Who Buys Asava from India — 173 Importers Behind a $458.0K Market
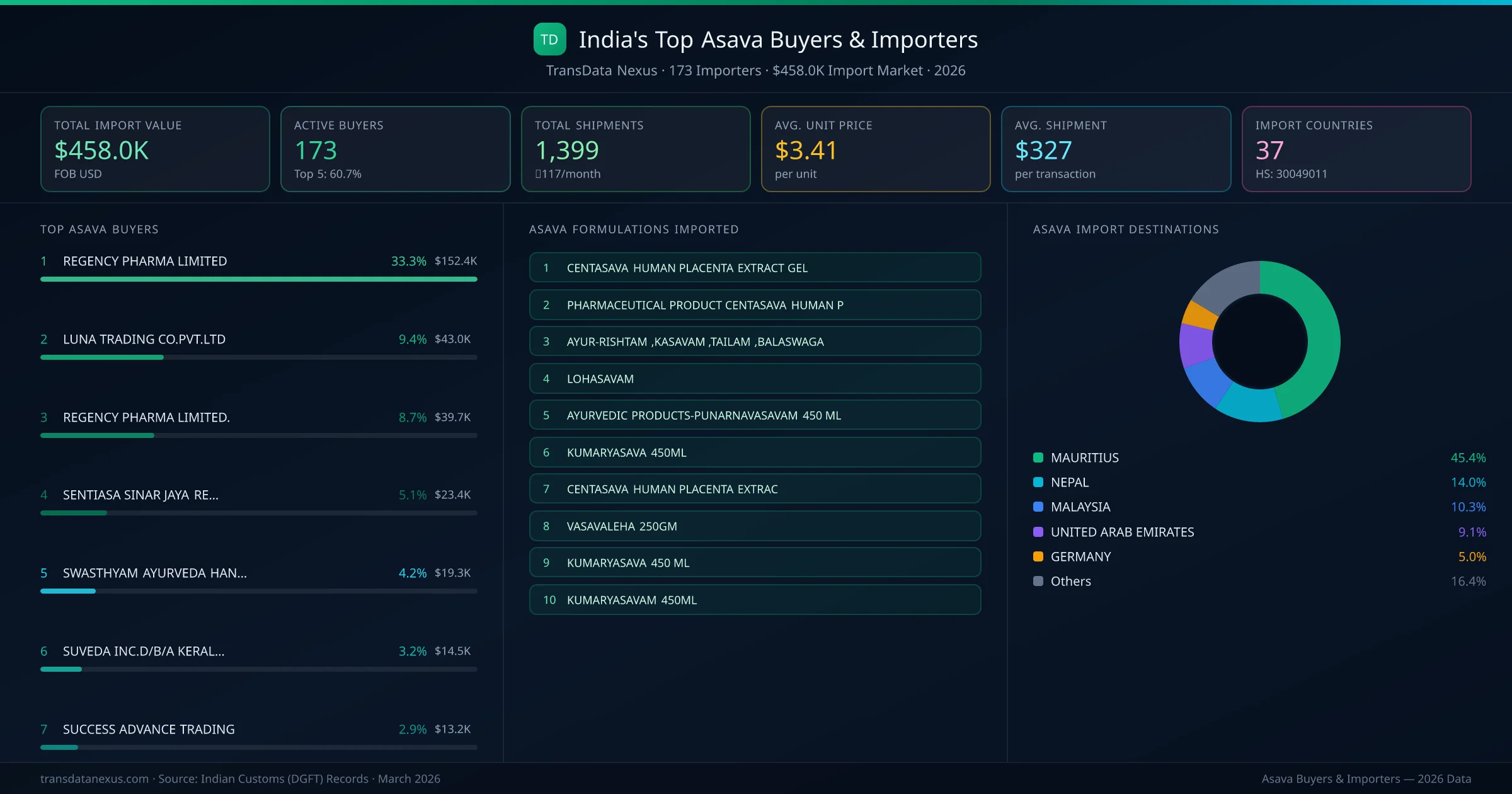
India's asava import market is served by 173 active buyers who collectively imported $458.0K across 1,399 shipments. REGENCY PHARMA LIMITED (MAURITIUS) leads with a 33.3% market share, followed by LUNA TRADING CO.PVT.LTD and SENTIASA SINAR JAYA RESOURCES. The top 5 buyers together control 55.2% of total import value, reflecting a moderately competitive buyer landscape.
Top Asava Importers — Ranked by Import Value
REGENCY PHARMA LIMITED (MAURITIUS) is the leading asava importer from India, holding a 33.3% share of the $458.0K market across 1,399 shipments from 173 buyers. The top 5 buyers — REGENCY PHARMA LIMITED (MAURITIUS), LUNA TRADING CO.PVT.LTD (NEPAL), SENTIASA SINAR JAYA RESOURCES (MALAYSIA), SWASTHYAM AYURVEDA HANDEL (GERMANY), SUVEDA INC.D/B/A KERALA AYURVEDA (UNITED STATES) — collectively control 55.2% of total import value.
Top Asava Buyers & Importers
Ranked by import value · 173 active buyers · Indian Customs (DGFT) data
| # | Buyer & Formulations | Value | Suppliers | Share |
|---|---|---|---|---|
| 1 | REGENCY PHARMA LIMITEDMAURITIUS CENTASAVA HUMAN PLACENTA EXTRACT GELPHARMACEUTICAL PRODUCT CENTASAVA HUMAN PCENTASAVA HUMAN PLACENTA EXTRACT GEL PACK SIZE 1 X 20 GM | $152.4K | 5 | 33.3% |
| 2 | LUNA TRADING CO.PVT.LTDNEPAL KUMARYASAVA 450MLVASAVALEHA 250GMKUMARYASAVA 450 ML | $43.0K | 5 | 9.4% |
| 3 | SENTIASA SINAR JAYA RESOURCESMALAYSIA AYUR-RISHTAM ,KASAVAM ,TAILAM ,BALASWAGAAYURVEDIC : PUNAMNAVASAVAM - 80 NOS, BHRAYURVEDIC PRODUCT KANAKASAVAM | $23.4K | 5 | 5.1% |
| 4 | SWASTHYAM AYURVEDA HANDELGERMANY LOHASAVAMKUMARYASAVAM 450 MLLOHASAVAM 450 ML | $19.3K | 5 | 4.2% |
| 5 | SUVEDA INC.D/B/A KERALA AYURVEDAUNITED STATES AYURVEDIC PRODUCTS-PUNARNAVASAVAM 450 MLBA BHRINGRAJASAVA DRINKPUNARNAVASAVAM 450 ML | $14.5K | 5 | 3.2% |
| 6 | SUCCESS ADVANCE TRADINGMALAYSIA AYUR-RISHTAM ,KASAVAM ,TAILAM ,BALASWAGAAYURVEDIC : PUNAMNAVASAVAM - 80 NOS, BHRAYURVEDIC PRODUCT KANAKASAVAM | $13.2K | 5 | 2.9% |
