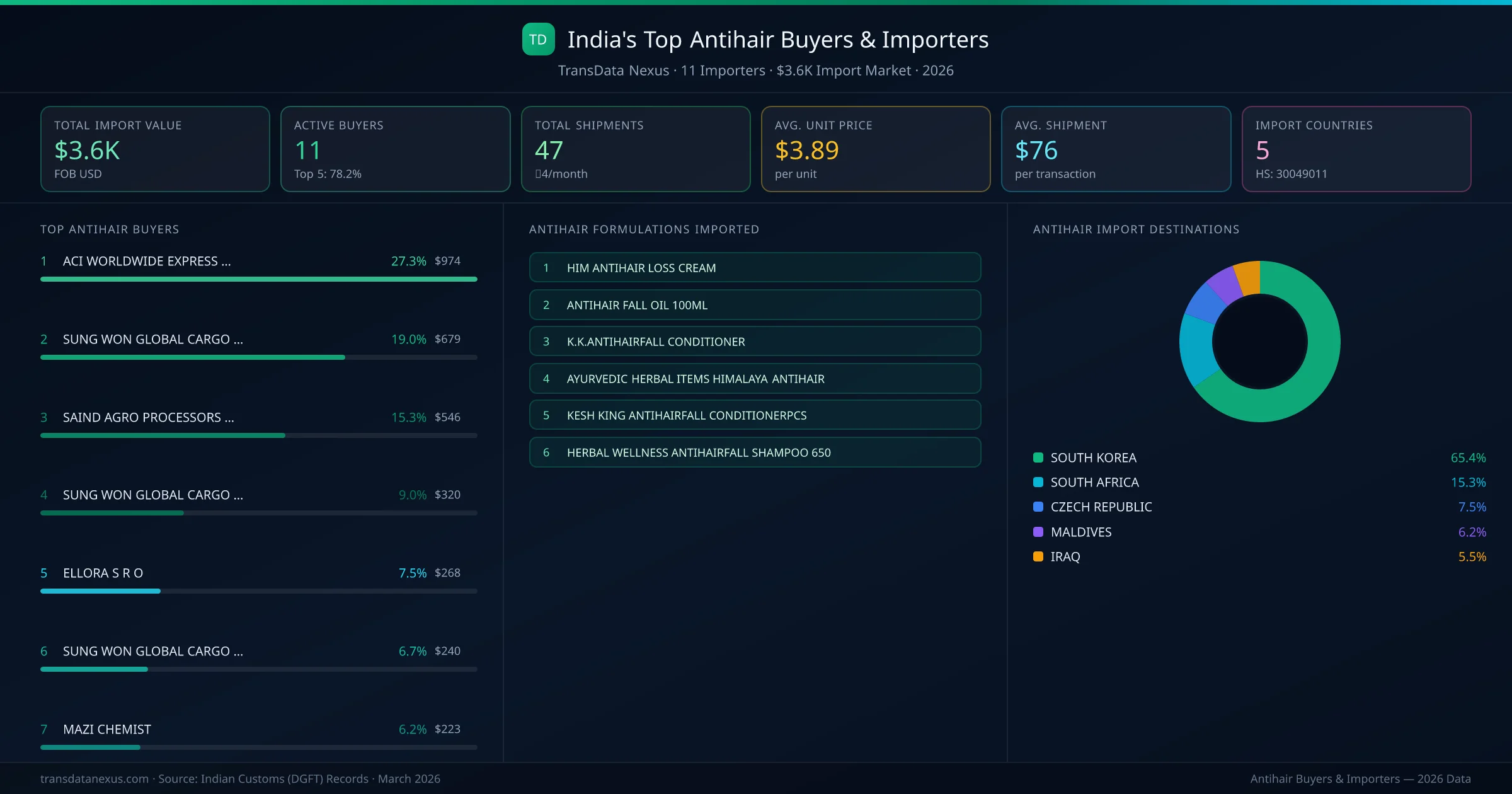
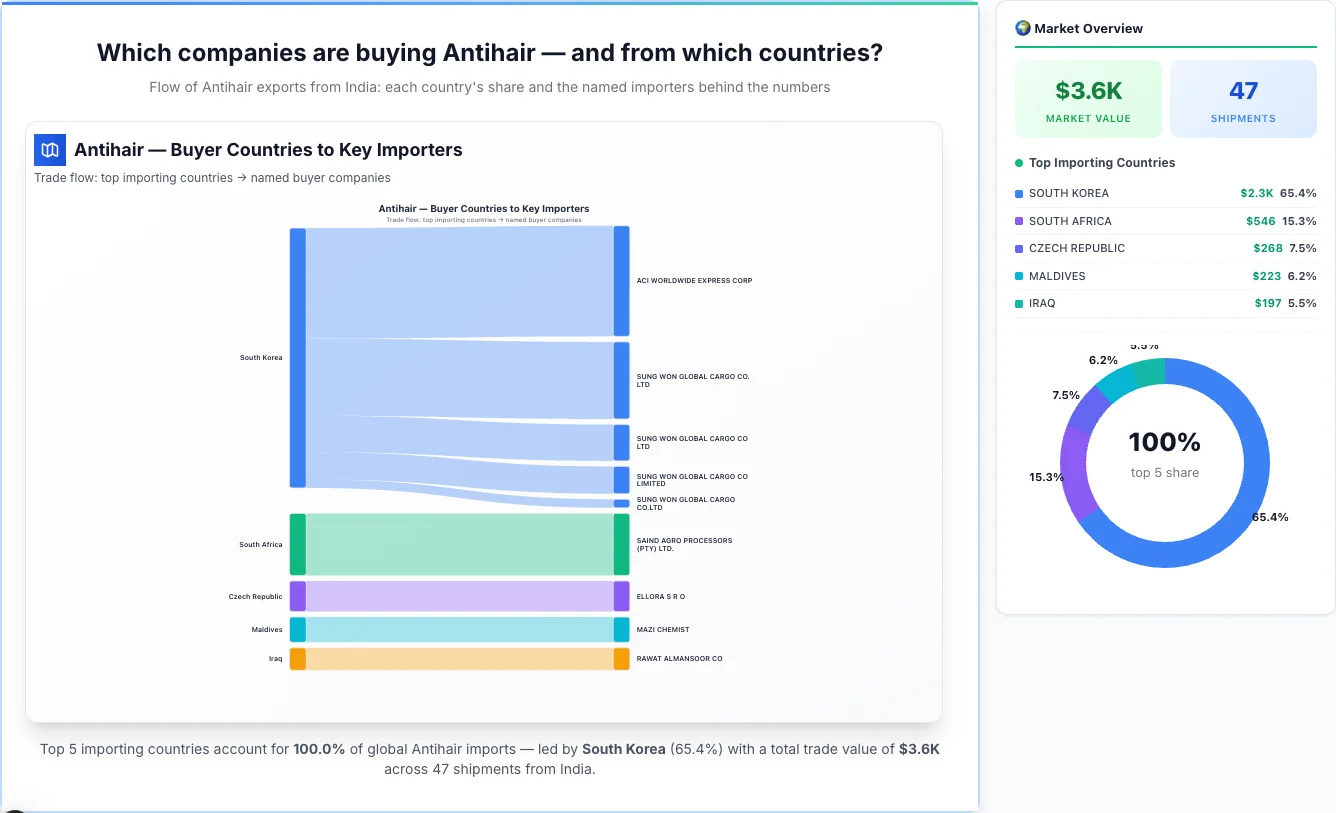
South Korea
1. Drug Regulatory Authority: Ministry of Food and Drug Safety (MFDS).
2. Import Registration Process: Importers must submit a New Drug Application (NDA) to the MFDS, including data on safety, efficacy, and Good Manufacturing Practice (GMP) compliance. The MFDS reviews the application and may request additional information. Upon approval, a product license is issued.
3. Estimated Timeline for Registration: The approval process typically takes several months, depending on the completeness of the application and any additional data requests.
4. Import Tariff/Duty for HS 30049011: Pharmaceutical products under HS code 30049011 are generally subject to a 0% import tariff in South Korea.
South Africa
1. Drug Regulatory Authority: South African Health Products Regulatory Authority (SAHPRA).
2. Import Registration Process: Importers must obtain approval from SAHPRA, which involves submitting an application detailing the product's safety, efficacy, and quality. An import license is required, and the product must comply with local regulations.
3. Estimated Timeline for Registration: The registration process can take several months, depending on the complexity of the application and the need for additional information.
4. Import Tariff/Duty for HS 30049011: Pharmaceutical products under HS code 30049011 are typically subject to a 0% import tariff in South Africa.
Czech Republic
1. Drug Regulatory Authority: State Institute for Drug Control (SÚKL).
2. Import Registration Process: Importers must submit a marketing authorization application to SÚKL, including comprehensive data on the product's quality, safety, and efficacy. The product must comply with European Union regulations.