Belgium to India Pharmaceutical Import
Bilateral Trade Intelligence · $217.6M Total Trade · 94 Foreign Suppliers · 78 Indian Buyers · DGFT Verified · Updated March 2026
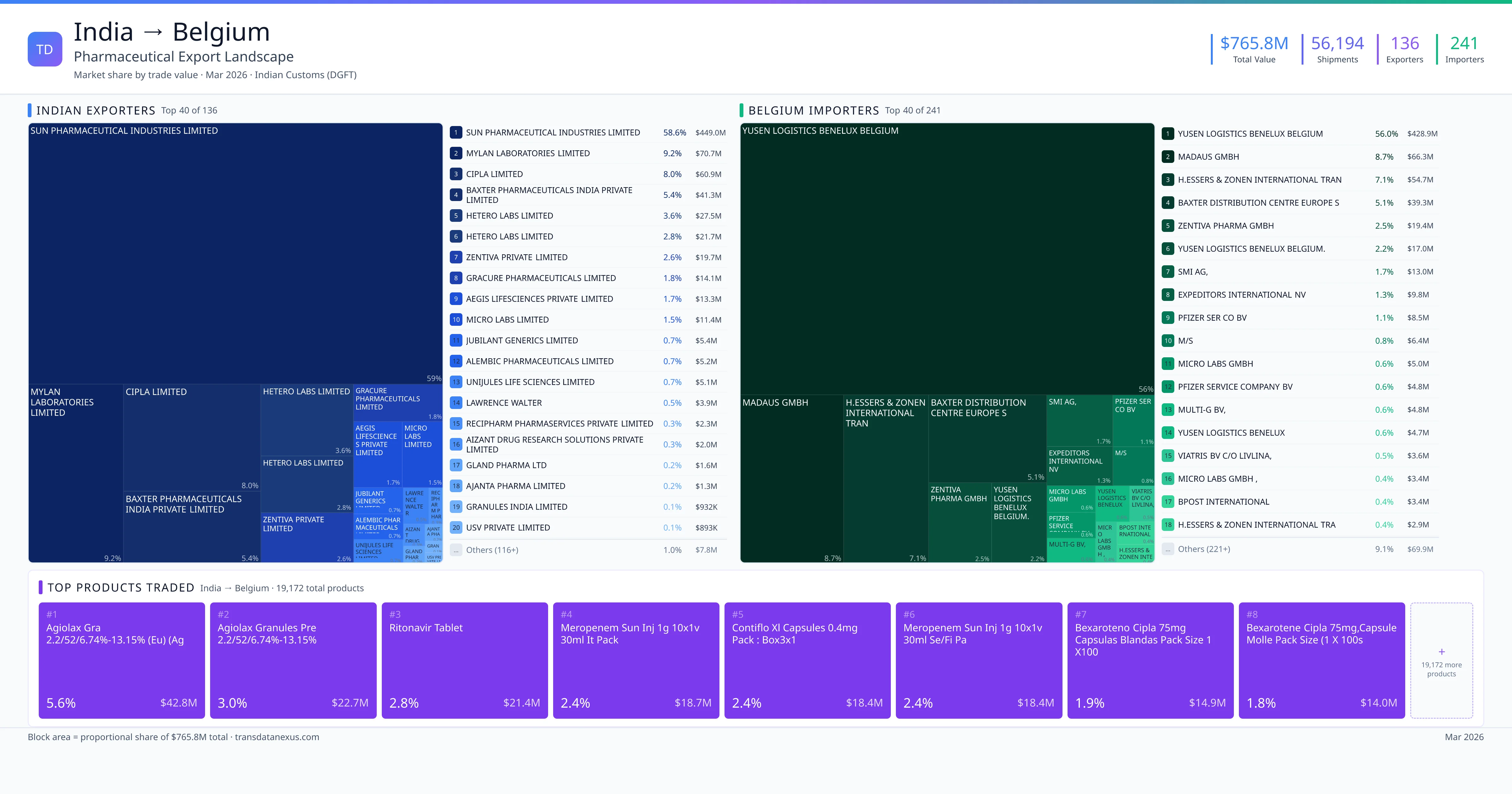
India imported $217.6M worth of pharmaceutical formulations from Belgium across 968 verified shipments, from 94 foreign suppliers to 78 Indian buyers. The top suppliers are PFIZER SERVICE COMPANY BV ($92.1M) and GLAXOSMITHKLINE BIOLOGICALS SA ($58.0M). The leading products are Vaccine ($111.5M) and Clindamycin ($33.5M). Average shipment value: $224.8K.
Top Pharmaceutical Formulations — Belgium to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Vaccine | $111.5M | 51.2% |
| 2 | Clindamycin | $33.5M | 15.4% |
| 3 | Sodium | $28.5M | 13.1% |
| 4 | Heparin | $14.0M | 6.4% |
| 5 | Prednisolone | $8.3M | 3.8% |
| 6 | Methylprednisolone | $8.3M | 3.8% |
| 7 | Daratumumab | $4.9M | 2.3% |
| 8 | Pembrolizumab | $888.8K | 0.4% |
| 9 | Cladribine | $823.6K | 0.4% |
| 10 | Sunitinib | $720.4K | 0.3% |
| 11 | Plasma | $717.5K | 0.3% |
| 12 | Doxorubicin | $508.7K | 0.2% |
| 13 | Etanercept | $457.7K | 0.2% |
| 14 | Ibrutinib | $450.7K | 0.2% |
| 15 | Pyrantel | $418.4K | 0.2% |
India imports 20+ pharmaceutical formulations from Belgium with a combined trade value of $217.6M. Key products include Vaccine ($111.5M), Clindamycin ($33.5M), Sodium ($28.5M), Heparin ($14.0M), Prednisolone ($8.3M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 968 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Belgium to India Trade Routes
These are the top pharmaceutical products exported from India to Belgium, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Belgium buyers, regulatory requirements, and logistics for that specific product corridor. Products include Vaccine ($111.5M), Clindamycin ($33.5M), Sodium ($28.5M), Heparin ($14.0M), Prednisolone ($8.3M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Belgium Suppliers to India
94 Indian pharmaceutical companies export finished formulations to Belgium. Leading exporters include Pfizer Service Company Bv, Glaxosmithkline Biologicals Sa, Janssen Pharmaceutica N.v., M S Glaxosmithkline Biologicals Sa. The top exporter accounts for 42.3% of total India–Belgium pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Belgium
78 companies in Belgium import pharmaceutical formulations from India. Top buyers include Glaxosmithkline Pharmaceuticals Limited, Pfizer Products India Private Limited, Pfizer Limited, Johnson & Johnson Private Limited. The largest buyer accounts for 37.9% of India–Belgium pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–Belgium Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between Belgium and India has experienced significant growth over the past decade. In 2022, the total import value stood at $217.6 million USD, with 968 shipments from 94 Belgian suppliers to 78 Indian buyers. This robust trade relationship underscores Belgium's pivotal role in supplying pharmaceutical products to the Indian market.
2Belgium's Role in India's Pharma Imports
Belgium has established itself as a key supplier in India's pharmaceutical import landscape. In 2022, Belgium's pharmaceutical exports to India were valued at $138.01 million USD, accounting for a substantial portion of India's total pharmaceutical imports. The primary categories imported include vaccines, antibiotics, and other essential medications, reflecting Belgium's diverse pharmaceutical offerings.
3Recent Developments
Between 2024 and 2026, the Central Drugs Standard Control Organisation (CDSCO) implemented several policy changes to streamline the import process. These included revisions to the registration procedures for foreign pharmaceutical products and updates to the guidelines for Good Manufacturing Practice (GMP) compliance. Additionally, India engaged in bilateral trade negotiations with Belgium to enhance the efficiency of pharmaceutical imports, focusing on reducing tariffs and simplifying customs procedures.
Belgium Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
Belgian pharmaceutical companies seeking to import products into India must navigate the CDSCO registration process. This involves submitting an Integrated Registration Form (IRF) through the CDSCO's SUGAM portal, accompanied by necessary documents such as a valid address proof of the firm and an authorized person's ID proof. The application undergoes verification by CDSCO Headquarters, and upon approval, a registration certificate is issued, authorizing the import and sale of the pharmaceutical products in India.
2GMP & Manufacturing Standards
India requires that foreign manufacturing sites adhere to Good Manufacturing Practice (GMP) standards as outlined by the World Health Organization (WHO). Belgian manufacturers must provide a GMP certificate in WHO format or a Certificate of Pharmaceutical Product (CPP) issued by the National Regulatory Authority of the country of origin. This certification ensures that the manufacturing processes meet the safety and quality standards mandated by Indian regulations.
3Import Documentation
To import pharmaceutical products into India, Belgian companies must obtain an import license (Form 10) from the CDSCO. This license is granted upon submission of a registration certificate, a No Objection Certificate (NOC), and a test license. The import process also involves compliance with customs procedures, including the submission of necessary documentation and adherence to import regulations to ensure the smooth clearance of goods at Indian ports.
Product Categories & Therapeutic Trends — India to Belgium
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
The pharmaceutical imports from Belgium to India are predominantly in the vaccine category, reflecting the high demand for immunization products in India. Other significant imports include antibiotics such as clindamycin and heparin, as well as corticosteroids like prednisolone and methylprednisolone. These products are essential in treating a range of medical conditions prevalent in India, highlighting Belgium's role in supplying critical medications.
2Innovation & Specialty Imports
Belgium exports several innovator drugs and novel formulations to India, including biologics like daratumumab and pembrolizumab. These advanced therapies are not manufactured domestically and are crucial in the treatment of complex diseases such as cancer. The importation of these specialty products underscores the importance of international collaboration in providing cutting-edge medical treatments to the Indian population.
3Import Demand Drivers
India's importation of pharmaceutical products from Belgium is driven by several factors, including the need for patented drugs not available domestically, technological advancements in drug manufacturing, and stringent quality requirements. The high disease burden in India necessitates access to a wide range of effective medications, making imports from Belgium a vital component of India's healthcare system.
Trade Policy & Tariff Intelligence — India and Belgium
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes the Most Favored Nation (MFN) tariff rates, basic customs duty, Integrated Goods and Services Tax (IGST), and health cess. These tariffs are subject to change based on government policies and trade agreements. Importers must stay informed about the current tariff rates to ensure compliance and optimize import costs.
2Trade Agreements & Preferences
India and Belgium have engaged in bilateral trade negotiations to enhance the efficiency of pharmaceutical imports. These discussions focus on reducing tariffs, simplifying customs procedures, and addressing regulatory harmonization to facilitate smoother trade flows between the two countries.
3IP, Patents & Price Control
India's patent regime, particularly Section 3(d) of the Indian Patents Act, aims to prevent evergreening of patents and ensures that only genuine innovations receive patent protection. The National Pharmaceutical Pricing Authority (NPPA) regulates the prices of essential drugs, including imported pharmaceuticals, to make them affordable for the Indian population. These policies impact the pricing and availability of imported drugs from Belgium.
Supply Chain & Logistics — India to Belgium Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from Belgium to India primarily utilize sea routes, with major ports such as Antwerp and Zeebrugge in Belgium serving as export points. The transit time for sea shipments to Indian ports like Mumbai and Chennai typically ranges from 20 to 30 days, depending on the shipping line and specific route taken. Air freight is also utilized for time-sensitive deliveries, with transit times of approximately 7 to 10 days.
2Port Infrastructure
Belgium's key export ports include Antwerp and Zeebrugge, both equipped with advanced facilities to handle pharmaceutical shipments. In India, major import ports such as Mumbai, Chennai, and Kolkata are equipped with specialized infrastructure to manage pharmaceutical imports, ensuring compliance with regulatory standards and efficient processing of goods.
3Indian Customs & Cold Chain
Indian customs procedures for pharmaceutical imports involve detailed documentation and adherence to regulatory requirements set by the CDSCO. Imported pharmaceuticals are subject to testing by the CDSCO to ensure compliance with safety and efficacy standards. Maintaining the cold chain is critical for temperature-sensitive products, and importers must ensure that their logistics partners adhere to Good Distribution Practice (GDP) guidelines to preserve product integrity throughout the supply chain.
Market Opportunity Assessment — Belgium for Indian Pharma
Market size, healthcare system, growth outlook
1India's Import Dependency
Despite being a major pharmaceutical manufacturer, India imports a significant portion of its pharmaceutical products from Belgium due to the need for patented drugs, advanced biologics, and specialized formulations not produced domestically. This dependency highlights the importance of international trade in meeting India's diverse healthcare needs.
2Belgium Companies in India
Major Belgian pharmaceutical companies, such as Pfizer Service Company BV and GlaxoSmithKline Biologicals SA, supply a range of products to the Indian market. These companies often engage in local partnerships and contract manufacturing to enhance their market presence and cater to the specific needs of Indian consumers.
3Outlook & Self-Reliance
India's Atmanirbhar Bharat initiative and the Production-Linked Incentive (PLI) scheme aim to boost domestic manufacturing capabilities, potentially reducing the reliance on pharmaceutical imports. However, the demand for specialized and patented drugs may continue to sustain imports from Belgium, albeit at a potentially reduced scale as domestic production increases.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Belgium
Competing origins, India's edge, challenges and threats
1Competing Import Origins
India imports pharmaceutical products from various countries, including the USA, other EU nations, China, Switzerland, and Japan. Belgium's share in India's pharmaceutical imports is significant, but it faces competition from these countries, each offering unique products and advantages.
2Belgium's Competitive Advantages
Belgian pharmaceutical products are favored in India due to their innovation, adherence to high-quality manufacturing standards, and strong brand reputation. The country's compliance with international regulatory standards and its focus on research and development contribute to the appeal of its products in the Indian market.
3Domestic Competition & Threats
Indian manufacturers are increasingly developing alternatives to imported pharmaceuticals, including biosimilars and generic versions of patented drugs. The PLI scheme has incentivized domestic production, potentially reducing the market share for imported products. However, the complexity of certain biologics and specialized formulations may continue to necessitate imports from countries like Belgium.
FAQ — India to Belgium Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Belgium?
India exported pharmaceuticals worth $217.6M to Belgium across 968 verified shipments.
Who are the top Indian pharmaceutical exporters to Belgium?
1. PFIZER SERVICE COMPANY BV — $92.1M. 2. GLAXOSMITHKLINE BIOLOGICALS SA — $58.0M. 3. JANSSEN PHARMACEUTICA N.V. — $5.6M. Total: 94 suppliers.
Which companies in Belgium import pharmaceuticals from India?
1. GLAXOSMITHKLINE PHARMACEUTICALS LIMITED — $82.5M. 2. PFIZER PRODUCTS INDIA PRIVATE LIMITED — $80.3M. 3. PFIZER LIMITED — $15.6M. 78 buyers total.
What pharmaceutical products does India export most to Belgium?
1. Vaccine ($111.5M, 51.2%); 2. Clindamycin ($33.5M, 15.4%); 3. Sodium ($28.5M, 13.1%); 4. Heparin ($14.0M, 6.4%); 5. Prednisolone ($8.3M, 3.8%)
Which ports handle pharmaceutical shipments from India to Belgium?
Export: . Import: .
Why does Belgium import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $217.6M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Belgium?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Belgium pharmaceutical trade?
$224.8K per consignment across 968 shipments.
How many Indian pharmaceutical companies export to Belgium?
94 Indian companies. Largest: PFIZER SERVICE COMPANY BV with $92.1M.
How can I find verified Indian pharmaceutical suppliers for Belgium?
TransData Nexus covers 94 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Belgium Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 968 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Belgium identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 968 verified shipments from 94 Indian exporters to 78 Belgium buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
94 Exporters
78 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists