Loading...
Atorvastatin
Who Supplies Atorvastatin to India from GERMANY
India's atorvastatin imports from GERMANY total $40.8K across 27 shipments from 8 foreign suppliers. NEWLIFE BIOPHARMA LIMITED leads with $11.6K in import value; the top 5 suppliers together control 93.8% of this origin. Leading Indian buyers include HETERO LABS LIMITED. This corridor reflects India's pharmaceutical import demand for atorvastatin — a diversified sourcing base with multiple active suppliers from GERMANY.
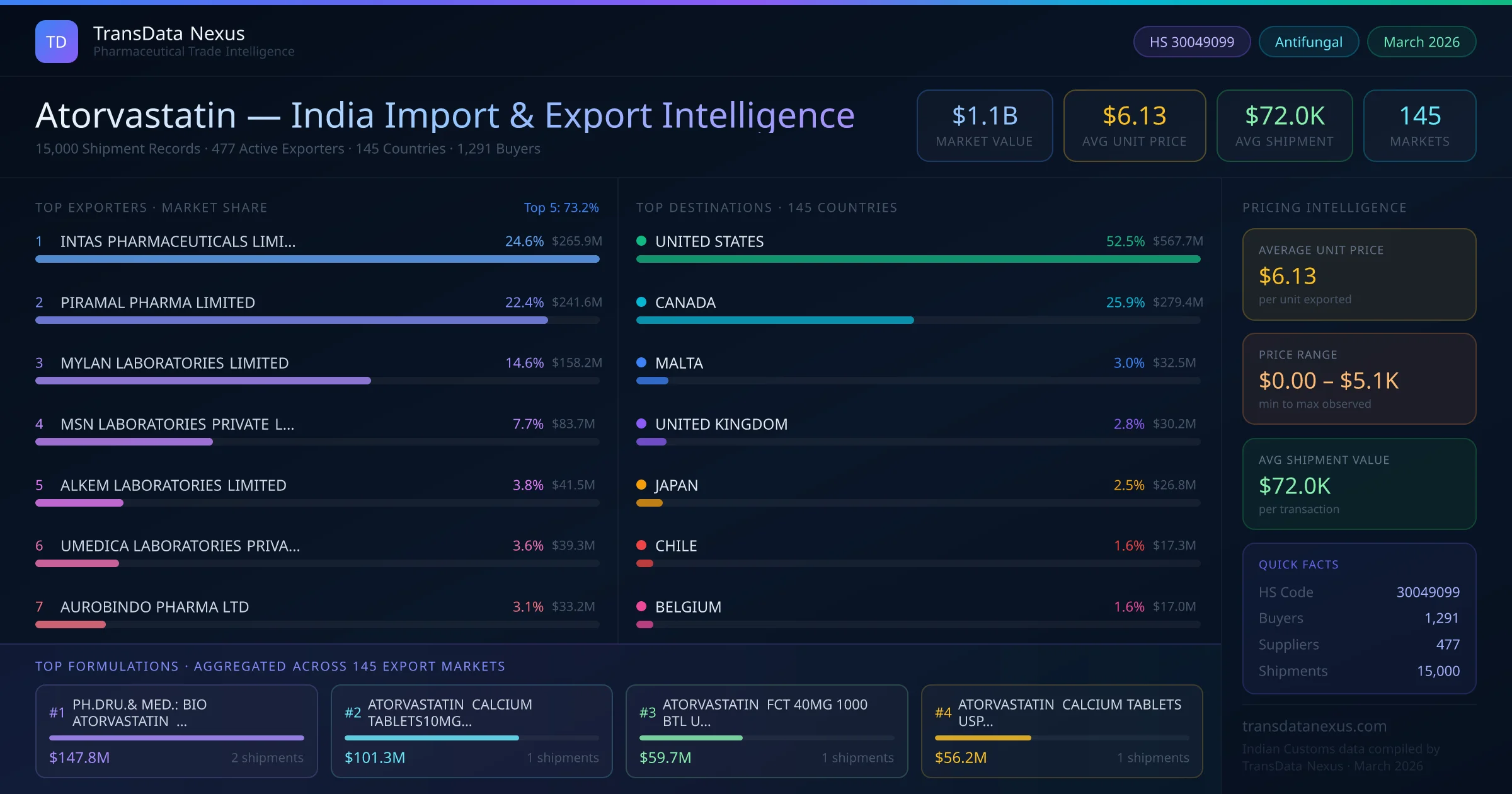
Top foreign suppliers (GERMANY) — Ranked by import value
NEWLIFE BIOPHARMA LIMITED is the leading Atorvastatin supplier from GERMANY to India, with import value of $11.6K across 4 shipments. The top 5 suppliers — NEWLIFE BIOPHARMA LIMITED, ILAPO, PRIVAPOSERVICES GMBH, ILAPO-INTERNATIONALE LUDWIGS-, BILCARE GCS IRELAND LIMITED — collectively account for 93.8% of total import value from this origin.
Top foreign suppliers (GERMANY)
Ranked by import value (USD) · Indian Customs (DGFT) data
| # | Supplier | Value (USD) | Shipments | Share |
|---|---|---|---|---|
| 1 | NEWLIFE BIOPHARMA LIMITED | $11.6K | 4 | 28.4% |
| 2 | ILAPO | $10.0K | 4 | 24.6% |
| 3 | PRIVAPOSERVICES GMBH | $8.2K | 8 | 20.1% |
| 4 | ILAPO-INTERNATIONALE LUDWIGS- | $5.9K | 2 | 14.6% |
| 5 | BILCARE GCS IRELAND LIMITED | $2.5K | 1 | 6.1% |
| 6 | NEW LIFE BIOPHARMA LIMITED | $2.2K | 2 | 5.3% |
| 7 | ESPEE PHARMA (UK) LTD | $324 | 1 | 0.8% |
| 8 | ALFRED E.TIEFENBACHER (GMBH & CO.KG) | $23 | 5 | 0.1% |
Top Indian buyers (importing from GERMANY)
Ranked by import value (USD)
| # | Buyer | Value (USD) | Shipments | Share |
|---|---|---|---|---|
| 1 | HETERO LABS LIMITED | $10.0K | 4 | 24.6% |
| 2 | UMEDICA LABORATORIES PRIVATE LIMITED | $8.8K | 1 | 21.5% |
| 3 | COPMED PHARMACEUTICALS PRIVATE LIMITED | $6.7K | 6 | 16.4% |
| 4 | WATSON PHARMA PVT.LTD. | $5.8K | 1 | 14.2% |
| 5 | INNOVA CAPTAB LIMITED | $2.5K | 1 | 6.1% |
| 6 | ALKEM LABORATORIES LIMITED | $1.6K | 1 | 4.0% |
| 7 | AUROBINDO PHARMA LIMITED | $1.5K | 1 | 3.7% |
| 8 | INTAS PHARMACEUTICALS LIMITED | $1.2K | 1 | 3.0% |
| 9 | PINNACLE LIFE SCIENCE PRIVATE LIMITED | $965 | 1 | 2.4% |
| 10 | AUROBINDO PHARMA LTD | $909 | 1 | 2.2% |
| 11 | LAMBDA THERAPEUTIC RESEARCH LIMITED | $324 | 1 | 0.8% |
| 12 | WINDLAS BIOTECH LIMITED | $311 | 2 | 0.8% |
| 13 | WATSON PHARMA PRIVATE LIMITED | $168 | 1 | 0.4% |
| 14 | LAURUS LABS LIMITED | $23 | 5 | 0.1% |
Import Statistics
- Import Value
- $40.8K
- Shipments
- 27
- Foreign Suppliers
- 8
- Indian Buyers
- 14
- Avg. Shipment
- $1.5K
Other Atorvastatin Import Origins
Related Analysis
India Import License & CDSCO Registration
GERMANY → India trade corridor intelligence
1India Import License & CDSCO Registration
Importing finished pharmaceutical formulations containing Atorvastatin into India requires compliance with the Drugs and Cosmetics Act, 1940, and associated rules. The Central Drugs Standard Control Organization (CDSCO) mandates that all imported drugs, including formulations under HS Code 30049099, obtain an Import Registration Certificate and an Import License. The registration process involves submitting Form 40/41 to CDSCO, along with necessary documents such as the Certificate of Pharmaceutical Product (CoPP), Free Sale Certificate, and Good Manufacturing Practice (GMP) certificate. The timeline for import drug registration can vary but typically ranges from 6 to 12 months, depending on the completeness of the application and regulatory reviews. Additionally, a No Objection Certificate (NOC) from the Directorate General of Foreign Trade (DGFT) is required for the importation of drugs.
Quality Testing & Batch Certification for Imports
GERMANY → India trade corridor intelligence
1Quality Testing & Batch Certification for Imports
Imported Atorvastatin formulations must undergo quality testing at CDSCO-approved laboratories in India. Each batch requires a Certificate of Analysis (CoA) confirming compliance with Indian Pharmacopoeia standards. Stability data, particularly for ICH Zone IV conditions, must be provided to demonstrate the product's stability in India's climate. Port inspection by customs drug inspectors is mandatory to verify the authenticity and quality of the imported drugs. Failure to meet these requirements can result in delays, rejections, or destruction of the consignment.
Recent Import Policy Changes (2024-2026)
GERMANY → India trade corridor intelligence
1Recent Import Policy Changes (2024-2026)
Between 2024 and 2026, India has implemented several regulatory updates affecting pharmaceutical imports. The introduction of the Production Linked Incentive (PLI) scheme has incentivized domestic manufacturing, potentially impacting the volume of finished formulation imports. Additionally, the Directorate General of Foreign Trade (DGFT) has issued notifications restricting imports of certain pharmaceutical chemicals, such as ATS-8, to regulate low-cost imports and support domestic manufacturers. These measures aim to balance the need for imported formulations with the promotion of domestic production capabilities.
MARKET
India's Atorvastatin Import Demand
GERMANY → India trade corridor intelligence
1India's Atorvastatin Import Demand
India imports finished Atorvastatin formulations to meet the demand for branded and patented products not manufactured domestically. Specific dosage forms, such as extended-release tablets, may not be produced locally, necessitating imports. Despite a robust domestic pharmaceutical industry, certain specialized formulations are sourced from countries like Germany, known for high-quality manufacturing standards. The market size for Atorvastatin formulations in India is substantial, with imports contributing to a significant portion of the total market.
India's Import Duty & Customs Structure
GERMANY → India trade corridor intelligence
1India's Import Duty & Customs Structure
The import duty structure for Atorvastatin formulations under HS Code 30049099 includes:
- Basic Customs Duty (BCD): 10%
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12%
Additional cess or duties, such as Education Cess or National Calamity Contingent Duty (NCCD), do not apply to this product category. Exemptions or preferential duty rates are not available for Germany-origin products under this HS Code. The total landed duty percentage is approximately 32.2%, calculated as follows:
- Total Duty = BCD + SWS + IGST = 10% + 10% + 12% = 32%
- Total Landed Duty Percentage = Total Duty / (1 - Total Duty) ≈ 32.2%
Germany as a Source — Competitive Position
GERMANY → India trade corridor intelligence
1Germany as a Source — Competitive Position
Germany's pharmaceutical industry is renowned for its stringent quality standards and advanced manufacturing technologies. This reputation makes German Atorvastatin formulations highly attractive to Indian importers seeking reliable and high-quality products. While other countries like China, the United States, and other European nations also supply Atorvastatin formulations, Germany's consistent quality and adherence to international regulatory standards provide a competitive edge. Germany's share in India's Atorvastatin import market is significant, reflecting its strong position as a preferred supplier.
CORRIDOR
Live Corridor Intelligence
GERMANY → India trade corridor intelligence
1Live Corridor Intelligence
The Germany to India pharmaceutical trade corridor is currently stable. Major ports such as Jawaharlal Nehru Port Trust (JNPT), Chennai, and Mundra are operating efficiently, with no significant congestion reported. Freight rates have remained consistent over the past year, and the exchange rate between the Indian Rupee (INR) and the Euro has shown minimal fluctuation, ensuring predictable costs for importers.
India's Import Policy & Self-Reliance Push
GERMANY → India trade corridor intelligence
1India's Import Policy & Self-Reliance Push
India's Production Linked Incentive (PLI) scheme aims to boost domestic manufacturing and reduce reliance on imports. While this initiative encourages local production, it may impact the volume of finished formulation imports, including Atorvastatin. However, the demand for specialized formulations not produced domestically continues to support the need for imports from countries like Germany.
Bilateral Trade Relations
GERMANY → India trade corridor intelligence
1Bilateral Trade Relations
India and Germany share a strong trade relationship, with ongoing discussions to enhance pharmaceutical trade. Negotiations for Free Trade Agreements (FTAs) and mutual recognition of Good Manufacturing Practices (GMP) are underway to facilitate smoother trade flows. These efforts aim to streamline regulatory processes and strengthen bilateral trade ties in the pharmaceutical sector.
Landed Cost Estimate (Germany to India)
GERMANY → India trade corridor intelligence
1Landed Cost Estimate (Germany to India)
The estimated landed cost for importing Atorvastatin formulations from Germany to India is as follows:
- FOB Price: $11,600
- Freight and Insurance: $1,500
- Basic Customs Duty (BCD): 10% of CIF value
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12% of CIF value
- Port Handling and CHA Charges: $500
Total Landed Cost = FOB Price + Freight and Insurance + BCD + SWS + IGST + Port Handling and CHA Charges
Total Landed Cost = $11,600 + $1,500 + ($1,500 * 10%) + ($1,500 * 10% * 10%) + ($1,500 * 12%) + $500
Total Landed Cost ≈ $15,000
Per-Unit Estimate: Assuming 10,000 units per shipment, the per-unit landed cost is approximately $1.50.
SOURCING
Why India Imports Atorvastatin from Germany
GERMANY → India trade corridor intelligence
1Why India Imports Atorvastatin from Germany
India imports Atorvastatin formulations from Germany due to the country's advanced pharmaceutical manufacturing capabilities and adherence to international quality standards. German manufacturers offer specialized dosage forms and patented formulations not available domestically, fulfilling specific market needs. The consistent quality and reliability of German products make them a preferred choice for Indian importers.
Germany vs. Other Origins — Comparison
GERMANY → India trade corridor intelligence
1Germany vs. Other Origins — Comparison
Compared to other origins like China, the United States, and other European countries, Germany offers superior quality assurance, regulatory compliance, and product reliability. While other suppliers may offer lower prices, Germany's products justify the higher cost through enhanced quality and consistency. This quality advantage positions Germany as a leading supplier of Atorvastatin formulations to India.
Supply Chain Risk for Indian Importers
GERMANY → India trade corridor intelligence
1Supply Chain Risk for Indian Importers
Potential risks for Indian importers include currency fluctuations, regulatory changes, and shipping disruptions. To mitigate these risks, importers should consider dual-sourcing strategies, maintain adequate inventory levels, and
India's Import Regulations for Atorvastatin from GERMANY
CDSCO registration, import licensing, and quality testing requirements
1India Import License & CDSCO Registration
Importing finished pharmaceutical formulations containing Atorvastatin into India requires compliance with the Drugs and Cosmetics Act, 1940, and associated rules. The Central Drugs Standard Control Organization (CDSCO) mandates that all imported drugs, including formulations under HS Code 30049099, obtain an Import Registration Certificate and an Import License. The registration process involves submitting Form 40/41 to CDSCO, along with necessary documents such as the Certificate of Pharmaceutical Product (CoPP), Free Sale Certificate, and Good Manufacturing Practice (GMP) certificate. The timeline for import drug registration can vary but typically ranges from 6 to 12 months, depending on the completeness of the application and regulatory reviews. Additionally, a No Objection Certificate (NOC) from the Directorate General of Foreign Trade (DGFT) is required for the importation of drugs.
2Quality Testing & Batch Certification for Imports
Imported Atorvastatin formulations must undergo quality testing at CDSCO-approved laboratories in India. Each batch requires a Certificate of Analysis (CoA) confirming compliance with Indian Pharmacopoeia standards. Stability data, particularly for ICH Zone IV conditions, must be provided to demonstrate the product's stability in India's climate. Port inspection by customs drug inspectors is mandatory to verify the authenticity and quality of the imported drugs. Failure to meet these requirements can result in delays, rejections, or destruction of the consignment.
3Recent Import Policy Changes (2024-2026)
Between 2024 and 2026, India has implemented several regulatory updates affecting pharmaceutical imports. The introduction of the Production Linked Incentive (PLI) scheme has incentivized domestic manufacturing, potentially impacting the volume of finished formulation imports. Additionally, the Directorate General of Foreign Trade (DGFT) has issued notifications restricting imports of certain pharmaceutical chemicals, such as ATS-8, to regulate low-cost imports and support domestic manufacturers. These measures aim to balance the need for imported formulations with the promotion of domestic production capabilities.
MARKET
4India's Atorvastatin Import Demand
India imports finished Atorvastatin formulations to meet the demand for branded and patented products not manufactured domestically. Specific dosage forms, such as extended-release tablets, may not be produced locally, necessitating imports. Despite a robust domestic pharmaceutical industry, certain specialized formulations are sourced from countries like Germany, known for high-quality manufacturing standards. The market size for Atorvastatin formulations in India is substantial, with imports contributing to a significant portion of the total market.
5India's Import Duty & Customs Structure
The import duty structure for Atorvastatin formulations under HS Code 30049099 includes:
- Basic Customs Duty (BCD): 10%
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12%
Additional cess or duties, such as Education Cess or National Calamity Contingent Duty (NCCD), do not apply to this product category. Exemptions or preferential duty rates are not available for Germany-origin products under this HS Code. The total landed duty percentage is approximately 32.2%, calculated as follows:
- Total Duty = BCD + SWS + IGST = 10% + 10% + 12% = 32%
- Total Landed Duty Percentage = Total Duty / (1 - Total Duty) ≈ 32.2%
6Germany as a Source — Competitive Position
Germany's pharmaceutical industry is renowned for its stringent quality standards and advanced manufacturing technologies. This reputation makes German Atorvastatin formulations highly attractive to Indian importers seeking reliable and high-quality products. While other countries like China, the United States, and other European nations also supply Atorvastatin formulations, Germany's consistent quality and adherence to international regulatory standards provide a competitive edge. Germany's share in India's Atorvastatin import market is significant, reflecting its strong position as a preferred supplier.
CORRIDOR
7Live Corridor Intelligence
The Germany to India pharmaceutical trade corridor is currently stable. Major ports such as Jawaharlal Nehru Port Trust (JNPT), Chennai, and Mundra are operating efficiently, with no significant congestion reported. Freight rates have remained consistent over the past year, and the exchange rate between the Indian Rupee (INR) and the Euro has shown minimal fluctuation, ensuring predictable costs for importers.
8India's Import Policy & Self-Reliance Push
India's Production Linked Incentive (PLI) scheme aims to boost domestic manufacturing and reduce reliance on imports. While this initiative encourages local production, it may impact the volume of finished formulation imports, including Atorvastatin. However, the demand for specialized formulations not produced domestically continues to support the need for imports from countries like Germany.
9Bilateral Trade Relations
India and Germany share a strong trade relationship, with ongoing discussions to enhance pharmaceutical trade. Negotiations for Free Trade Agreements (FTAs) and mutual recognition of Good Manufacturing Practices (GMP) are underway to facilitate smoother trade flows. These efforts aim to streamline regulatory processes and strengthen bilateral trade ties in the pharmaceutical sector.
10Landed Cost Estimate (Germany to India)
The estimated landed cost for importing Atorvastatin formulations from Germany to India is as follows:
- FOB Price: $11,600
- Freight and Insurance: $1,500
- Basic Customs Duty (BCD): 10% of CIF value
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12% of CIF value
- Port Handling and CHA Charges: $500
Total Landed Cost = FOB Price + Freight and Insurance + BCD + SWS + IGST + Port Handling and CHA Charges
Total Landed Cost = $11,600 + $1,500 + ($1,500 * 10%) + ($1,500 * 10% * 10%) + ($1,500 * 12%) + $500
Total Landed Cost ≈ $15,000
Per-Unit Estimate: Assuming 10,000 units per shipment, the per-unit landed cost is approximately $1.50.
SOURCING
11Why India Imports Atorvastatin from Germany
India imports Atorvastatin formulations from Germany due to the country's advanced pharmaceutical manufacturing capabilities and adherence to international quality standards. German manufacturers offer specialized dosage forms and patented formulations not available domestically, fulfilling specific market needs. The consistent quality and reliability of German products make them a preferred choice for Indian importers.
12Germany vs. Other Origins — Comparison
Compared to other origins like China, the United States, and other European countries, Germany offers superior quality assurance, regulatory compliance, and product reliability. While other suppliers may offer lower prices, Germany's products justify the higher cost through enhanced quality and consistency. This quality advantage positions Germany as a leading supplier of Atorvastatin formulations to India.
13Supply Chain Risk for Indian Importers
Potential risks for Indian importers include currency fluctuations, regulatory changes, and shipping disruptions. To mitigate these risks, importers should consider dual-sourcing strategies, maintain adequate inventory levels, and
India's Atorvastatin Import Market & Tariffs
Market demand, customs duty structure, and competitive landscape
1India Import License & CDSCO Registration
Importing finished pharmaceutical formulations containing Atorvastatin into India requires compliance with the Drugs and Cosmetics Act, 1940, and associated rules. The Central Drugs Standard Control Organization (CDSCO) mandates that all imported drugs, including formulations under HS Code 30049099, obtain an Import Registration Certificate and an Import License. The registration process involves submitting Form 40/41 to CDSCO, along with necessary documents such as the Certificate of Pharmaceutical Product (CoPP), Free Sale Certificate, and Good Manufacturing Practice (GMP) certificate. The timeline for import drug registration can vary but typically ranges from 6 to 12 months, depending on the completeness of the application and regulatory reviews. Additionally, a No Objection Certificate (NOC) from the Directorate General of Foreign Trade (DGFT) is required for the importation of drugs.
2Quality Testing & Batch Certification for Imports
Imported Atorvastatin formulations must undergo quality testing at CDSCO-approved laboratories in India. Each batch requires a Certificate of Analysis (CoA) confirming compliance with Indian Pharmacopoeia standards. Stability data, particularly for ICH Zone IV conditions, must be provided to demonstrate the product's stability in India's climate. Port inspection by customs drug inspectors is mandatory to verify the authenticity and quality of the imported drugs. Failure to meet these requirements can result in delays, rejections, or destruction of the consignment.
3Recent Import Policy Changes (2024-2026)
Between 2024 and 2026, India has implemented several regulatory updates affecting pharmaceutical imports. The introduction of the Production Linked Incentive (PLI) scheme has incentivized domestic manufacturing, potentially impacting the volume of finished formulation imports. Additionally, the Directorate General of Foreign Trade (DGFT) has issued notifications restricting imports of certain pharmaceutical chemicals, such as ATS-8, to regulate low-cost imports and support domestic manufacturers. These measures aim to balance the need for imported formulations with the promotion of domestic production capabilities.
MARKET
4India's Atorvastatin Import Demand
India imports finished Atorvastatin formulations to meet the demand for branded and patented products not manufactured domestically. Specific dosage forms, such as extended-release tablets, may not be produced locally, necessitating imports. Despite a robust domestic pharmaceutical industry, certain specialized formulations are sourced from countries like Germany, known for high-quality manufacturing standards. The market size for Atorvastatin formulations in India is substantial, with imports contributing to a significant portion of the total market.
5India's Import Duty & Customs Structure
The import duty structure for Atorvastatin formulations under HS Code 30049099 includes:
- Basic Customs Duty (BCD): 10%
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12%
Additional cess or duties, such as Education Cess or National Calamity Contingent Duty (NCCD), do not apply to this product category. Exemptions or preferential duty rates are not available for Germany-origin products under this HS Code. The total landed duty percentage is approximately 32.2%, calculated as follows:
- Total Duty = BCD + SWS + IGST = 10% + 10% + 12% = 32%
- Total Landed Duty Percentage = Total Duty / (1 - Total Duty) ≈ 32.2%
6Germany as a Source — Competitive Position
Germany's pharmaceutical industry is renowned for its stringent quality standards and advanced manufacturing technologies. This reputation makes German Atorvastatin formulations highly attractive to Indian importers seeking reliable and high-quality products. While other countries like China, the United States, and other European nations also supply Atorvastatin formulations, Germany's consistent quality and adherence to international regulatory standards provide a competitive edge. Germany's share in India's Atorvastatin import market is significant, reflecting its strong position as a preferred supplier.
CORRIDOR
7Live Corridor Intelligence
The Germany to India pharmaceutical trade corridor is currently stable. Major ports such as Jawaharlal Nehru Port Trust (JNPT), Chennai, and Mundra are operating efficiently, with no significant congestion reported. Freight rates have remained consistent over the past year, and the exchange rate between the Indian Rupee (INR) and the Euro has shown minimal fluctuation, ensuring predictable costs for importers.
8India's Import Policy & Self-Reliance Push
India's Production Linked Incentive (PLI) scheme aims to boost domestic manufacturing and reduce reliance on imports. While this initiative encourages local production, it may impact the volume of finished formulation imports, including Atorvastatin. However, the demand for specialized formulations not produced domestically continues to support the need for imports from countries like Germany.
9Bilateral Trade Relations
India and Germany share a strong trade relationship, with ongoing discussions to enhance pharmaceutical trade. Negotiations for Free Trade Agreements (FTAs) and mutual recognition of Good Manufacturing Practices (GMP) are underway to facilitate smoother trade flows. These efforts aim to streamline regulatory processes and strengthen bilateral trade ties in the pharmaceutical sector.
10Landed Cost Estimate (Germany to India)
The estimated landed cost for importing Atorvastatin formulations from Germany to India is as follows:
- FOB Price: $11,600
- Freight and Insurance: $1,500
- Basic Customs Duty (BCD): 10% of CIF value
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12% of CIF value
- Port Handling and CHA Charges: $500
Total Landed Cost = FOB Price + Freight and Insurance + BCD + SWS + IGST + Port Handling and CHA Charges
Total Landed Cost = $11,600 + $1,500 + ($1,500 * 10%) + ($1,500 * 10% * 10%) + ($1,500 * 12%) + $500
Total Landed Cost ≈ $15,000
Per-Unit Estimate: Assuming 10,000 units per shipment, the per-unit landed cost is approximately $1.50.
SOURCING
11Why India Imports Atorvastatin from Germany
India imports Atorvastatin formulations from Germany due to the country's advanced pharmaceutical manufacturing capabilities and adherence to international quality standards. German manufacturers offer specialized dosage forms and patented formulations not available domestically, fulfilling specific market needs. The consistent quality and reliability of German products make them a preferred choice for Indian importers.
12Germany vs. Other Origins — Comparison
Compared to other origins like China, the United States, and other European countries, Germany offers superior quality assurance, regulatory compliance, and product reliability. While other suppliers may offer lower prices, Germany's products justify the higher cost through enhanced quality and consistency. This quality advantage positions Germany as a leading supplier of Atorvastatin formulations to India.
13Supply Chain Risk for Indian Importers
Potential risks for Indian importers include currency fluctuations, regulatory changes, and shipping disruptions. To mitigate these risks, importers should consider dual-sourcing strategies, maintain adequate inventory levels, and
Why India Imports Atorvastatin from GERMANY
Import rationale, competitive comparison, supply chain risk, and procurement strategy
1India Import License & CDSCO Registration
Importing finished pharmaceutical formulations containing Atorvastatin into India requires compliance with the Drugs and Cosmetics Act, 1940, and associated rules. The Central Drugs Standard Control Organization (CDSCO) mandates that all imported drugs, including formulations under HS Code 30049099, obtain an Import Registration Certificate and an Import License. The registration process involves submitting Form 40/41 to CDSCO, along with necessary documents such as the Certificate of Pharmaceutical Product (CoPP), Free Sale Certificate, and Good Manufacturing Practice (GMP) certificate. The timeline for import drug registration can vary but typically ranges from 6 to 12 months, depending on the completeness of the application and regulatory reviews. Additionally, a No Objection Certificate (NOC) from the Directorate General of Foreign Trade (DGFT) is required for the importation of drugs.
2Quality Testing & Batch Certification for Imports
Imported Atorvastatin formulations must undergo quality testing at CDSCO-approved laboratories in India. Each batch requires a Certificate of Analysis (CoA) confirming compliance with Indian Pharmacopoeia standards. Stability data, particularly for ICH Zone IV conditions, must be provided to demonstrate the product's stability in India's climate. Port inspection by customs drug inspectors is mandatory to verify the authenticity and quality of the imported drugs. Failure to meet these requirements can result in delays, rejections, or destruction of the consignment.
3Recent Import Policy Changes (2024-2026)
Between 2024 and 2026, India has implemented several regulatory updates affecting pharmaceutical imports. The introduction of the Production Linked Incentive (PLI) scheme has incentivized domestic manufacturing, potentially impacting the volume of finished formulation imports. Additionally, the Directorate General of Foreign Trade (DGFT) has issued notifications restricting imports of certain pharmaceutical chemicals, such as ATS-8, to regulate low-cost imports and support domestic manufacturers. These measures aim to balance the need for imported formulations with the promotion of domestic production capabilities.
MARKET
4India's Atorvastatin Import Demand
India imports finished Atorvastatin formulations to meet the demand for branded and patented products not manufactured domestically. Specific dosage forms, such as extended-release tablets, may not be produced locally, necessitating imports. Despite a robust domestic pharmaceutical industry, certain specialized formulations are sourced from countries like Germany, known for high-quality manufacturing standards. The market size for Atorvastatin formulations in India is substantial, with imports contributing to a significant portion of the total market.
5India's Import Duty & Customs Structure
The import duty structure for Atorvastatin formulations under HS Code 30049099 includes:
- Basic Customs Duty (BCD): 10%
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12%
Additional cess or duties, such as Education Cess or National Calamity Contingent Duty (NCCD), do not apply to this product category. Exemptions or preferential duty rates are not available for Germany-origin products under this HS Code. The total landed duty percentage is approximately 32.2%, calculated as follows:
- Total Duty = BCD + SWS + IGST = 10% + 10% + 12% = 32%
- Total Landed Duty Percentage = Total Duty / (1 - Total Duty) ≈ 32.2%
6Germany as a Source — Competitive Position
Germany's pharmaceutical industry is renowned for its stringent quality standards and advanced manufacturing technologies. This reputation makes German Atorvastatin formulations highly attractive to Indian importers seeking reliable and high-quality products. While other countries like China, the United States, and other European nations also supply Atorvastatin formulations, Germany's consistent quality and adherence to international regulatory standards provide a competitive edge. Germany's share in India's Atorvastatin import market is significant, reflecting its strong position as a preferred supplier.
CORRIDOR
7Live Corridor Intelligence
The Germany to India pharmaceutical trade corridor is currently stable. Major ports such as Jawaharlal Nehru Port Trust (JNPT), Chennai, and Mundra are operating efficiently, with no significant congestion reported. Freight rates have remained consistent over the past year, and the exchange rate between the Indian Rupee (INR) and the Euro has shown minimal fluctuation, ensuring predictable costs for importers.
8India's Import Policy & Self-Reliance Push
India's Production Linked Incentive (PLI) scheme aims to boost domestic manufacturing and reduce reliance on imports. While this initiative encourages local production, it may impact the volume of finished formulation imports, including Atorvastatin. However, the demand for specialized formulations not produced domestically continues to support the need for imports from countries like Germany.
9Bilateral Trade Relations
India and Germany share a strong trade relationship, with ongoing discussions to enhance pharmaceutical trade. Negotiations for Free Trade Agreements (FTAs) and mutual recognition of Good Manufacturing Practices (GMP) are underway to facilitate smoother trade flows. These efforts aim to streamline regulatory processes and strengthen bilateral trade ties in the pharmaceutical sector.
10Landed Cost Estimate (Germany to India)
The estimated landed cost for importing Atorvastatin formulations from Germany to India is as follows:
- FOB Price: $11,600
- Freight and Insurance: $1,500
- Basic Customs Duty (BCD): 10% of CIF value
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12% of CIF value
- Port Handling and CHA Charges: $500
Total Landed Cost = FOB Price + Freight and Insurance + BCD + SWS + IGST + Port Handling and CHA Charges
Total Landed Cost = $11,600 + $1,500 + ($1,500 * 10%) + ($1,500 * 10% * 10%) + ($1,500 * 12%) + $500
Total Landed Cost ≈ $15,000
Per-Unit Estimate: Assuming 10,000 units per shipment, the per-unit landed cost is approximately $1.50.
SOURCING
11Why India Imports Atorvastatin from Germany
India imports Atorvastatin formulations from Germany due to the country's advanced pharmaceutical manufacturing capabilities and adherence to international quality standards. German manufacturers offer specialized dosage forms and patented formulations not available domestically, fulfilling specific market needs. The consistent quality and reliability of German products make them a preferred choice for Indian importers.
12Germany vs. Other Origins — Comparison
Compared to other origins like China, the United States, and other European countries, Germany offers superior quality assurance, regulatory compliance, and product reliability. While other suppliers may offer lower prices, Germany's products justify the higher cost through enhanced quality and consistency. This quality advantage positions Germany as a leading supplier of Atorvastatin formulations to India.
13Supply Chain Risk for Indian Importers
Potential risks for Indian importers include currency fluctuations, regulatory changes, and shipping disruptions. To mitigate these risks, importers should consider dual-sourcing strategies, maintain adequate inventory levels, and
Import Due Diligence — Atorvastatin from GERMANY
Import license checklist, document requirements, quality testing, and compliance
1India Import License & CDSCO Registration
Importing finished pharmaceutical formulations containing Atorvastatin into India requires compliance with the Drugs and Cosmetics Act, 1940, and associated rules. The Central Drugs Standard Control Organization (CDSCO) mandates that all imported drugs, including formulations under HS Code 30049099, obtain an Import Registration Certificate and an Import License. The registration process involves submitting Form 40/41 to CDSCO, along with necessary documents such as the Certificate of Pharmaceutical Product (CoPP), Free Sale Certificate, and Good Manufacturing Practice (GMP) certificate. The timeline for import drug registration can vary but typically ranges from 6 to 12 months, depending on the completeness of the application and regulatory reviews. Additionally, a No Objection Certificate (NOC) from the Directorate General of Foreign Trade (DGFT) is required for the importation of drugs.
2Quality Testing & Batch Certification for Imports
Imported Atorvastatin formulations must undergo quality testing at CDSCO-approved laboratories in India. Each batch requires a Certificate of Analysis (CoA) confirming compliance with Indian Pharmacopoeia standards. Stability data, particularly for ICH Zone IV conditions, must be provided to demonstrate the product's stability in India's climate. Port inspection by customs drug inspectors is mandatory to verify the authenticity and quality of the imported drugs. Failure to meet these requirements can result in delays, rejections, or destruction of the consignment.
3Recent Import Policy Changes (2024-2026)
Between 2024 and 2026, India has implemented several regulatory updates affecting pharmaceutical imports. The introduction of the Production Linked Incentive (PLI) scheme has incentivized domestic manufacturing, potentially impacting the volume of finished formulation imports. Additionally, the Directorate General of Foreign Trade (DGFT) has issued notifications restricting imports of certain pharmaceutical chemicals, such as ATS-8, to regulate low-cost imports and support domestic manufacturers. These measures aim to balance the need for imported formulations with the promotion of domestic production capabilities.
MARKET
4India's Atorvastatin Import Demand
India imports finished Atorvastatin formulations to meet the demand for branded and patented products not manufactured domestically. Specific dosage forms, such as extended-release tablets, may not be produced locally, necessitating imports. Despite a robust domestic pharmaceutical industry, certain specialized formulations are sourced from countries like Germany, known for high-quality manufacturing standards. The market size for Atorvastatin formulations in India is substantial, with imports contributing to a significant portion of the total market.
5India's Import Duty & Customs Structure
The import duty structure for Atorvastatin formulations under HS Code 30049099 includes:
- Basic Customs Duty (BCD): 10%
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12%
Additional cess or duties, such as Education Cess or National Calamity Contingent Duty (NCCD), do not apply to this product category. Exemptions or preferential duty rates are not available for Germany-origin products under this HS Code. The total landed duty percentage is approximately 32.2%, calculated as follows:
- Total Duty = BCD + SWS + IGST = 10% + 10% + 12% = 32%
- Total Landed Duty Percentage = Total Duty / (1 - Total Duty) ≈ 32.2%
6Germany as a Source — Competitive Position
Germany's pharmaceutical industry is renowned for its stringent quality standards and advanced manufacturing technologies. This reputation makes German Atorvastatin formulations highly attractive to Indian importers seeking reliable and high-quality products. While other countries like China, the United States, and other European nations also supply Atorvastatin formulations, Germany's consistent quality and adherence to international regulatory standards provide a competitive edge. Germany's share in India's Atorvastatin import market is significant, reflecting its strong position as a preferred supplier.
CORRIDOR
7Live Corridor Intelligence
The Germany to India pharmaceutical trade corridor is currently stable. Major ports such as Jawaharlal Nehru Port Trust (JNPT), Chennai, and Mundra are operating efficiently, with no significant congestion reported. Freight rates have remained consistent over the past year, and the exchange rate between the Indian Rupee (INR) and the Euro has shown minimal fluctuation, ensuring predictable costs for importers.
8India's Import Policy & Self-Reliance Push
India's Production Linked Incentive (PLI) scheme aims to boost domestic manufacturing and reduce reliance on imports. While this initiative encourages local production, it may impact the volume of finished formulation imports, including Atorvastatin. However, the demand for specialized formulations not produced domestically continues to support the need for imports from countries like Germany.
9Bilateral Trade Relations
India and Germany share a strong trade relationship, with ongoing discussions to enhance pharmaceutical trade. Negotiations for Free Trade Agreements (FTAs) and mutual recognition of Good Manufacturing Practices (GMP) are underway to facilitate smoother trade flows. These efforts aim to streamline regulatory processes and strengthen bilateral trade ties in the pharmaceutical sector.
10Landed Cost Estimate (Germany to India)
The estimated landed cost for importing Atorvastatin formulations from Germany to India is as follows:
- FOB Price: $11,600
- Freight and Insurance: $1,500
- Basic Customs Duty (BCD): 10% of CIF value
- Social Welfare Surcharge (SWS): 10% of BCD
- Integrated Goods and Services Tax (IGST): 12% of CIF value
- Port Handling and CHA Charges: $500
Total Landed Cost = FOB Price + Freight and Insurance + BCD + SWS + IGST + Port Handling and CHA Charges
Total Landed Cost = $11,600 + $1,500 + ($1,500 * 10%) + ($1,500 * 10% * 10%) + ($1,500 * 12%) + $500
Total Landed Cost ≈ $15,000
Per-Unit Estimate: Assuming 10,000 units per shipment, the per-unit landed cost is approximately $1.50.
SOURCING
11Why India Imports Atorvastatin from Germany
India imports Atorvastatin formulations from Germany due to the country's advanced pharmaceutical manufacturing capabilities and adherence to international quality standards. German manufacturers offer specialized dosage forms and patented formulations not available domestically, fulfilling specific market needs. The consistent quality and reliability of German products make them a preferred choice for Indian importers.
12Germany vs. Other Origins — Comparison
Compared to other origins like China, the United States, and other European countries, Germany offers superior quality assurance, regulatory compliance, and product reliability. While other suppliers may offer lower prices, Germany's products justify the higher cost through enhanced quality and consistency. This quality advantage positions Germany as a leading supplier of Atorvastatin formulations to India.
13Supply Chain Risk for Indian Importers
Potential risks for Indian importers include currency fluctuations, regulatory changes, and shipping disruptions. To mitigate these risks, importers should consider dual-sourcing strategies, maintain adequate inventory levels, and
Frequently Asked Questions — Atorvastatin Imports from GERMANY
Answers based on Indian Customs (DGFT) import records compiled by TransData Nexus
QWho are the top Atorvastatin suppliers from GERMANY to India?
The top Atorvastatin suppliers from GERMANY to India include NEWLIFE BIOPHARMA LIMITED, ILAPO, PRIVAPOSERVICES GMBH. The leading supplier is NEWLIFE BIOPHARMA LIMITED with import value of $11.6K USD across 4 shipments. India imported Atorvastatin worth $40.8K USD from GERMANY in total across 27 shipments.
QHow much Atorvastatin does India import from GERMANY?
India imported Atorvastatin worth $40.8K USD from GERMANY across 27 shipments. Data is from Indian Customs (DGFT) records. Values are in USD.
QWho are the main Indian buyers of Atorvastatin from GERMANY?
The main Indian buyers of Atorvastatin sourced from GERMANY include HETERO LABS LIMITED, UMEDICA LABORATORIES PRIVATE LIMITED, COPMED PHARMACEUTICALS PRIVATE LIMITED. The largest buyer is HETERO LABS LIMITED with $10.0K in imports across 4 shipments.
QWhat is the total value of Atorvastatin imports from GERMANY to India?
The total value of Atorvastatin imports from GERMANY to India is $40.8K USD, across 27 shipments and 8 foreign suppliers. Data source: Indian Customs (DGFT).
Official References & Regulatory Resources
- India Trade Statistics (DGFT)
- Invest India — Pharma Sector
- WCO HS Nomenclature
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory and trade status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Route Identification: GERMANY to India pharmaceutical import trade corridor identified from Indian Customs (DGFT) shipping bill records for Atorvastatin.
- 2.Supplier/Buyer Matching: 8 foreign suppliers from GERMANY and 14 Indian buyers identified using company name normalization across customs records.
- 3.Value Aggregation: Total import value of $40.8K aggregated from 27 verified shipping bill records. All values reported in FOB USD as recorded by Indian Customs.
- 4.Market Share Analysis: Each supplier and buyer contribution calculated as a percentage of total route value. Statistical normalization applied to ensure accurate representation across varying shipment sizes.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
27 Verified Shipments
8 suppliers, 14 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists