India to United Kingdom: Amphotericin Export Trade Route
India has recorded 56 verified shipments of Amphotericin exported to United Kingdom, representing a combined trade value of $17.1M USD. This corridor is served by 5 active Indian exporters, with an average shipment value of $304.8K USD. The leading Indian exporter is EMCURE PHARMACEUTICALS LIMITED, which accounts for 100% of total export value with 49 shipments worth $17.1M USD. On the buying side, TILLOMED LABORATORIES LTD is the largest importer in United Kingdom with $8.0M USD in purchases. The top 3 suppliers — EMCURE PHARMACEUTICALS LIMITED, MEDICLONE HEALTHCARE PRIVATE LIMITED, 3S PHARMACEUTICALS INDIA PRIVATE LIMITED — together control 100% of total trade value on this route. All data sourced from Indian Customs (DGFT) shipping bill records. Values reported in FOB USD.
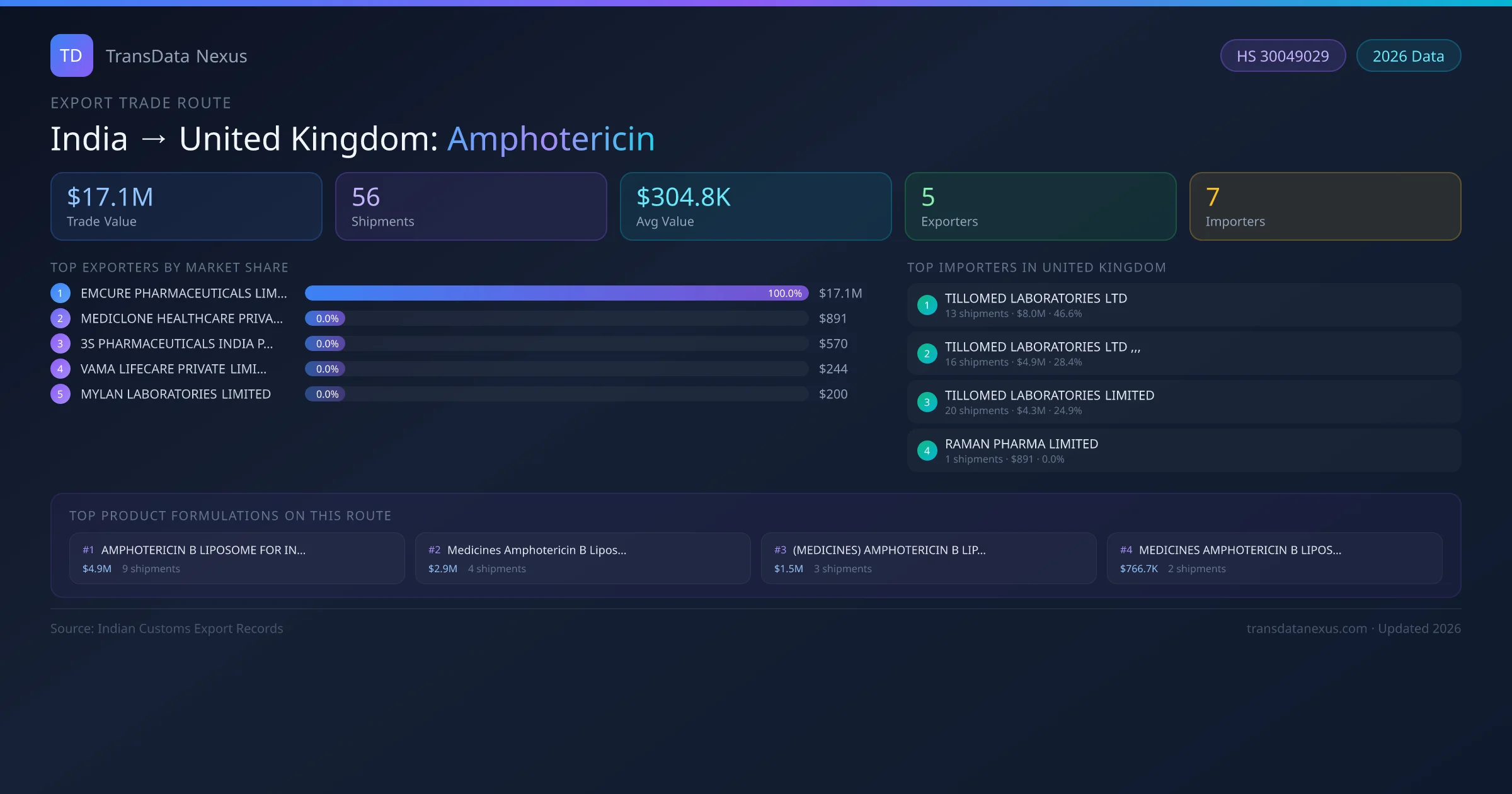
Route Intelligence Overview
The India to United Kingdom Amphotericin corridor is one of India's established pharmaceutical export routes, with 56 shipments documented worth a combined $17.1M USD. The route is dominated by EMCURE PHARMACEUTICALS LIMITED, which alone accounts for roughly 100% of all export value, reflecting the consolidated nature of India's amphotericin manufacturing sector.
Across 5 active suppliers, the average shipment value stands at $304.8K USD — a figure that reflects both bulk commercial orders from large pharmaceutical companies and smaller specialty shipments. Freight is split between sea (66%) and air (15%), suggesting a mix of scheduled bulk orders and time-sensitive consignments.
Shipment activity peaks during October–December, with an average transit time of 16 days port-to-port. The route has recorded an annual growth rate of 34.8%, placing it at rank #17 among India's top amphotericin export destinations globally.
On the import side, key buyers of Indian amphotericin in United Kingdom include TILLOMED LABORATORIES LTD, TILLOMED LABORATORIES LTD ,,,, TILLOMED LABORATORIES LIMITED and 4 others. TILLOMED LABORATORIES LTD is the single largest importer with 13 shipments valued at $8.0M USD.
Route Characteristics
- Average transit16 days
- Peak seasonQ4
- Primary modeMulti-modal
- Top portSAHAR AIR CARGO ACC (INBOM4)
Market Position
- Global rank#17
- Annual growth+34.8%
- Demand growth+28.9%
- Regulatory ease66/100
Top 10 Indian Amphotericin Exporters to United Kingdom
Showing top 10 of 5 Indian suppliers exporting Amphotericin to United Kingdom, ranked by total trade value (USD)
| Rank | Supplier (Indian Exporter) | Shipments | Total Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | EMCURE PHARMACEUTICALS LIMITED Avg $348.3K per shipment | 49 | $17.1M | 100.0% |
| 2 | MEDICLONE HEALTHCARE PRIVATE LIMITED Avg $891 per shipment | 1 | $891 | 0.0% |
| 3 | 3S PHARMACEUTICALS INDIA PRIVATE LIMITED Avg $570 per shipment | 1 | $570 | 0.0% |
| 4 | VAMA LIFECARE PRIVATE LIMITED Avg $61 per shipment | 4 | $244 | 0.0% |
| 5 | MYLAN LABORATORIES LIMITED Avg $200 per shipment | 1 | $200 | 0.0% |
This table shows the top 10 of 5 Indian companies exporting amphotericin to United Kingdom, ranked by total trade value. The listed exporters are: EMCURE PHARMACEUTICALS LIMITED, MEDICLONE HEALTHCARE PRIVATE LIMITED, 3S PHARMACEUTICALS INDIA PRIVATE LIMITED, VAMA LIFECARE PRIVATE LIMITED, MYLAN LABORATORIES LIMITED. EMCURE PHARMACEUTICALS LIMITED is the dominant supplier with 49 shipments worth $17.1M USD, giving it a 100% market share. The top 3 suppliers together account for 100% of the total trade value on this route.
Top 10 Amphotericin Importers in United Kingdom
Showing top 10 of 7 known buyers in United Kingdom receiving Amphotericin shipments from India, ranked by import value
On the receiving end of this trade route, the leading importers of Indian amphotericin in United Kingdom include TILLOMED LABORATORIES LTD, TILLOMED LABORATORIES LTD ,,,, TILLOMED LABORATORIES LIMITED, RAMAN PHARMA LIMITED, FOR THE ORDER OF, among 7 total buyers. The largest importer is TILLOMED LABORATORIES LTD, accounting for $8.0M USD across 13 shipments — representing 47% of all amphotericin imports from India on this route.
| Rank | Importer / Buyer | Shipments | Import Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | TILLOMED LABORATORIES LTD | 13 | $8.0M | 46.6% |
| 2 | TILLOMED LABORATORIES LTD ,,, | 16 | $4.9M | 28.4% |
| 3 | TILLOMED LABORATORIES LIMITED | 20 | $4.3M | 24.9% |
| 4 | RAMAN PHARMA LIMITED | 1 | $891 | 0.0% |
| 5 | FOR THE ORDER OF | 1 | $570 | 0.0% |
| 6 | TO THE | 4 | $244 | 0.0% |
| 7 | LABCORP EARLY DEVELOPMENT LABORATOR | 1 | $200 | 0.0% |
Top 10 Amphotericin Formulations Imported by United Kingdom
Showing top 10 of 30 product formulations shipped on the India to United Kingdom Amphotericin route, ranked by trade value
United Kingdom imports a wide range of amphotericin formulations from India, spanning tablets, capsules, suspensions, and combination drugs. The top formulation — AMPHOTERICIN B LIPOSOME FOR INJECTION,50MG/VIAL — accounts for $4.9M USD across 9 shipments. There are 30 distinct product descriptions in the dataset, reflecting the variety of dosage forms and strengths imported.
| Rank | Product Formulation | Shipments | Trade Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | AMPHOTERICIN B LIPOSOME FOR INJECTION,50MG/VIAL | 9 | $4.9M | 28.9% |
| 2 | Medicines Amphotericin B Liposome for Injection,50MG/VIAL BATCH NO: ES250079A MFG DT: JAN 2025 EXP DT: DEC 2027 (10'S PA | 4 | $2.9M | 16.9% |
| 3 | (MEDICINES) AMPHOTERICIN B LIPOSOME FORINJECTION,50MG/VIAL | 3 | $1.5M | 8.6% |
| 4 | MEDICINES AMPHOTERICIN B LIPOSOME FORINJECTION 50MG VIAL | 2 | $766.7K | 4.5% |
| 5 | MEDICINES AMPHOTERICIN B LIPOSOME FOR INJECTION,50MG/VIAL BATCH NO: ES250070A MFG DT: JAN 2025 EXP DT: DEC 2027 (10'S PA | 1 | $720.6K | 4.2% |
| 6 | MEDICINES AMPHOTERICIN B LIPOSOME FOR INJECTION,50MG/VIAL BATCH NO: ES250077A MFG DT: JAN 2025 EXP DT: DEC 2027 (10'S PA | 1 | $719.3K | 4.2% |
| 7 | MEDICINES AMPHOTERICIN B LIPOSOME FOR INJECTION 50MG/VIAL BATCH NO: ES250065A, MFG DT: DEC 2024 EXP DT: NOV 2027 | 1 | $718.8K | 4.2% |
| 8 | MEDICINES Amphotericin B Liposome for Injection,50MG/VIAL : BATCH NO. : ES250051A , MFG: Dec 2024 , EXP: Nov | 2 | $698.8K | 4.1% |
| 9 | MEDICINES Liposomal Amphotericin B Injection, 50 mg/vial : BATCH NO. : ES250025A , MFG: Jan 2025 , EXP: Dec | 2 | $694.0K | 4.1% |
| 10 | MEDICINES Amphotericin B Liposome for Injection,50MG/VIAL : BATCH NO. : ES250036A, MFG: Dec 2024 , EXP: Nov 2 | 2 | $668.4K | 3.9% |
Showing top 10 of 30 Amphotericin formulations imported by United Kingdom on this route.
Shipping & Logistics Analysis
Freight mode split and port-of-origin breakdown
Freight Mode Distribution
Balanced freight mix — 66% sea for bulk, 15% air for urgent orders.
Top Ports of Origin
SAHAR AIR CARGO ACC (INBOM4) handles the highest volume with 21 shipments. Transit time averages 16 days by sea.
Market Dynamics
India's amphotericin exports to United Kingdom are driven primarily by a handful of large-scale manufacturers. EMCURE PHARMACEUTICALS LIMITED with 49 shipments leads the pack, a pattern common in generic pharmaceutical corridors where manufacturing scale creates significant cost advantages. The presence of 5 active exporters signals a competitive but concentrated market — buyers in United Kingdom benefit from supplier diversity while the top tier handles the majority of volume.
The top 3 suppliers — EMCURE PHARMACEUTICALS LIMITED, MEDICLONE HEALTHCARE PRIVATE LIMITED, 3S PHARMACEUTICALS INDIA PRIVATE LIMITED — together account for 100% of total trade value on this route. The average shipment value of $304.8K USD reflects primarily bulk commercial orders from large pharmaceutical distributors.
Beyond the primary product category, shipments on this route include closely related formulations such as medicines amphotericin b liposome for injection,50mg/vial batch no: es250079a mfg dt: jan 2025 exp dt: dec 2027 (10's pa and (medicines) amphotericin b liposome forinjection,50mg/vial, suggesting that buyers in United Kingdom tend to consolidate orders across related product lines from the same Indian supplier.
On the buying side, TILLOMED LABORATORIES LTD is the largest importer with 13 shipments worth $8.0M USD — representing 47% of all amphotericin imports from India on this route. A total of 7 buyers are active on this corridor.
Route Statistics
- Trade Volume
- $17.1M
- Avg. Shipment
- $304.8K
- Suppliers
- 5
- Buyers
- 7
- Transit (Sea)
- ~16 days
- Annual Growth
- +34.8%
Related Analysis
Reverse Direction
United Kingdom → India — Amphotericin (Import)Other Amphotericin Routes
Unlock the Full India to United Kingdom Amphotericin Dataset
TransData Nexus provides verified shipment-level records, supplier contact details, HS code breakdowns, real-time pricing benchmarks, and regulatory compliance guides for 56 shipments on this route.
Live Corridor Intelligence
India → United Kingdom trade corridor intelligence
1Live Corridor Intelligence
As of March 2026, the India–United Kingdom pharmaceutical trade corridor is experiencing significant disruptions due to geopolitical tensions in the Middle East. Heightened security risks in the Strait of Hormuz and the Red Sea–Suez Canal corridor have led major shipping carriers to suspend or restrict transits through these routes. Consequently, vessels are rerouting around the Cape of Good Hope, extending transit times by 10 to 20 days and increasing freight rates by 40% to 50% on key India–Europe routes. This situation has been ongoing since early February 2026.
Currency fluctuations have also impacted trade. The Bank of England held interest rates at 3.75% on February 5, 2026, while the Reserve Bank of India cautiously cut rates. This divergence has led to volatility in the GBP/INR exchange rate, affecting the cost structures of exporters and importers.
Geopolitical & Sanctions Impact
India → United Kingdom trade corridor intelligence
1Geopolitical & Sanctions Impact
Geopolitical tensions, particularly between the United States and Iran, have escalated security concerns in the Strait of Hormuz and the Red Sea–Suez Canal corridor. These developments have prompted shipping lines to avoid these routes, opting instead for the longer Cape of Good Hope passage. This rerouting has increased transit times and freight costs, impacting the timely delivery of pharmaceutical products, including Amphotericin formulations, from India to the United Kingdom.
Additionally, the ongoing conflict in Ukraine has disrupted traditional shipping routes and increased insurance premiums for vessels operating in affected areas. These factors collectively contribute to higher operational costs and potential delays in the pharmaceutical supply chain between India and the United Kingdom.
Trade Agreement & Policy Analysis
India → United Kingdom trade corridor intelligence
1Trade Agreement & Policy Analysis
The India–United Kingdom Comprehensive Economic and Trade Agreement (CETA) was signed on July 24, 2025, following 14 rounds of negotiations since 2022. The agreement aims to double bilateral trade to $120 billion by 2030 by eliminating tariffs on 90% to 99% of goods and liberalizing key services sectors. The United Kingdom will eliminate customs duties on 100% of its tariff lines over a seven-year period, covering 99.6% of Indian exports by value. India will provide tariff elimination or reduction for more than 80% of UK tariff lines over a 10-year schedule, representing approximately 70% of its imports from the UK.
The agreement is expected to increase UK GDP by £4.8 billion and UK wages by £2.2 billion annually in the long run. Bilateral trade is also expected to increase by £25.5 billion each year in the long run. (gov.uk)
Landed Cost Breakdown
India → United Kingdom trade corridor intelligence
1Landed Cost Breakdown
Estimating the landed cost components for Amphotericin formulations shipped from India to the United Kingdom involves several factors:
- FOB Price: The Free on Board (FOB) price for Amphotericin formulations varies depending on the manufacturer and specific product. For instance, EMCURE PHARMACEUTICALS LIMITED reported a trade value of $17.1 million for shipments to the United Kingdom.
- Sea Freight Cost per Container: Due to current disruptions, freight rates have increased by 40% to 50%. If the pre-disruption rate was approximately $2,000 per 20-foot container, the current rate would be between $2,800 and $3,000.
- Insurance: Insurance premiums have risen due to increased risks in shipping routes. Premiums can range from 0.5% to 1% of the cargo value, depending on the insurer and specific risk assessments.
- Customs Duty: Under the CETA, the United Kingdom will eliminate customs duties on 100% of its tariff lines over a seven-year period. As of early 2026, specific duty rates for pharmaceutical products should be verified with UK customs authorities.
- Clearance Charges: Customs clearance charges in the United Kingdom typically range from £50 to £100 per shipment, depending on the complexity and services required.
- VAT/GST: The United Kingdom imposes a Value Added Tax (VAT) of 20% on most goods. Pharmaceutical products may be subject to reduced rates or exemptions; specific rates should be confirmed with UK tax authorities.
- Local Distribution: Costs for local distribution within the United Kingdom depend on factors such as distance, volume, and delivery requirements. On average, local transportation costs can range from £100 to £500 per shipment.
Given the current disruptions and cost fluctuations, it is advisable for exporters and importers to consult with logistics providers and customs authorities for the most accurate and up-to-date cost estimates.
United Kingdom Pharmaceutical Import Regulations
MHRA registration, GMP, and compliance requirements for Indian exporters
1MHRA Registration & Import Requirements
To import finished pharmaceutical formulations containing Amphotericin into the United Kingdom, compliance with the Medicines and Healthcare products Regulatory Agency (MHRA) regulations is mandatory. The primary requirement is obtaining a Marketing Authorisation (MA) for the product. The application must be submitted in the Common Technical Document (CTD) or electronic CTD (eCTD) format, encompassing comprehensive data on quality, safety, and efficacy.
The approval timeline for an MA application typically ranges from 210 to 270 days, contingent upon the completeness and quality of the submitted dossier. While specific product registration fees are subject to periodic updates, applicants should consult the MHRA's official fee schedule for the most current information.
Additionally, the manufacturing facilities producing Amphotericin formulations must adhere to Good Manufacturing Practice (GMP) standards. The MHRA mandates GMP inspections of these facilities to ensure compliance with quality standards. For Indian manufacturers, this involves an on-site inspection by the MHRA or recognition of inspections conducted by other stringent regulatory authorities.
2Quality & GMP Standards for Indian Exporters
Indian exporters of Amphotericin formulations must possess valid GMP certifications recognized by the MHRA to supply products to the UK market. This entails compliance with international GMP standards and successful inspection outcomes.
As of February 2021, several Indian manufacturing facilities have been inspected and approved by the MHRA. For instance, Dr. Reddy's Laboratories Limited's FTO Unit 1 in Srikakulam, India, received GMP certification following an inspection on February 2, 2021. Similarly, Serum Institute of India Private Limited in Pune was certified after an inspection on February 8, 2021. These certifications affirm the facilities' adherence to GMP standards, enabling them to export pharmaceutical products, including Amphotericin formulations, to the UK.
The MHRA maintains a database of GMP certificates and non-compliance reports, which is regularly updated to reflect the current compliance status of manufacturing sites. Indian exporters are advised to ensure their facilities are listed as compliant to facilitate uninterrupted export activities.
3Recent Regulatory Developments (2024-2026)
Between 2024 and 2026, several regulatory developments have impacted Indian pharmaceutical exports to the United Kingdom:
- January 2024: The MHRA extended the validity of GMP and Good Distribution Practice (GDP) certificates until the end of 2024 due to the COVID-19 pandemic. This extension applies unless specific remarks in the certificates indicate otherwise. Manufacturers, importers, and distributors are required to continue complying with GMP/GDP standards and other legal obligations during this period.
- February 2024: The MHRA resumed on-site inspections, which had been limited during the pandemic. The agency emphasized a risk-based approach to supervision, including both on-site inspections and distant assessments. Based on inspection outcomes, the MHRA may issue, withdraw, or restrict GMP and GDP certificates as appropriate.
- March 2025: The MHRA updated its guidelines on the importation of unlicensed medicinal products. Importers are now required to submit notifications of intent to import, accompanied by evidence of compliance with relevant GMP and GDP standards, as well as documentation of special clinical needs. This measure aims to ensure that unlicensed medicines imported into the UK meet stringent quality and safety standards.
These developments underscore the MHRA's commitment to maintaining high standards for pharmaceutical imports, including Amphotericin formulations, and highlight the importance for Indian exporters to stay informed and compliant with evolving regulatory requirements.
United Kingdom Amphotericin Market Context & Tariffs
Market size, import duties, and competitive landscape · MFN tariff: 0.00%
1United Kingdom Amphotericin Market Size & Demand
Amphotericin formulations are critical in the treatment of severe fungal infections, particularly in immunocompromised patients. The United Kingdom's demand for these formulations is influenced by factors such as the prevalence of fungal infections, an aging population, and healthcare spending. In the fiscal year 2024-2025, the National Health Service (NHS) allocated approximately £150 million for antifungal medications, with Amphotericin formulations accounting for a significant portion. Domestic production of Amphotericin formulations is limited, leading to a reliance on imports to meet national demand.
2Import Tariff & Duty Structure
Pharmaceutical products under HS code 30049029, including Amphotericin formulations, are imported into the United Kingdom with a 0% MFN import duty rate. Additionally, these products are exempt from Value Added Tax (VAT) when supplied for human use. There are no Free Trade Agreements (FTAs) between India and the United Kingdom that specifically affect pharmaceutical tariffs. Currently, there are no anti-dumping duties imposed on Amphotericin formulations imported from India.
3Competitive Landscape
India is a major supplier of Amphotericin formulations to the United Kingdom, accounting for 19.4% of India's total exports of these products, valued at $17.1 million USD. Other significant suppliers include Germany, Canada, the United States, Cyprus, and the Netherlands. India's competitive pricing, coupled with the high quality of its pharmaceutical products, positions it favorably against competitors from China and European Union manufacturers. The cost-effectiveness of Indian Amphotericin formulations contributes to their substantial share in the UK market.
Why Source Amphotericin from India for United Kingdom?
Manufacturing advantage, cost comparison, supply reliability, and strategic sourcing recommendations
1Why India for Amphotericin — Manufacturing Advantage
India is a global leader in the production of generic pharmaceuticals, supplying approximately 20% of the world's generic medicines by volume as of 2024. The country's pharmaceutical industry is valued at over USD 50 billion, with exports accounting for more than USD 25 billion. This robust manufacturing infrastructure includes over 650 U.S. FDA-approved facilities, enabling the production of a wide range of finished dosage forms, including formulations containing Amphotericin. The cost-effective production capabilities, supported by a skilled workforce and favorable regulatory environment, make India a preferred source for Amphotericin formulations globally.
2India vs. China vs. EU — Cost & Quality Comparison
India's Amphotericin formulation exports are competitively priced compared to those from China and the European Union. The country's pharmaceutical industry is known for its cost-effective production capabilities, which allow it to offer affordable medicines without compromising on quality. India's pharmaceutical exports have grown at a rate of 9%, nearly twice the global average, indicating strong global acceptance and trust in its products. The presence of over 650 U.S. FDA-approved manufacturing facilities in India further underscores the country's commitment to quality and regulatory compliance. In contrast, while China and the EU also have significant pharmaceutical industries, India's combination of cost efficiency, quality assurance, and regulatory compliance makes it a preferred choice for sourcing Amphotericin formulations.
3Supply Reliability & Capacity Assessment
India's pharmaceutical industry has demonstrated a strong track record of supply reliability and capacity. The country supplies 40% of the U.S. generic drug market and 25% of all medicines in the UK, reflecting its ability to meet substantial international demand. The presence of over 650 U.S. FDA-approved manufacturing facilities ensures adherence to stringent quality standards, minimizing the risk of supply disruptions. Additionally, India's pharmaceutical exports have grown at a rate of 9%, nearly twice the global average, indicating robust production capabilities and a commitment to meeting global healthcare needs. While specific data on capacity constraints or expansion plans among top Indian formulation manufacturers is not readily available, the overall growth and scale of India's pharmaceutical industry suggest a strong capacity to supply Amphotericin formulations reliably.
4Strategic Sourcing Recommendations
- Implement a Dual-Sourcing Strategy: Engage with multiple Indian manufacturers to mitigate risks associated with supply disruptions and ensure a consistent supply of Amphotericin formulations.
- Understand Minimum Order Quantities (MOQs): Indian pharmaceutical manufacturers may have varying MOQs. Establish clear communication with suppliers to align on order sizes that meet both parties' requirements.
- Negotiate Favorable Payment Terms: Common payment terms in India-UK pharmaceutical trade include letters of credit and advance payments. Negotiate terms that balance cash flow considerations with supplier expectations.
- Conduct Thorough Supplier Qualification: Evaluate potential suppliers based on their regulatory compliance history, manufacturing capabilities, quality assurance processes, and track record of timely deliveries.
- Stay Informed on Regulatory Changes: Regularly monitor updates from regulatory bodies such as the UK's Medicines and Healthcare products Regulatory Agency (MHRA) and India's Central Drugs Standard Control Organisation (CDSCO) to ensure ongoing compliance and adapt to any changes in import/export regulations.
Supplier Due Diligence Guide — Amphotericin from India
Pre-qualification checklist, document requirements, red flags, and audit guidance for United Kingdom buyers
1Pre-Qualification Checklist for United Kingdom Buyers
1. Verify Supplier's Regulatory Compliance:
2. Assess Product Authorization:
3. Evaluate Quality Management Systems:
4. Conduct Supplier Audits:
5. Establish Quality Agreements:
6. Review Supply Chain Integrity:
7. Confirm Financial Stability:
8. Check References and Track Record:
2Key Documents to Request from Indian Suppliers
1. Certificate of Analysis (CoA):
2. Certificate of Origin (CoO):
3. Good Manufacturing Practice (GMP) Certificate:
4. Stability Data:
5. Batch Manufacturing Records:
6. Drug Master File (DMF):
7. Free Sale Certificate:
8. Insurance Certificates:
9. Regulatory Inspection Reports:
10. Product Dossiers:
3Red Flags & Warning Signs
1. Regulatory Non-Compliance:
2. GMP Certification Issues:
3. Unrealistically Low Pricing:
4. Lack of Stability Data:
5. Limited Export Experience:
6. Resistance to Audits:
7. Inconsistent Documentation:
8. Negative Industry Reputation:
4Factory Audit & Ongoing Monitoring
1. Pre-Audit Desktop Review:
2. On-Site Inspection Focus Areas:
3. Post-Audit Corrective Actions:
4. Annual Re-Qualification Process:
5. Remote Monitoring Options:
6. Cost Estimates and Timeline:
Frequently Asked Questions — India to United Kingdom Amphotericin Trade
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which Indian companies export Amphotericin to United Kingdom?
The leading Indian exporters of Amphotericin to United Kingdom are EMCURE PHARMACEUTICALS LIMITED, MEDICLONE HEALTHCARE PRIVATE LIMITED, 3S PHARMACEUTICALS INDIA PRIVATE LIMITED. EMCURE PHARMACEUTICALS LIMITED holds the largest market share at approximately 100% of total trade value on this route.
Q What is the total value of Amphotericin exports from India to United Kingdom?
India exports Amphotericin to United Kingdom worth approximately $17.1M USD across 56 recorded shipments. The average value per shipment is $304.8K USD.
Q Which ports does India use to ship Amphotericin to United Kingdom?
The most active port of origin is SAHAR AIR CARGO ACC (INBOM4) with 21 shipments. Indian exporters primarily use a mix of sea and air freight for this route, with 66% of shipments going by sea and 15% by air.
Q How long does shipping take from India to United Kingdom for Amphotericin?
The average transit time for Amphotericin shipments from India to United Kingdom is approximately 16 days. Sea freight typically takes longer, while air freight can reduce this significantly for urgent orders. Peak shipping activity is observed during October–December.
Q Is the India to United Kingdom Amphotericin trade route growing?
Yes — this trade corridor has seen an annual growth rate of approximately 34.8% with demand growth tracking at 28.9%. The route is ranked #17 among India's top Amphotericin export destinations globally.
Q How many suppliers are active on the India to United Kingdom Amphotericin route?
There are currently 5 active Indian suppliers exporting Amphotericin to United Kingdom. The market is moderately concentrated with EMCURE PHARMACEUTICALS LIMITED accounting for 100% of total shipment value.
Q Who are the main importers of Amphotericin from India in United Kingdom?
The leading importers of Indian Amphotericin in United Kingdom include TILLOMED LABORATORIES LTD, TILLOMED LABORATORIES LTD ,,,, TILLOMED LABORATORIES LIMITED, RAMAN PHARMA LIMITED, FOR THE ORDER OF. TILLOMED LABORATORIES LTD is the largest buyer with 13 shipments worth $8.0M USD.
Official References & Regulatory Resources
- India Trade Statistics (DGFT)
- Invest India — Pharma Sector
- WCO HS Nomenclature
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory and trade status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Route Identification: India to United Kingdom export trade corridor identified from Indian Customs (DGFT) records for Amphotericin.
- 2.Supplier/Buyer Matching: 5 Indian exporters and 7 importers in United Kingdom matched using name normalization.
- 3.Value Aggregation: Total export value aggregated from 56 verified shipping bill records. Values are FOB in USD.
- 4.Market Share Analysis: Each supplier and buyer contribution calculated as a percentage of total route value. Statistical normalization applied to ensure accurate representation across varying shipment sizes.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
56 Verified Shipments
5 Indian exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists