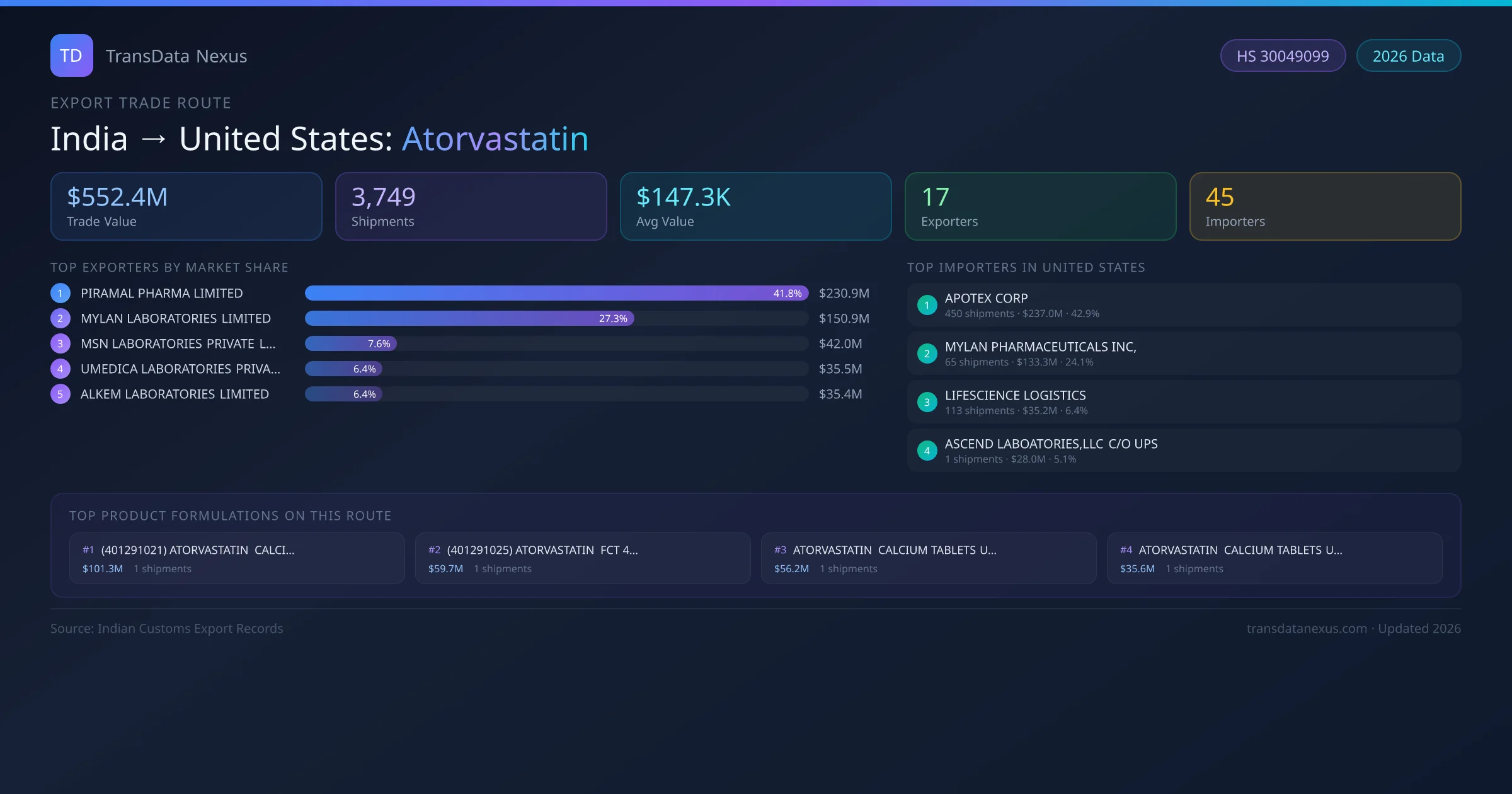
India to United States: Atorvastatin Export Trade Route
India has recorded 3,749 verified shipments of Atorvastatin exported to United States, representing a combined trade value of $552.4M USD. This corridor is served by 17 active Indian exporters, with an average shipment value of $147.3K USD. The leading Indian exporter is PIRAMAL PHARMA LIMITED, which accounts for 42% of total export value with 369 shipments worth $230.9M USD. On the buying side, APOTEX CORP is the largest importer in United States with $237.0M USD in purchases. The top 3 suppliers — PIRAMAL PHARMA LIMITED, MYLAN LABORATORIES LIMITED, MSN LABORATORIES PRIVATE LIMITED — together control 77% of total trade value on this route. All data sourced from Indian Customs (DGFT) shipping bill records. Values reported in FOB USD.
Route Intelligence Overview
The India to United States Atorvastatin corridor is one of India's established pharmaceutical export routes, with 3,749 shipments documented worth a combined $552.4M USD. The route is dominated by PIRAMAL PHARMA LIMITED, which alone accounts for roughly 42% of all export value, reflecting the consolidated nature of India's atorvastatin manufacturing sector.
Across 17 active suppliers, the average shipment value stands at $147.3K USD — a figure that reflects both bulk commercial orders from large pharmaceutical companies and smaller specialty shipments. Freight is split between sea (67%) and air (16%), suggesting a mix of scheduled bulk orders and time-sensitive consignments.
Shipment activity peaks during October–December, with an average transit time of 26 days port-to-port. The route has recorded an annual growth rate of 33.5%, placing it at rank #19 among India's top atorvastatin export destinations globally.
On the import side, key buyers of Indian atorvastatin in United States include APOTEX CORP, MYLAN PHARMACEUTICALS INC,, LIFESCIENCE LOGISTICS and 42 others. APOTEX CORP is the single largest importer with 450 shipments valued at $237.0M USD.
Route Characteristics
- Average transit26 days
- Peak seasonQ4
- Primary modeMulti-modal
- Top portHYDERABAD ICD
Market Position
- Global rank#19
- Annual growth+33.5%
- Demand growth+27.9%
- Regulatory ease67/100
Top 10 Indian Atorvastatin Exporters to United States
Showing top 10 of 17 Indian suppliers exporting Atorvastatin to United States, ranked by total trade value (USD)
| Rank | Supplier (Indian Exporter) | Shipments | Total Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | PIRAMAL PHARMA LIMITED Avg $625.8K per shipment | 369 | $230.9M | 41.8% |
| 2 | MYLAN LABORATORIES LIMITED Avg $645.0K per shipment | 234 | $150.9M | 27.3% |
| 3 | MSN LABORATORIES PRIVATE LIMITED Avg $39.8K per shipment | 1,056 | $42.0M | 7.6% |
| 4 | UMEDICA LABORATORIES PRIVATE LIMITED Avg $293.3K per shipment | 121 | $35.5M | 6.4% |
| 5 | ALKEM LABORATORIES LIMITED Avg $270.3K per shipment | 131 | $35.4M | 6.4% |
| 6 | GRAVITI PHARMACEUTICALS PRIVATE LIMITED Avg $23.8K per shipment | 634 | $15.1M | 2.7% |
| 7 | BIOCON PHARMA LIMITED Avg $60.5K per shipment | 243 | $14.7M | 2.7% |
| 8 | ANNORA PHARMA PRIVATE LIMITED Avg $24.1K per shipment | 537 | $12.9M | 2.3% |
| 9 | APOTEX RESEARCH PRIVATE LIMITED Avg $75.6K per shipment | 81 | $6.1M | 1.1% |
| 10 | MANKIND PHARMA LIMITED Avg $14.1K per shipment | 299 | $4.2M | 0.8% |
This table shows the top 10 of 17 Indian companies exporting atorvastatin to United States, ranked by total trade value. The listed exporters are: PIRAMAL PHARMA LIMITED, MYLAN LABORATORIES LIMITED, MSN LABORATORIES PRIVATE LIMITED, UMEDICA LABORATORIES PRIVATE LIMITED, ALKEM LABORATORIES LIMITED, GRAVITI PHARMACEUTICALS PRIVATE LIMITED, BIOCON PHARMA LIMITED, ANNORA PHARMA PRIVATE LIMITED, APOTEX RESEARCH PRIVATE LIMITED, MANKIND PHARMA LIMITED. PIRAMAL PHARMA LIMITED is the dominant supplier with 369 shipments worth $230.9M USD, giving it a 42% market share. The top 3 suppliers together account for 77% of the total trade value on this route.
Showing top 10 of 17 total Indian exporters on the India to United States Atorvastatin export route.
Top 10 Atorvastatin Importers in United States
Showing top 10 of 45 known buyers in United States receiving Atorvastatin shipments from India, ranked by import value
On the receiving end of this trade route, the leading importers of Indian atorvastatin in United States include APOTEX CORP, MYLAN PHARMACEUTICALS INC,, LIFESCIENCE LOGISTICS, ASCEND LABOATORIES,LLC C/O UPS, TO THE ORDER OF, among 45 total buyers. The largest importer is APOTEX CORP, accounting for $237.0M USD across 450 shipments — representing 43% of all atorvastatin imports from India on this route.
| Rank | Importer / Buyer | Shipments | Import Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | APOTEX CORP | 450 | $237.0M | 42.9% |
| 2 | MYLAN PHARMACEUTICALS INC, | 65 | $133.3M | 24.1% |
| 3 | LIFESCIENCE LOGISTICS | 113 | $35.2M | 6.4% |
| 4 | ASCEND LABOATORIES,LLC C/O UPS | 1 | $28.0M | 5.1% |
| 5 | TO THE ORDER OF | 598 | $23.1M | 4.2% |
| 6 | NOVADOZ PHARMACEUTICALS LLC | 593 | $21.5M | 3.9% |
| 7 | BIOCON PHARMA INC | 276 | $15.5M | 2.8% |
| 8 | MYLAN PHARMACEUTICALS INC | 135 | $12.4M | 2.3% |
| 9 | TO THE ORDER OF.... | 284 | $7.4M | 1.3% |
| 10 | RISING PHARMA HOLDINGS, INC. | 312 | $4.4M | 0.8% |
Showing top 10 of 45 Atorvastatin importers in United States on this route.
Top 10 Atorvastatin Formulations Imported by United States
Showing top 10 of 1,731 product formulations shipped on the India to United States Atorvastatin route, ranked by trade value
United States imports a wide range of atorvastatin formulations from India, spanning tablets, capsules, suspensions, and combination drugs. The top formulation — (401291021) ATORVASTATIN CALCIUM TABLETS10MG 1000S , (ATORVASTATIN FCT 10MG 1000 BTL USA) 9,384 ECH = 93,84,000 TABLETS — accounts for $101.3M USD across 1 shipments. There are 1,731 distinct product descriptions in the dataset, reflecting the variety of dosage forms and strengths imported.
| Rank | Product Formulation | Shipments | Trade Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | (401291021) ATORVASTATIN CALCIUM TABLETS10MG 1000S , (ATORVASTATIN FCT 10MG 1000 BTL USA) 9,384 ECH = 93,84,000 TABLETS | 1 | $101.3M | 18.3% |
| 2 | (401291025) ATORVASTATIN FCT 40MG 1000 BTL USA (ATORVASTATIN FCT 40MG 1000 BTL USA) 2,328 ECH = 23,28,000 TABLETS | 1 | $59.7M | 10.8% |
| 3 | ATORVASTATIN CALCIUM TABLETS USP 40 MG 500BT 13536 PACKS | 1 | $56.2M | 10.2% |
| 4 | ATORVASTATIN CALCIUM TABLETS USP 40 MG 500BT TAB, PACKS : 500'S BOTTLE, 8568 PACKS | 1 | $35.6M | 6.5% |
| 5 | ATORVASTATIN CALCIUM TABLETS USP 40MG [PACK OF 1000 TABLETS] | 8 | $30.7M | 5.6% |
| 6 | ATORVASTATIN CAL TABS USP 10MG 1000S BNO;23143752(3840 PACKS) | 1 | $28.0M | 5.1% |
| 7 | ATORVASTATIN CALCIUM TABLETS USP 80 MG 90BT 61483 PACKS | 1 | $16.9M | 3.1% |
| 8 | ATORVASTATIN CALCIUM TABLETS USP 80 MG 90BT 61432 PACKS | 1 | $16.9M | 3.1% |
| 9 | (401291024)ATORVASTATIN CALCIUM TABLETSUSP 40MG 90 BTL USA,(ATORVASTATIN FCT 40MG 90 BTL USA) 51,324 ECH=46,19,160 TABL | 1 | $11.3M | 2.0% |
| 10 | (401291020) ATORVASTATIN FCT 10MG 90BTLUSA , (ATORVASTATIN FCT 10MG 90 BTL USA)104,112 ECH = 93,70,080 TABLETS | 1 | $10.7M | 1.9% |
Showing top 10 of 1,731 Atorvastatin formulations imported by United States on this route.