| 7 | M/S. REGENCY PHARMA LIMITEDMAURITIUS CENTASAVA HUMAN PLACENTA EXTRACT GELPHARMACEUTICAL PRODUCT CENTASAVA HUMAN PCENTASAVA HUMAN PLACENTA EXTRACT GEL PACK SIZE 1 X 20 GM | $12.8K | 5 | 2.8% |
| 8 | M S RAWAFED MUSCAT NATIONALOMAN CENTASAVA HUMAN PLACENTA EXTRACT GELPHARMACEUTICAL PRODUCT CENTASAVA HUMAN PCENTASAVA HUMAN PLACENTA EXTRACT GEL PACK SIZE 1 X 20 GM | $9.3K | 5 | 2.0% |
| 9 | LUNA TRADINGNEPAL KUMARYASAVA 450MLVASAVALEHA 250GMKUMARYASAVA 450 ML | $7.6K | 5 | 1.7% |
| 10 | KOTTAKKAL AYURVEDIC MEDICALS LLCUNITED ARAB EMIRATES PIPPALYASAVAM 450MLLOHASAVAM 450MLCHANDANASAVAM 450ML | $6.2K | 5 | 1.4% |
| 11 | M/SKOTTAKAL AYURVEDIC MEDICALS LLCUNITED ARAB EMIRATES PIPPALYASAVAM 450MLLOHASAVAM 450MLKANAKASAVAM 450ML | $4.2K | 5 | 0.9% |
| 12 | LUNA TRADING CO.PVT.LTD.NEPAL KUMARYASAVA 450MLVASAVALEHA 250GMKUMARYASAVA 450 ML | $3.9K | 5 | 0.8% |
| 13 | LUNA TRADING CO PVT LTDNEPAL KUMARYASAVA 450MLVASAVALEHA 250GMKUMARYASAVA 450 ML | $3.5K | 5 | 0.8% |
| 14 | AYUS AYURVEDIC HERBS TRADING LLCUNITED ARAB EMIRATES LOHASAVAM -450MLSARIBADYASAVAM -450MLKUMARYASAVAM -450ML | $3.4K | 5 | 0.8% |
| 15 | M/S.RAWAFED MUSCAT NATIONALOMAN NIMBAMRUTHASAVAM 450MLARDRAKASAVAM 450MLPUNARNAVASAVAM 450ML | $3.3K | 5 | 0.7% |
Related Analysis
Market Analysis: Top Importing Countries & Importers
Comprehensive geographic market intelligence dashboard
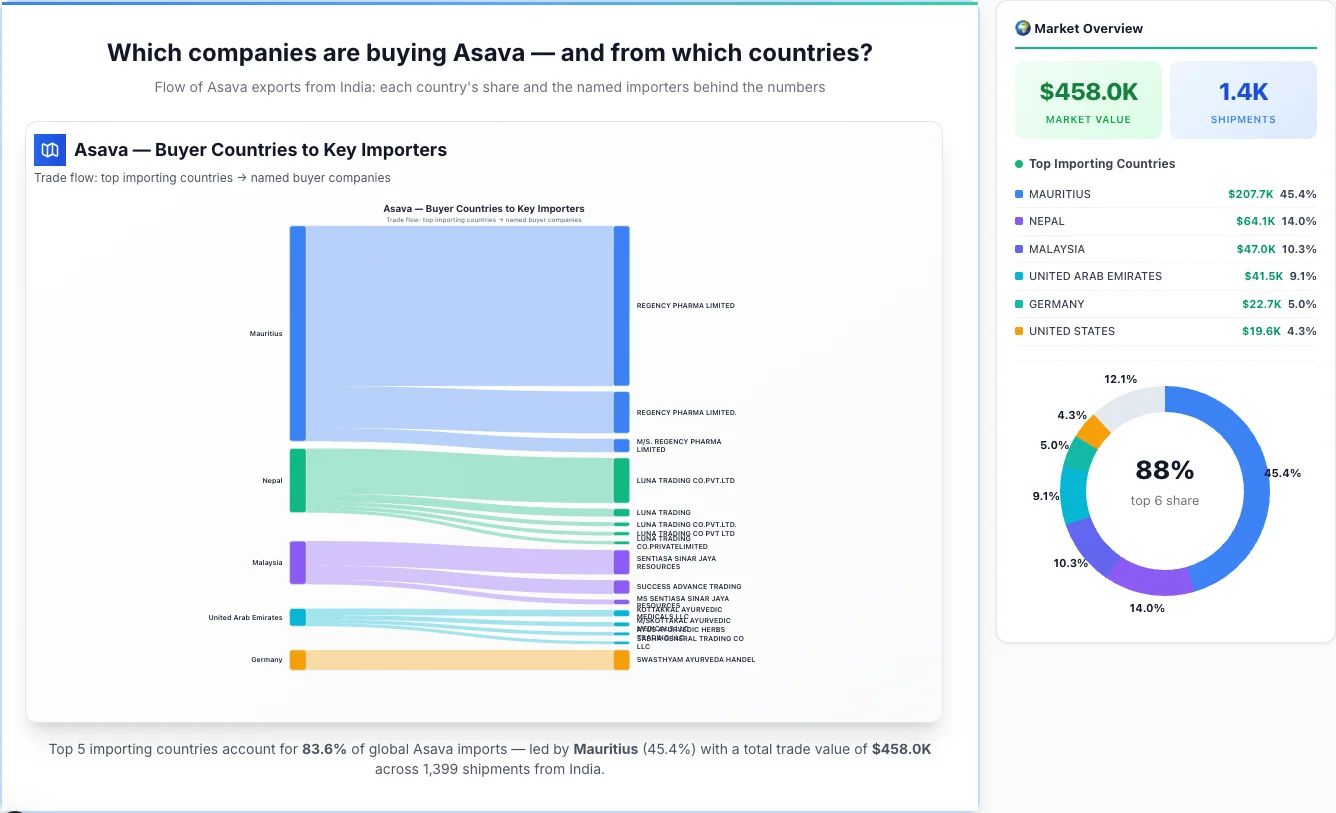
Which companies are buying Asava — and from which countries?
Flow of Asava exports from India: each country's share and the named importers behind the numbers
Asava — Buyer Countries to Key Importers
Trade flow: top importing countries → named buyer companies
Market Analysis: Top Importing Countries & Their Importers
1Mauritius
Mauritius emerges as the first largest importing country, achieving a total trade value of $207.7K through 8 shipments. This represents a market share of 45.4% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Mauritius has a diverse importer base, with their top importers including:
• REGENCY PHARMA LIMITED - $152.4K (73.4% of country's total business)
• M/S. REGENCY PHARMA LIMITED - $12.8K (6.2% of country's total business)
Key Insight: Mauritius demonstrates strong market positioning with an average shipment value of $26.0K, indicating premium pricing strategy compared to the market average of $327.
Mauritius is a key market for Asava imports, representing 45.4% of total trade value.
2Nepal
Nepal emerges as the second largest importing country, achieving a total trade value of $64.1K through 127 shipments. This represents a market share of 14.0% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Nepal has a diverse importer base, with their top importers including:
• LUNA TRADING CO.PVT.LTD - $43.0K (67.0% of country's total business)
• LUNA TRADING - $7.6K (11.9% of country's total business)
• LUNA TRADING CO.PVT.LTD. - $3.9K (6.0% of country's total business)
• LUNA TRADING CO PVT LTD - $3.5K (5.5% of country's total business)
• LUNA TRADING CO.PRIVATELIMITED - $3.2K (4.9% of country's total business)
• Rest of Nepal's importers - $3.0K (4.7% of country's total business)
Key Insight: Nepal demonstrates strong market positioning with an average shipment value of $505, indicating premium pricing strategy compared to the market average of $327.
Nepal is a key market for Asava imports, representing 14.0% of total trade value.
3Malaysia
Malaysia emerges as the third largest importing country, achieving a total trade value of $47.0K through 4 shipments. This represents a market share of 10.3% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Malaysia has a diverse importer base, with their top importers including:
• SENTIASA SINAR JAYA RESOURCES - $23.4K (49.7% of country's total business)
• SUCCESS ADVANCE TRADING - $13.2K (28.1% of country's total business)
Key Insight: Malaysia demonstrates strong market positioning with an average shipment value of $11.7K, indicating premium pricing strategy compared to the market average of $327.
Malaysia is a key market for Asava imports, representing 10.3% of total trade value.
4United Arab Emirates
United Arab Emirates emerges as the fourth largest importing country, achieving a total trade value of $41.5K through 231 shipments. This represents a market share of 9.1% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: United Arab Emirates has a diverse importer base, with their top importers including:
• KOTTAKKAL AYURVEDIC MEDICALS LLC - $6.2K (14.9% of country's total business)
• M/SKOTTAKAL AYURVEDIC MEDICALS LLC - $4.2K (10.2% of country's total business)
• AYUS AYURVEDIC HERBS TRADING LLC - $3.4K (8.3% of country's total business)
• SABHA GENERAL TRADING CO LLC - $3.0K (7.2% of country's total business)
Key Insight: United Arab Emirates demonstrates strong market positioning with an average shipment value of $180, indicating competitive pricing strategy compared to the market average of $327.
United Arab Emirates is a key market for Asava imports, representing 9.1% of total trade value.
5Germany
Germany emerges as the fifth largest importing country, achieving a total trade value of $22.7K through 3 shipments. This represents a market share of 5.0% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Germany has a diverse importer base, with their top importers including:
• SWASTHYAM AYURVEDA HANDEL - $19.3K (85.0% of country's total business)
Key Insight: Germany demonstrates strong market positioning with an average shipment value of $7.6K, indicating premium pricing strategy compared to the market average of $327.
Germany is a key market for Asava imports, representing 5.0% of total trade value.
Buyer Segment Analysis
TransData Nexus Asava buyer market intelligence
1Buyer Segment Analysis
The importers of Asava from India can be categorized into several segments:
1. Distributors: Entities like REGENCY PHARMA LIMITED (Mauritius) and LUNA TRADING CO. PVT. LTD (Nepal) function as intermediaries, purchasing Asava in bulk to supply to local retailers, healthcare providers, or other businesses. Their buying behavior typically involves large, regular orders to maintain a steady supply chain.
2. Retail Chains: Companies such as SUVEDA INC. D/B/A KERALA AYURVEDA (United States) and KOTTAKKAL AYURVEDIC MEDICALS LLC (United Arab Emirates) operate retail outlets specializing in Ayurvedic products. They procure Asava to stock their shelves, with order patterns influenced by consumer demand and seasonal trends.
3. Re-exporters: Firms like SENTIASA SINAR JAYA RESOURCES (Malaysia) and SUCCESS ADVANCE TRADING (Malaysia) may import Asava with the intent to re-export to neighboring countries or regions. Their purchasing behavior is often characterized by opportunistic buying, capitalizing on market demands in various territories.
4. Contract Manufacturers: Entities such as SWASTHYAM AYURVEDA HANDEL (Germany) might import Asava as a component for further processing or inclusion in proprietary formulations. Their orders are typically aligned with production schedules and client requirements.
Understanding these segments is crucial for tailoring marketing strategies, forecasting demand, and optimizing supply chain logistics.
Country-Specific Import Regulations
TransData Nexus Asava buyer market intelligence
1Country-Specific Import Regulations
Mauritius
1. Drug Regulatory Authority: Mauritius Pharmacy Board.
2. Import Registration Process: Importers must obtain a pharmaceutical import license from the Mauritius Pharmacy Board, ensuring compliance with local standards and regulations.
3. Estimated Timeline for Registration: The registration process typically takes 4-6 weeks, subject to the completeness of the application and any required inspections.
4. Import Tariff/Duty for HS 30049011: Mauritius imposes a standard import duty of 0% on pharmaceutical products under HS Code 30049011, promoting access to essential medicines.
Nepal
1. Drug Regulatory Authority: Department of Drug Administration (DDA).
2. Import Registration Process: Importers must submit an application to the DDA, including a certified copy of the manufacturer's Good Manufacturing Practices (GMP) compliance, product specifications, lab test reports, and a sample of the drug. (dda.gov.np)
3. Estimated Timeline for Registration: The registration process can take several months, depending on the complexity of the application and the need for additional information or inspections.
4. Import Tariff/Duty for HS 30049011: Nepal imposes an import duty of 14% for SAARC countries and 15% for other countries on pharmaceutical products under HS Code 30049011. (naya.com.np)
Malaysia
1. Drug Regulatory Authority: National Pharmaceutical Regulatory Agency (NPRA).
2. Import Registration Process: Importers must register pharmaceutical products with the NPRA, providing comprehensive documentation, including product details, manufacturing information, and evidence of compliance with international standards. (npra.gov.my)
3. Estimated Timeline for Registration: The registration process typically takes 6-12 months, depending on the product's complexity and the completeness of the application.
4. Import Tariff/Duty for HS 30049011: Malaysia generally imposes a 0% import duty on pharmaceutical products under HS Code 30049011, facilitating access to medicines.
United Arab Emirates (UAE)
1. Drug Regulatory Authority: Ministry of Health and Prevention (MOHAP).
2. Import Registration Process: Importers must register pharmaceutical products with MOHAP, submitting detailed documentation, including product composition, manufacturing processes, and compliance with GMP standards.
3. Estimated Timeline for Registration: The registration process can take 6-9 months, subject to the product's nature and the thoroughness of the application.
4. Import Tariff/Duty for HS 30049011: The UAE imposes a 5% import duty on pharmaceutical products under HS Code 30049011, with certain exemptions for essential medicines.
Germany
1. Drug Regulatory Authority: Federal Institute for Drugs and Medical Devices (BfArM).
2. Import Registration Process: Importers must obtain a marketing authorization from BfArM, providing extensive documentation, including clinical data, manufacturing details, and proof of compliance with European Union regulations.
3. Estimated Timeline for Registration: The registration process typically takes 12-15 months, depending on the product's complexity and the completeness of the application.
4. Import Tariff/Duty for HS 30049011: Germany, as part of the European Union, imposes a 0% import duty on pharmaceutical products under HS Code 30049011, promoting the availability of medicines.
Demand Drivers & Market Opportunity
TransData Nexus Asava buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Asava in importing countries is driven by several factors:
1. Disease Prevalence: Asava formulations are traditionally used to address various health conditions prevalent in these regions, such as digestive disorders, respiratory issues, and general wellness. The increasing awareness and acceptance of Ayurvedic medicine contribute to sustained demand.
2. Government Healthcare Programs: Many countries are integrating traditional medicine into their national healthcare systems. For instance, Nepal's National Health Policy emphasizes the promotion of Ayurvedic medicine, leading to increased procurement and utilization of products like Asava.
3. Universal Health Coverage Expansion: Countries like Malaysia and Mauritius are expanding their universal health coverage, incorporating alternative and traditional medicines to provide comprehensive healthcare solutions. This expansion creates opportunities for Asava to be included in public health programs.
4. WHO Essential Medicines List Procurement: While Asava may not be listed on the WHO Essential Medicines List, the growing recognition of traditional medicines' role in primary healthcare encourages their inclusion in national essential medicines lists, influencing procurement decisions.
5. Tender-Based Purchasing: Government and private healthcare institutions often procure medicines through tender systems. The repeat buyer rate of 69.4% indicates a stable demand, suggesting that Asava is a preferred choice in such procurement processes.
With 173 buyers across 37 countries, the market for Asava demonstrates a robust and expanding landscape, presenting significant opportunities for growth and increased market penetration.
Common Questions — Asava Buyers & Importers
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
QWho is the largest asava buyer importing from India?
Based on import volume and value, REGENCY PHARMA LIMITED (MAURITIUS) leads with $152.4K in imports and a 33.3% market share — the highest of any single asava importer. LUNA TRADING CO.PVT.LTD (NEPAL) and SENTIASA SINAR JAYA RESOURCES (MALAYSIA) are the next largest buyers.
QHow many companies buy asava from India?
There are 173 active asava buyers importing from India, with a combined market of $458.0K across 1,399 shipments to 37 countries. The top 5 buyers hold 55.2% of total import value, while the remaining 168 buyers handle the other 44.8%.
QWhich countries import the most asava from India?
The top importing countries for asava from India are Mauritius (45.4%), Nepal (14.0%), Malaysia (10.3%), United Arab Emirates (9.1%), Germany (5.0%). These markets represent the largest demand centres for Indian pharmaceutical exports of asava, with buyers ranging from government procurement agencies to private pharmaceutical distributors.
QWhat is the average import order value for asava from India?
The average import transaction value for asava from India is $327, with an average unit price of $3.41 per unit. Order sizes vary significantly based on buyer type — government tenders tend to be larger while private distributors place more frequent smaller orders.
Buyer Segment Analysis
TransData Nexus Asava buyer market intelligence
1Buyer Segment Analysis
The importers of Asava from India can be categorized into several segments:
1. Distributors: Entities like REGENCY PHARMA LIMITED (Mauritius) and LUNA TRADING CO. PVT. LTD (Nepal) function as intermediaries, purchasing Asava in bulk to supply to local retailers, healthcare providers, or other businesses. Their buying behavior typically involves large, regular orders to maintain a steady supply chain.
2. Retail Chains: Companies such as SUVEDA INC. D/B/A KERALA AYURVEDA (United States) and KOTTAKKAL AYURVEDIC MEDICALS LLC (United Arab Emirates) operate retail outlets specializing in Ayurvedic products. They procure Asava to stock their shelves, with order patterns influenced by consumer demand and seasonal trends.
3. Re-exporters: Firms like SENTIASA SINAR JAYA RESOURCES (Malaysia) and SUCCESS ADVANCE TRADING (Malaysia) may import Asava with the intent to re-export to neighboring countries or regions. Their purchasing behavior is often characterized by opportunistic buying, capitalizing on market demands in various territories.
4. Contract Manufacturers: Entities such as SWASTHYAM AYURVEDA HANDEL (Germany) might import Asava as a component for further processing or inclusion in proprietary formulations. Their orders are typically aligned with production schedules and client requirements.
Understanding these segments is crucial for tailoring marketing strategies, forecasting demand, and optimizing supply chain logistics.
Country-Specific Import Regulations
TransData Nexus Asava buyer market intelligence
1Country-Specific Import Regulations
Mauritius
1. Drug Regulatory Authority: Mauritius Pharmacy Board.
2. Import Registration Process: Importers must obtain a pharmaceutical import license from the Mauritius Pharmacy Board, ensuring compliance with local standards and regulations.
3. Estimated Timeline for Registration: The registration process typically takes 4-6 weeks, subject to the completeness of the application and any required inspections.
4. Import Tariff/Duty for HS 30049011: Mauritius imposes a standard import duty of 0% on pharmaceutical products under HS Code 30049011, promoting access to essential medicines.
Nepal
1. Drug Regulatory Authority: Department of Drug Administration (DDA).
2. Import Registration Process: Importers must submit an application to the DDA, including a certified copy of the manufacturer's Good Manufacturing Practices (GMP) compliance, product specifications, lab test reports, and a sample of the drug. (dda.gov.np)
3. Estimated Timeline for Registration: The registration process can take several months, depending on the complexity of the application and the need for additional information or inspections.
4. Import Tariff/Duty for HS 30049011: Nepal imposes an import duty of 14% for SAARC countries and 15% for other countries on pharmaceutical products under HS Code 30049011. (naya.com.np)
Malaysia
1. Drug Regulatory Authority: National Pharmaceutical Regulatory Agency (NPRA).
2. Import Registration Process: Importers must register pharmaceutical products with the NPRA, providing comprehensive documentation, including product details, manufacturing information, and evidence of compliance with international standards. (npra.gov.my)
3. Estimated Timeline for Registration: The registration process typically takes 6-12 months, depending on the product's complexity and the completeness of the application.
4. Import Tariff/Duty for HS 30049011: Malaysia generally imposes a 0% import duty on pharmaceutical products under HS Code 30049011, facilitating access to medicines.
United Arab Emirates (UAE)
1. Drug Regulatory Authority: Ministry of Health and Prevention (MOHAP).
2. Import Registration Process: Importers must register pharmaceutical products with MOHAP, submitting detailed documentation, including product composition, manufacturing processes, and compliance with GMP standards.
3. Estimated Timeline for Registration: The registration process can take 6-9 months, subject to the product's nature and the thoroughness of the application.
4. Import Tariff/Duty for HS 30049011: The UAE imposes a 5% import duty on pharmaceutical products under HS Code 30049011, with certain exemptions for essential medicines.
Germany
1. Drug Regulatory Authority: Federal Institute for Drugs and Medical Devices (BfArM).
2. Import Registration Process: Importers must obtain a marketing authorization from BfArM, providing extensive documentation, including clinical data, manufacturing details, and proof of compliance with European Union regulations.
3. Estimated Timeline for Registration: The registration process typically takes 12-15 months, depending on the product's complexity and the completeness of the application.
4. Import Tariff/Duty for HS 30049011: Germany, as part of the European Union, imposes a 0% import duty on pharmaceutical products under HS Code 30049011, promoting the availability of medicines.
Demand Drivers & Market Opportunity
TransData Nexus Asava buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Asava in importing countries is driven by several factors:
1. Disease Prevalence: Asava formulations are traditionally used to address various health conditions prevalent in these regions, such as digestive disorders, respiratory issues, and general wellness. The increasing awareness and acceptance of Ayurvedic medicine contribute to sustained demand.
2. Government Healthcare Programs: Many countries are integrating traditional medicine into their national healthcare systems. For instance, Nepal's National Health Policy emphasizes the promotion of Ayurvedic medicine, leading to increased procurement and utilization of products like Asava.
3. Universal Health Coverage Expansion: Countries like Malaysia and Mauritius are expanding their universal health coverage, incorporating alternative and traditional medicines to provide comprehensive healthcare solutions. This expansion creates opportunities for Asava to be included in public health programs.
4. WHO Essential Medicines List Procurement: While Asava may not be listed on the WHO Essential Medicines List, the growing recognition of traditional medicines' role in primary healthcare encourages their inclusion in national essential medicines lists, influencing procurement decisions.
5. Tender-Based Purchasing: Government and private healthcare institutions often procure medicines through tender systems. The repeat buyer rate of 69.4% indicates a stable demand, suggesting that Asava is a preferred choice in such procurement processes.
With 173 buyers across 37 countries, the market for Asava demonstrates a robust and expanding landscape, presenting significant opportunities for growth and increased market penetration.
Official References & Regulatory Resources
- European Medicines Agency
- WHO Essential Medicines
- Invest India — Pharma Sector
- India Trade Statistics (DGFT)
Verify import regulations and drug registration requirements with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Identification: 173 global importers of Asava identified from consignee fields in DGFT shipping bill records.
- 2.Import Value Analysis: Total import value aggregated from 1,399 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Geographic Distribution: Buyer imports distributed across 37 destination countries with market share per buyer calculated from capped values.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
1,399 Verified Shipments
173 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists