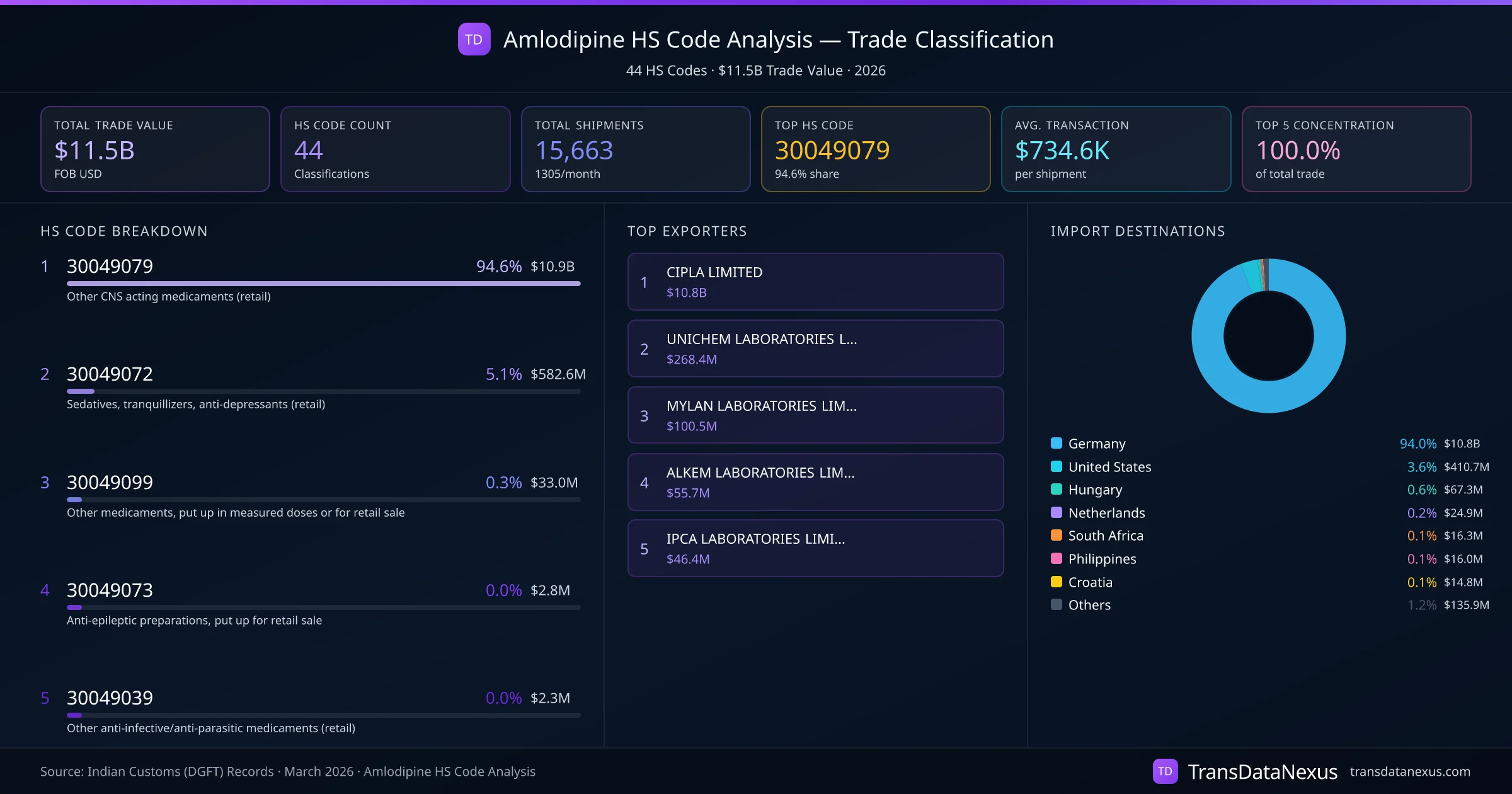
The classification of Amlodipine under the Harmonized System can present challenges, particularly when distinguishing between HS codes 3003 and 3004.
- HS Code 3003: This code pertains to "Medicaments consisting of two or more constituents which have been mixed together for therapeutic or prophylactic uses, not put up in measured doses or in forms or packings for retail sale."
- HS Code 3004: This code covers "Medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses or in forms or packings for retail sale."
Amlodipine, when formulated and packaged for retail sale in specific dosages, should be classified under HS code 3004. Misclassification can occur if the product is incorrectly declared under HS code 3003, leading to potential customs penalties, delays in clearance, and compliance issues. Given that 94.6% of Amlodipine trade utilizes HS code 30049079, it is evident that the majority of shipments are correctly classified. However, vigilance is necessary to avoid errors, especially when dealing with bulk shipments not intended for retail sale, which may fall under HS code 3003.
Recent rulings by the World Customs Organization (WCO) and national customs authorities emphasize the importance of accurate classification. For instance, a WCO case in 2025 highlighted the misclassification of bulk pharmaceutical ingredients, underscoring the need for precise declarations to ensure compliance and avoid penalties.