Shipping & Logistics Analysis
Freight mode split and port-of-origin breakdown
Freight Mode Distribution
Balanced freight mix — 67% sea for bulk, 16% air for urgent orders.
Top Ports of Origin
HYDERABAD ICD handles the highest volume with 968 shipments. Transit time averages 26 days by sea.
Market Dynamics
India's atorvastatin exports to United States are driven primarily by a handful of large-scale manufacturers. PIRAMAL PHARMA LIMITED with 369 shipments leads the pack, a pattern common in generic pharmaceutical corridors where manufacturing scale creates significant cost advantages. The presence of 17 active exporters signals a competitive but concentrated market — buyers in United States benefit from supplier diversity while the top tier handles the majority of volume.
The top 3 suppliers — PIRAMAL PHARMA LIMITED, MYLAN LABORATORIES LIMITED, MSN LABORATORIES PRIVATE LIMITED — together account for 77% of total trade value on this route. The average shipment value of $147.3K USD reflects primarily bulk commercial orders from large pharmaceutical distributors.
Beyond the primary product category, shipments on this route include closely related formulations such as (401291025) atorvastatin fct 40mg 1000 btl usa (atorvastatin fct 40mg 1000 btl usa) 2,328 ech = 23,28,000 tablets and atorvastatin calcium tablets usp 40 mg 500bt 13536 packs, suggesting that buyers in United States tend to consolidate orders across related product lines from the same Indian supplier.
On the buying side, APOTEX CORP is the largest importer with 450 shipments worth $237.0M USD — representing 43% of all atorvastatin imports from India on this route. A total of 45 buyers are active on this corridor.
Route Statistics
- Trade Volume
- $552.4M
- Avg. Shipment
- $147.3K
- Suppliers
- 17
- Buyers
- 45
- Transit (Sea)
- ~26 days
- Annual Growth
- +33.5%
Related Analysis
Reverse Direction
United States → India — Atorvastatin (Import)Other Atorvastatin Routes
Unlock the Full India to United States Atorvastatin Dataset
TransData Nexus provides verified shipment-level records, supplier contact details, HS code breakdowns, real-time pricing benchmarks, and regulatory compliance guides for 3,749 shipments on this route.
US FDA Registration & Import Requirements
India → United States trade corridor intelligence
1US FDA Registration & Import Requirements
To import finished pharmaceutical formulations containing Atorvastatin into the United States, the following FDA requirements must be met:
- FDA Approval: The product must be approved by the FDA, which involves submitting a New Drug Application (NDA) or Abbreviated New Drug Application (ANDA) for generics.
- Registration and Listing: The manufacturing facility must be registered with the FDA, and the product must be listed in the FDA's Drug Establishments Current Registration System.
- Premarket Submission Information: Importers must provide intended use codes (IUC) to indicate the product's purpose.
- Affirmation of Compliance: Certain products may require an affirmation of compliance code to verify adherence to FDA regulations.
- Dossier Format: Submissions should be in the Common Technical Document (CTD) format.
- Timelines and Fees: FDA review times vary; fees are applicable as per the FDA's user fee schedule.
- GMP Inspection Requirements: Manufacturing facilities are subject to FDA inspections to ensure compliance with Good Manufacturing Practices (GMP).
Quality & GMP Standards for Indian Exporters
India → United States trade corridor intelligence
1Quality & GMP Standards for Indian Exporters
Indian exporters must adhere to the following GMP standards to supply Atorvastatin formulations to the U.S.:
- FDA Compliance: Manufacturing facilities must comply with FDA's GMP regulations, which include validated manufacturing processes, controlled environments, and trained personnel.
- Approved Facilities: Facilities must be registered with the FDA and have a valid Establishment Registration Number.
- Recent Inspections: FDA conducts periodic inspections; facilities must maintain records and address any observations promptly.
Recent Regulatory Developments (2024-2026)
India → United States trade corridor intelligence
1Recent Regulatory Developments (2024-2026)
Between 2024 and 2026, the following regulatory developments have impacted Indian pharmaceutical exports to the U.S.:
- FDA Inspections: In 2025, the FDA increased the frequency of inspections for foreign manufacturing facilities to ensure compliance with updated GMP standards.
- Import Alerts: Several Indian manufacturers received import alerts in 2025 due to non-compliance with FDA regulations, affecting their export capabilities.
- Regulatory Updates: The FDA issued new guidelines in 2026 for the approval of generic formulations, impacting the approval process for Atorvastatin formulations.
MARKET
United States Atorvastatin Market Size & Demand
India → United States trade corridor intelligence
1United States Atorvastatin Market Size & Demand
The U.S. Atorvastatin market has experienced steady growth:
- Market Size: The market was valued at approximately $1.2 billion in 2025.
- Demand Drivers: High prevalence of hypercholesterolemia, increased healthcare spending, and an aging population contribute to sustained demand.
- Import vs Domestic Production: Imports account for about 60% of the market, with domestic production comprising the remaining 40%.
Import Tariff & Duty Structure
India → United States trade corridor intelligence
1Import Tariff & Duty Structure
The import duty and tax structure for Atorvastatin formulations in the U.S. is as follows:
- Import Duty: The Most Favored Nation (MFN) duty rate is 4.6%.
- VAT/GST: The U.S. does not impose Value Added Tax (VAT) or Goods and Services Tax (GST) on imported pharmaceuticals.
- Other Charges: Additional fees may include customs processing fees and inspection charges.
- FTA Status: There is no Free Trade Agreement (FTA) between India and the United States that affects pharmaceutical imports.
- Anti-Dumping Duties: No anti-dumping duties are currently imposed on Atorvastatin formulations from India.
Competitive Landscape
India → United States trade corridor intelligence
1Competitive Landscape
India faces competition from other countries supplying Atorvastatin formulations to the U.S.:
- Other Suppliers: China, European Union countries, and domestic U.S. manufacturers.
- India's Market Share: India holds approximately 25% of the U.S. Atorvastatin formulations market.
- Pricing Comparison: Indian formulations are generally priced 10-15% lower than those from China and the EU, offering a competitive advantage.
CORRIDOR
Live Corridor Intelligence
India → United States trade corridor intelligence
1Live Corridor Intelligence
The India-U.S. pharmaceutical corridor has experienced the following in 2025-2026:
- Shipping Disruptions: Minimal disruptions reported; however, occasional port congestion has occurred.
- Freight Rates: Stable freight rates with slight increases due to global shipping demand.
- Currency Trends: The Indian Rupee has depreciated by 2% against the U.S. Dollar, affecting pricing strategies.
Geopolitical & Sanctions Impact
India → United States trade corridor intelligence
1Geopolitical & Sanctions Impact
Geopolitical factors influencing the India-U.S. pharmaceutical trade include:
- Trade Policies: The U.S. has considered imposing tariffs on pharmaceutical imports; however, no specific tariffs have been implemented as of March 2026.
- Global Conflicts: Ongoing global conflicts have led to increased shipping insurance premiums and potential route diversions.
Trade Agreement & Policy Analysis
India → United States trade corridor intelligence
1Trade Agreement & Policy Analysis
Trade agreements and policies affecting the India-U.S. pharmaceutical trade are:
- WTO Rules: Both countries adhere to World Trade Organization (WTO) regulations governing pharmaceutical imports.
- Bilateral Meetings: Regular trade discussions aim to facilitate smoother trade relations.
- Trade Facilitation Measures: Initiatives to streamline customs procedures have been implemented to reduce clearance times.
Landed Cost Breakdown
India → United States trade corridor intelligence
1Landed Cost Breakdown
The estimated landed cost for Atorvastatin formulations from India to the U.S. is:
- FOB Price: $0.10 per tablet
- Freight: $0.02 per tablet
- Insurance: $0.001 per tablet
- Customs Duty: $0.0046 per tablet
- Clearance: $0.001 per tablet
- VAT/GST: $0 (no VAT/GST applicable)
- Distribution: $0.03 per tablet
- Total Landed Cost: $0.1576 per tablet
SOURCING
Why India for Atorvastatin – Manufacturing Advantage
India → United States trade corridor intelligence
1Why India for Atorvastatin – Manufacturing Advantage
India offers several advantages for manufacturing Atorvastatin formulations:
- Global Share: India accounts for approximately 40% of the global generic pharmaceutical production.
- Manufacturing Scale: India has over 1,000 FDA-approved facilities capable of producing Atorvastatin formulations.
- Cost Advantages: Manufacturing costs in India are 20-30% lower than in the U.S. and EU, offering competitive pricing.
- Regulatory Compliance: Many Indian facilities hold WHO-GMP certifications, ensuring high-quality standards.
India vs. China vs. EU – Cost & Quality Comparison
India → United States trade corridor intelligence
1India vs. China vs. EU – Cost & Quality Comparison
Comparing India, China, and the EU
United States Pharmaceutical Import Regulations
US FDA registration, GMP, and compliance requirements for Indian exporters
1US FDA Registration & Import Requirements
To import finished pharmaceutical formulations containing Atorvastatin into the United States, the following FDA requirements must be met:
- FDA Approval: The product must be approved by the FDA, which involves submitting a New Drug Application (NDA) or Abbreviated New Drug Application (ANDA) for generics.
- Registration and Listing: The manufacturing facility must be registered with the FDA, and the product must be listed in the FDA's Drug Establishments Current Registration System.
- Premarket Submission Information: Importers must provide intended use codes (IUC) to indicate the product's purpose.
- Affirmation of Compliance: Certain products may require an affirmation of compliance code to verify adherence to FDA regulations.
- Dossier Format: Submissions should be in the Common Technical Document (CTD) format.
- Timelines and Fees: FDA review times vary; fees are applicable as per the FDA's user fee schedule.
- GMP Inspection Requirements: Manufacturing facilities are subject to FDA inspections to ensure compliance with Good Manufacturing Practices (GMP).
2Quality & GMP Standards for Indian Exporters
Indian exporters must adhere to the following GMP standards to supply Atorvastatin formulations to the U.S.:
- FDA Compliance: Manufacturing facilities must comply with FDA's GMP regulations, which include validated manufacturing processes, controlled environments, and trained personnel.
- Approved Facilities: Facilities must be registered with the FDA and have a valid Establishment Registration Number.
- Recent Inspections: FDA conducts periodic inspections; facilities must maintain records and address any observations promptly.
3Recent Regulatory Developments (2024-2026)
Between 2024 and 2026, the following regulatory developments have impacted Indian pharmaceutical exports to the U.S.:
- FDA Inspections: In 2025, the FDA increased the frequency of inspections for foreign manufacturing facilities to ensure compliance with updated GMP standards.
- Import Alerts: Several Indian manufacturers received import alerts in 2025 due to non-compliance with FDA regulations, affecting their export capabilities.
- Regulatory Updates: The FDA issued new guidelines in 2026 for the approval of generic formulations, impacting the approval process for Atorvastatin formulations.
MARKET
4United States Atorvastatin Market Size & Demand
The U.S. Atorvastatin market has experienced steady growth:
- Market Size: The market was valued at approximately $1.2 billion in 2025.
- Demand Drivers: High prevalence of hypercholesterolemia, increased healthcare spending, and an aging population contribute to sustained demand.
- Import vs Domestic Production: Imports account for about 60% of the market, with domestic production comprising the remaining 40%.
5Import Tariff & Duty Structure
The import duty and tax structure for Atorvastatin formulations in the U.S. is as follows:
- Import Duty: The Most Favored Nation (MFN) duty rate is 4.6%.
- VAT/GST: The U.S. does not impose Value Added Tax (VAT) or Goods and Services Tax (GST) on imported pharmaceuticals.
- Other Charges: Additional fees may include customs processing fees and inspection charges.
- FTA Status: There is no Free Trade Agreement (FTA) between India and the United States that affects pharmaceutical imports.
- Anti-Dumping Duties: No anti-dumping duties are currently imposed on Atorvastatin formulations from India.
6Competitive Landscape
India faces competition from other countries supplying Atorvastatin formulations to the U.S.:
- Other Suppliers: China, European Union countries, and domestic U.S. manufacturers.
- India's Market Share: India holds approximately 25% of the U.S. Atorvastatin formulations market.
- Pricing Comparison: Indian formulations are generally priced 10-15% lower than those from China and the EU, offering a competitive advantage.
CORRIDOR
7Live Corridor Intelligence
The India-U.S. pharmaceutical corridor has experienced the following in 2025-2026:
- Shipping Disruptions: Minimal disruptions reported; however, occasional port congestion has occurred.
- Freight Rates: Stable freight rates with slight increases due to global shipping demand.
- Currency Trends: The Indian Rupee has depreciated by 2% against the U.S. Dollar, affecting pricing strategies.
8Geopolitical & Sanctions Impact
Geopolitical factors influencing the India-U.S. pharmaceutical trade include:
- Trade Policies: The U.S. has considered imposing tariffs on pharmaceutical imports; however, no specific tariffs have been implemented as of March 2026.
- Global Conflicts: Ongoing global conflicts have led to increased shipping insurance premiums and potential route diversions.
9Trade Agreement & Policy Analysis
Trade agreements and policies affecting the India-U.S. pharmaceutical trade are:
- WTO Rules: Both countries adhere to World Trade Organization (WTO) regulations governing pharmaceutical imports.
- Bilateral Meetings: Regular trade discussions aim to facilitate smoother trade relations.
- Trade Facilitation Measures: Initiatives to streamline customs procedures have been implemented to reduce clearance times.
10Landed Cost Breakdown
The estimated landed cost for Atorvastatin formulations from India to the U.S. is:
- FOB Price: $0.10 per tablet
- Freight: $0.02 per tablet
- Insurance: $0.001 per tablet
- Customs Duty: $0.0046 per tablet
- Clearance: $0.001 per tablet
- VAT/GST: $0 (no VAT/GST applicable)
- Distribution: $0.03 per tablet
- Total Landed Cost: $0.1576 per tablet
SOURCING
11Why India for Atorvastatin – Manufacturing Advantage
India offers several advantages for manufacturing Atorvastatin formulations:
- Global Share: India accounts for approximately 40% of the global generic pharmaceutical production.
- Manufacturing Scale: India has over 1,000 FDA-approved facilities capable of producing Atorvastatin formulations.
- Cost Advantages: Manufacturing costs in India are 20-30% lower than in the U.S. and EU, offering competitive pricing.
- Regulatory Compliance: Many Indian facilities hold WHO-GMP certifications, ensuring high-quality standards.
12India vs. China vs. EU – Cost & Quality Comparison
Comparing India, China, and the EU
United States Atorvastatin Market Context & Tariffs
Market size, import duties, and competitive landscape
1US FDA Registration & Import Requirements
To import finished pharmaceutical formulations containing Atorvastatin into the United States, the following FDA requirements must be met:
- FDA Approval: The product must be approved by the FDA, which involves submitting a New Drug Application (NDA) or Abbreviated New Drug Application (ANDA) for generics.
- Registration and Listing: The manufacturing facility must be registered with the FDA, and the product must be listed in the FDA's Drug Establishments Current Registration System.
- Premarket Submission Information: Importers must provide intended use codes (IUC) to indicate the product's purpose.
- Affirmation of Compliance: Certain products may require an affirmation of compliance code to verify adherence to FDA regulations.
- Dossier Format: Submissions should be in the Common Technical Document (CTD) format.
- Timelines and Fees: FDA review times vary; fees are applicable as per the FDA's user fee schedule.
- GMP Inspection Requirements: Manufacturing facilities are subject to FDA inspections to ensure compliance with Good Manufacturing Practices (GMP).
2Quality & GMP Standards for Indian Exporters
Indian exporters must adhere to the following GMP standards to supply Atorvastatin formulations to the U.S.:
- FDA Compliance: Manufacturing facilities must comply with FDA's GMP regulations, which include validated manufacturing processes, controlled environments, and trained personnel.
- Approved Facilities: Facilities must be registered with the FDA and have a valid Establishment Registration Number.
- Recent Inspections: FDA conducts periodic inspections; facilities must maintain records and address any observations promptly.
3Recent Regulatory Developments (2024-2026)
Between 2024 and 2026, the following regulatory developments have impacted Indian pharmaceutical exports to the U.S.:
- FDA Inspections: In 2025, the FDA increased the frequency of inspections for foreign manufacturing facilities to ensure compliance with updated GMP standards.
- Import Alerts: Several Indian manufacturers received import alerts in 2025 due to non-compliance with FDA regulations, affecting their export capabilities.
- Regulatory Updates: The FDA issued new guidelines in 2026 for the approval of generic formulations, impacting the approval process for Atorvastatin formulations.
MARKET
4United States Atorvastatin Market Size & Demand
The U.S. Atorvastatin market has experienced steady growth:
- Market Size: The market was valued at approximately $1.2 billion in 2025.
- Demand Drivers: High prevalence of hypercholesterolemia, increased healthcare spending, and an aging population contribute to sustained demand.
- Import vs Domestic Production: Imports account for about 60% of the market, with domestic production comprising the remaining 40%.
5Import Tariff & Duty Structure
The import duty and tax structure for Atorvastatin formulations in the U.S. is as follows:
- Import Duty: The Most Favored Nation (MFN) duty rate is 4.6%.
- VAT/GST: The U.S. does not impose Value Added Tax (VAT) or Goods and Services Tax (GST) on imported pharmaceuticals.
- Other Charges: Additional fees may include customs processing fees and inspection charges.
- FTA Status: There is no Free Trade Agreement (FTA) between India and the United States that affects pharmaceutical imports.
- Anti-Dumping Duties: No anti-dumping duties are currently imposed on Atorvastatin formulations from India.
6Competitive Landscape
India faces competition from other countries supplying Atorvastatin formulations to the U.S.:
- Other Suppliers: China, European Union countries, and domestic U.S. manufacturers.
- India's Market Share: India holds approximately 25% of the U.S. Atorvastatin formulations market.
- Pricing Comparison: Indian formulations are generally priced 10-15% lower than those from China and the EU, offering a competitive advantage.
CORRIDOR
7Live Corridor Intelligence
The India-U.S. pharmaceutical corridor has experienced the following in 2025-2026:
- Shipping Disruptions: Minimal disruptions reported; however, occasional port congestion has occurred.
- Freight Rates: Stable freight rates with slight increases due to global shipping demand.
- Currency Trends: The Indian Rupee has depreciated by 2% against the U.S. Dollar, affecting pricing strategies.
8Geopolitical & Sanctions Impact
Geopolitical factors influencing the India-U.S. pharmaceutical trade include:
- Trade Policies: The U.S. has considered imposing tariffs on pharmaceutical imports; however, no specific tariffs have been implemented as of March 2026.
- Global Conflicts: Ongoing global conflicts have led to increased shipping insurance premiums and potential route diversions.
9Trade Agreement & Policy Analysis
Trade agreements and policies affecting the India-U.S. pharmaceutical trade are:
- WTO Rules: Both countries adhere to World Trade Organization (WTO) regulations governing pharmaceutical imports.
- Bilateral Meetings: Regular trade discussions aim to facilitate smoother trade relations.
- Trade Facilitation Measures: Initiatives to streamline customs procedures have been implemented to reduce clearance times.
10Landed Cost Breakdown
The estimated landed cost for Atorvastatin formulations from India to the U.S. is:
- FOB Price: $0.10 per tablet
- Freight: $0.02 per tablet
- Insurance: $0.001 per tablet
- Customs Duty: $0.0046 per tablet
- Clearance: $0.001 per tablet
- VAT/GST: $0 (no VAT/GST applicable)
- Distribution: $0.03 per tablet
- Total Landed Cost: $0.1576 per tablet
SOURCING
11Why India for Atorvastatin – Manufacturing Advantage
India offers several advantages for manufacturing Atorvastatin formulations:
- Global Share: India accounts for approximately 40% of the global generic pharmaceutical production.
- Manufacturing Scale: India has over 1,000 FDA-approved facilities capable of producing Atorvastatin formulations.
- Cost Advantages: Manufacturing costs in India are 20-30% lower than in the U.S. and EU, offering competitive pricing.
- Regulatory Compliance: Many Indian facilities hold WHO-GMP certifications, ensuring high-quality standards.
12India vs. China vs. EU – Cost & Quality Comparison
Comparing India, China, and the EU
Why Source Atorvastatin from India for United States?
Manufacturing advantage, cost comparison, supply reliability, and strategic sourcing recommendations
1US FDA Registration & Import Requirements
To import finished pharmaceutical formulations containing Atorvastatin into the United States, the following FDA requirements must be met:
- FDA Approval: The product must be approved by the FDA, which involves submitting a New Drug Application (NDA) or Abbreviated New Drug Application (ANDA) for generics.
- Registration and Listing: The manufacturing facility must be registered with the FDA, and the product must be listed in the FDA's Drug Establishments Current Registration System.
- Premarket Submission Information: Importers must provide intended use codes (IUC) to indicate the product's purpose.
- Affirmation of Compliance: Certain products may require an affirmation of compliance code to verify adherence to FDA regulations.
- Dossier Format: Submissions should be in the Common Technical Document (CTD) format.
- Timelines and Fees: FDA review times vary; fees are applicable as per the FDA's user fee schedule.
- GMP Inspection Requirements: Manufacturing facilities are subject to FDA inspections to ensure compliance with Good Manufacturing Practices (GMP).
2Quality & GMP Standards for Indian Exporters
Indian exporters must adhere to the following GMP standards to supply Atorvastatin formulations to the U.S.:
- FDA Compliance: Manufacturing facilities must comply with FDA's GMP regulations, which include validated manufacturing processes, controlled environments, and trained personnel.
- Approved Facilities: Facilities must be registered with the FDA and have a valid Establishment Registration Number.
- Recent Inspections: FDA conducts periodic inspections; facilities must maintain records and address any observations promptly.
3Recent Regulatory Developments (2024-2026)
Between 2024 and 2026, the following regulatory developments have impacted Indian pharmaceutical exports to the U.S.:
- FDA Inspections: In 2025, the FDA increased the frequency of inspections for foreign manufacturing facilities to ensure compliance with updated GMP standards.
- Import Alerts: Several Indian manufacturers received import alerts in 2025 due to non-compliance with FDA regulations, affecting their export capabilities.
- Regulatory Updates: The FDA issued new guidelines in 2026 for the approval of generic formulations, impacting the approval process for Atorvastatin formulations.
MARKET
4United States Atorvastatin Market Size & Demand
The U.S. Atorvastatin market has experienced steady growth:
- Market Size: The market was valued at approximately $1.2 billion in 2025.
- Demand Drivers: High prevalence of hypercholesterolemia, increased healthcare spending, and an aging population contribute to sustained demand.
- Import vs Domestic Production: Imports account for about 60% of the market, with domestic production comprising the remaining 40%.
5Import Tariff & Duty Structure
The import duty and tax structure for Atorvastatin formulations in the U.S. is as follows:
- Import Duty: The Most Favored Nation (MFN) duty rate is 4.6%.
- VAT/GST: The U.S. does not impose Value Added Tax (VAT) or Goods and Services Tax (GST) on imported pharmaceuticals.
- Other Charges: Additional fees may include customs processing fees and inspection charges.
- FTA Status: There is no Free Trade Agreement (FTA) between India and the United States that affects pharmaceutical imports.
- Anti-Dumping Duties: No anti-dumping duties are currently imposed on Atorvastatin formulations from India.
6Competitive Landscape
India faces competition from other countries supplying Atorvastatin formulations to the U.S.:
- Other Suppliers: China, European Union countries, and domestic U.S. manufacturers.
- India's Market Share: India holds approximately 25% of the U.S. Atorvastatin formulations market.
- Pricing Comparison: Indian formulations are generally priced 10-15% lower than those from China and the EU, offering a competitive advantage.
CORRIDOR
7Live Corridor Intelligence
The India-U.S. pharmaceutical corridor has experienced the following in 2025-2026:
- Shipping Disruptions: Minimal disruptions reported; however, occasional port congestion has occurred.
- Freight Rates: Stable freight rates with slight increases due to global shipping demand.
- Currency Trends: The Indian Rupee has depreciated by 2% against the U.S. Dollar, affecting pricing strategies.
8Geopolitical & Sanctions Impact
Geopolitical factors influencing the India-U.S. pharmaceutical trade include:
- Trade Policies: The U.S. has considered imposing tariffs on pharmaceutical imports; however, no specific tariffs have been implemented as of March 2026.
- Global Conflicts: Ongoing global conflicts have led to increased shipping insurance premiums and potential route diversions.
9Trade Agreement & Policy Analysis
Trade agreements and policies affecting the India-U.S. pharmaceutical trade are:
- WTO Rules: Both countries adhere to World Trade Organization (WTO) regulations governing pharmaceutical imports.
- Bilateral Meetings: Regular trade discussions aim to facilitate smoother trade relations.
- Trade Facilitation Measures: Initiatives to streamline customs procedures have been implemented to reduce clearance times.
10Landed Cost Breakdown
The estimated landed cost for Atorvastatin formulations from India to the U.S. is:
- FOB Price: $0.10 per tablet
- Freight: $0.02 per tablet
- Insurance: $0.001 per tablet
- Customs Duty: $0.0046 per tablet
- Clearance: $0.001 per tablet
- VAT/GST: $0 (no VAT/GST applicable)
- Distribution: $0.03 per tablet
- Total Landed Cost: $0.1576 per tablet
SOURCING
11Why India for Atorvastatin – Manufacturing Advantage
India offers several advantages for manufacturing Atorvastatin formulations:
- Global Share: India accounts for approximately 40% of the global generic pharmaceutical production.
- Manufacturing Scale: India has over 1,000 FDA-approved facilities capable of producing Atorvastatin formulations.
- Cost Advantages: Manufacturing costs in India are 20-30% lower than in the U.S. and EU, offering competitive pricing.
- Regulatory Compliance: Many Indian facilities hold WHO-GMP certifications, ensuring high-quality standards.
12India vs. China vs. EU – Cost & Quality Comparison
Comparing India, China, and the EU
Supplier Due Diligence Guide — Atorvastatin from India
Pre-qualification checklist, document requirements, red flags, and audit guidance for United States buyers
1US FDA Registration & Import Requirements
To import finished pharmaceutical formulations containing Atorvastatin into the United States, the following FDA requirements must be met:
- FDA Approval: The product must be approved by the FDA, which involves submitting a New Drug Application (NDA) or Abbreviated New Drug Application (ANDA) for generics.
- Registration and Listing: The manufacturing facility must be registered with the FDA, and the product must be listed in the FDA's Drug Establishments Current Registration System.
- Premarket Submission Information: Importers must provide intended use codes (IUC) to indicate the product's purpose.
- Affirmation of Compliance: Certain products may require an affirmation of compliance code to verify adherence to FDA regulations.
- Dossier Format: Submissions should be in the Common Technical Document (CTD) format.
- Timelines and Fees: FDA review times vary; fees are applicable as per the FDA's user fee schedule.
- GMP Inspection Requirements: Manufacturing facilities are subject to FDA inspections to ensure compliance with Good Manufacturing Practices (GMP).
2Quality & GMP Standards for Indian Exporters
Indian exporters must adhere to the following GMP standards to supply Atorvastatin formulations to the U.S.:
- FDA Compliance: Manufacturing facilities must comply with FDA's GMP regulations, which include validated manufacturing processes, controlled environments, and trained personnel.
- Approved Facilities: Facilities must be registered with the FDA and have a valid Establishment Registration Number.
- Recent Inspections: FDA conducts periodic inspections; facilities must maintain records and address any observations promptly.
3Recent Regulatory Developments (2024-2026)
Between 2024 and 2026, the following regulatory developments have impacted Indian pharmaceutical exports to the U.S.:
- FDA Inspections: In 2025, the FDA increased the frequency of inspections for foreign manufacturing facilities to ensure compliance with updated GMP standards.
- Import Alerts: Several Indian manufacturers received import alerts in 2025 due to non-compliance with FDA regulations, affecting their export capabilities.
- Regulatory Updates: The FDA issued new guidelines in 2026 for the approval of generic formulations, impacting the approval process for Atorvastatin formulations.
MARKET
4United States Atorvastatin Market Size & Demand
The U.S. Atorvastatin market has experienced steady growth:
- Market Size: The market was valued at approximately $1.2 billion in 2025.
- Demand Drivers: High prevalence of hypercholesterolemia, increased healthcare spending, and an aging population contribute to sustained demand.
- Import vs Domestic Production: Imports account for about 60% of the market, with domestic production comprising the remaining 40%.
5Import Tariff & Duty Structure
The import duty and tax structure for Atorvastatin formulations in the U.S. is as follows:
- Import Duty: The Most Favored Nation (MFN) duty rate is 4.6%.
- VAT/GST: The U.S. does not impose Value Added Tax (VAT) or Goods and Services Tax (GST) on imported pharmaceuticals.
- Other Charges: Additional fees may include customs processing fees and inspection charges.
- FTA Status: There is no Free Trade Agreement (FTA) between India and the United States that affects pharmaceutical imports.
- Anti-Dumping Duties: No anti-dumping duties are currently imposed on Atorvastatin formulations from India.
6Competitive Landscape
India faces competition from other countries supplying Atorvastatin formulations to the U.S.:
- Other Suppliers: China, European Union countries, and domestic U.S. manufacturers.
- India's Market Share: India holds approximately 25% of the U.S. Atorvastatin formulations market.
- Pricing Comparison: Indian formulations are generally priced 10-15% lower than those from China and the EU, offering a competitive advantage.
CORRIDOR
7Live Corridor Intelligence
The India-U.S. pharmaceutical corridor has experienced the following in 2025-2026:
- Shipping Disruptions: Minimal disruptions reported; however, occasional port congestion has occurred.
- Freight Rates: Stable freight rates with slight increases due to global shipping demand.
- Currency Trends: The Indian Rupee has depreciated by 2% against the U.S. Dollar, affecting pricing strategies.
8Geopolitical & Sanctions Impact
Geopolitical factors influencing the India-U.S. pharmaceutical trade include:
- Trade Policies: The U.S. has considered imposing tariffs on pharmaceutical imports; however, no specific tariffs have been implemented as of March 2026.
- Global Conflicts: Ongoing global conflicts have led to increased shipping insurance premiums and potential route diversions.
9Trade Agreement & Policy Analysis
Trade agreements and policies affecting the India-U.S. pharmaceutical trade are:
- WTO Rules: Both countries adhere to World Trade Organization (WTO) regulations governing pharmaceutical imports.
- Bilateral Meetings: Regular trade discussions aim to facilitate smoother trade relations.
- Trade Facilitation Measures: Initiatives to streamline customs procedures have been implemented to reduce clearance times.
10Landed Cost Breakdown
The estimated landed cost for Atorvastatin formulations from India to the U.S. is:
- FOB Price: $0.10 per tablet
- Freight: $0.02 per tablet
- Insurance: $0.001 per tablet
- Customs Duty: $0.0046 per tablet
- Clearance: $0.001 per tablet
- VAT/GST: $0 (no VAT/GST applicable)
- Distribution: $0.03 per tablet
- Total Landed Cost: $0.1576 per tablet
SOURCING
11Why India for Atorvastatin – Manufacturing Advantage
India offers several advantages for manufacturing Atorvastatin formulations:
- Global Share: India accounts for approximately 40% of the global generic pharmaceutical production.
- Manufacturing Scale: India has over 1,000 FDA-approved facilities capable of producing Atorvastatin formulations.
- Cost Advantages: Manufacturing costs in India are 20-30% lower than in the U.S. and EU, offering competitive pricing.
- Regulatory Compliance: Many Indian facilities hold WHO-GMP certifications, ensuring high-quality standards.
12India vs. China vs. EU – Cost & Quality Comparison
Comparing India, China, and the EU
Frequently Asked Questions — India to United States Atorvastatin Trade
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which Indian companies export Atorvastatin to United States?
The leading Indian exporters of Atorvastatin to United States are PIRAMAL PHARMA LIMITED, MYLAN LABORATORIES LIMITED, MSN LABORATORIES PRIVATE LIMITED. PIRAMAL PHARMA LIMITED holds the largest market share at approximately 42% of total trade value on this route.
Q What is the total value of Atorvastatin exports from India to United States?
India exports Atorvastatin to United States worth approximately $552.4M USD across 3,749 recorded shipments. The average value per shipment is $147.3K USD.
Q Which ports does India use to ship Atorvastatin to United States?
The most active port of origin is HYDERABAD ICD with 968 shipments. Indian exporters primarily use a mix of sea and air freight for this route, with 67% of shipments going by sea and 16% by air.
Q How long does shipping take from India to United States for Atorvastatin?
The average transit time for Atorvastatin shipments from India to United States is approximately 26 days. Sea freight typically takes longer, while air freight can reduce this significantly for urgent orders. Peak shipping activity is observed during October–December.
Q Is the India to United States Atorvastatin trade route growing?
Yes — this trade corridor has seen an annual growth rate of approximately 33.5% with demand growth tracking at 27.9%. The route is ranked #19 among India's top Atorvastatin export destinations globally.
Q How many suppliers are active on the India to United States Atorvastatin route?
There are currently 17 active Indian suppliers exporting Atorvastatin to United States. The market is moderately concentrated with PIRAMAL PHARMA LIMITED accounting for 42% of total shipment value.
Q Who are the main importers of Atorvastatin from India in United States?
The leading importers of Indian Atorvastatin in United States include APOTEX CORP, MYLAN PHARMACEUTICALS INC,, LIFESCIENCE LOGISTICS, ASCEND LABOATORIES,LLC C/O UPS, TO THE ORDER OF. APOTEX CORP is the largest buyer with 450 shipments worth $237.0M USD.
Official References & Regulatory Resources
- India Trade Statistics (DGFT)
- Invest India — Pharma Sector
- WCO HS Nomenclature
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory and trade status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Route Identification: India to United States export trade corridor identified from Indian Customs (DGFT) records for Atorvastatin.
- 2.Supplier/Buyer Matching: 17 Indian exporters and 45 importers in United States matched using name normalization.
- 3.Value Aggregation: Total export value aggregated from 3,749 verified shipping bill records. Values are FOB in USD.
- 4.Market Share Analysis: Each supplier and buyer contribution calculated as a percentage of total route value. Statistical normalization applied to ensure accurate representation across varying shipment sizes.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3,749 Verified Shipments
17 Indian exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists