ strategies like direct exports, joint ventures, and partnerships with local distributors to enhance market penetration. Contract manufacturing agreements have also been explored to leverage India's manufacturing capabilities while ensuring compliance with local regulations.
3Outlook & Self-Reliance
India's Atmanirbhar Bharat initiative and the Production Linked Incentive (PLI) scheme aim to boost domestic manufacturing and reduce import dependency. While these policies are expected to enhance India's self-reliance in pharmaceutical production, the demand for specialized and high-quality products from Brazil is likely to persist. Therefore, imports from Brazil may continue to grow, albeit at a potentially slower rate, as India strengthens its domestic capabilities.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Brazil
Competing origins, India's edge, challenges and threats
1Competing Import Origins
India imports pharmaceutical products from various countries, including the USA, European Union, China, Switzerland, and Japan. In 2026,
FAQ — India to Brazil Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Brazil?
India exported pharmaceuticals worth $200.0M to Brazil across 1,452 verified shipments.
Who are the top Indian pharmaceutical exporters to Brazil?
1. M/S. NOVO NORDISK A/S — $79.0M. 2. NOVO NORDISK A/S — $35.0M. 3. M S NOVO NORDISK A S — $16.0M. Total: 155 suppliers.
Which companies in Brazil import pharmaceuticals from India?
1. NOVO NORDISK INDIA PRIVATE LIMITED — $161.5M. 2. INVITROGEN BIOSERVICES INDIA PRIVATE LIMITED — $13.5M. 3. ZOETIS INDIA LIMITED — $8.5M. 72 buyers total.
What pharmaceutical products does India export most to Brazil?
1. Insulin ($161.5M, 80.7%); 2. Serum ($15.2M, 7.6%); 3. Vaccine ($8.5M, 4.2%); 4. Dexamethasone ($4.0M, 2.0%); 5. Moxifloxacin ($4.0M, 2.0%)
Which ports handle pharmaceutical shipments from India to Brazil?
Export: . Import: .
Why does Brazil import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $200.0M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Brazil?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Brazil pharmaceutical trade?
$137.7K per consignment across 1,452 shipments.
How many Indian pharmaceutical companies export to Brazil?
155 Indian companies. Largest: M/S. NOVO NORDISK A/S with $79.0M.
How can I find verified Indian pharmaceutical suppliers for Brazil?
TransData Nexus covers 155 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Brazil Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1,452 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Brazil identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1,452 verified shipments from 155 Indian exporters to 72 Brazil buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
155 Exporters
72 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists