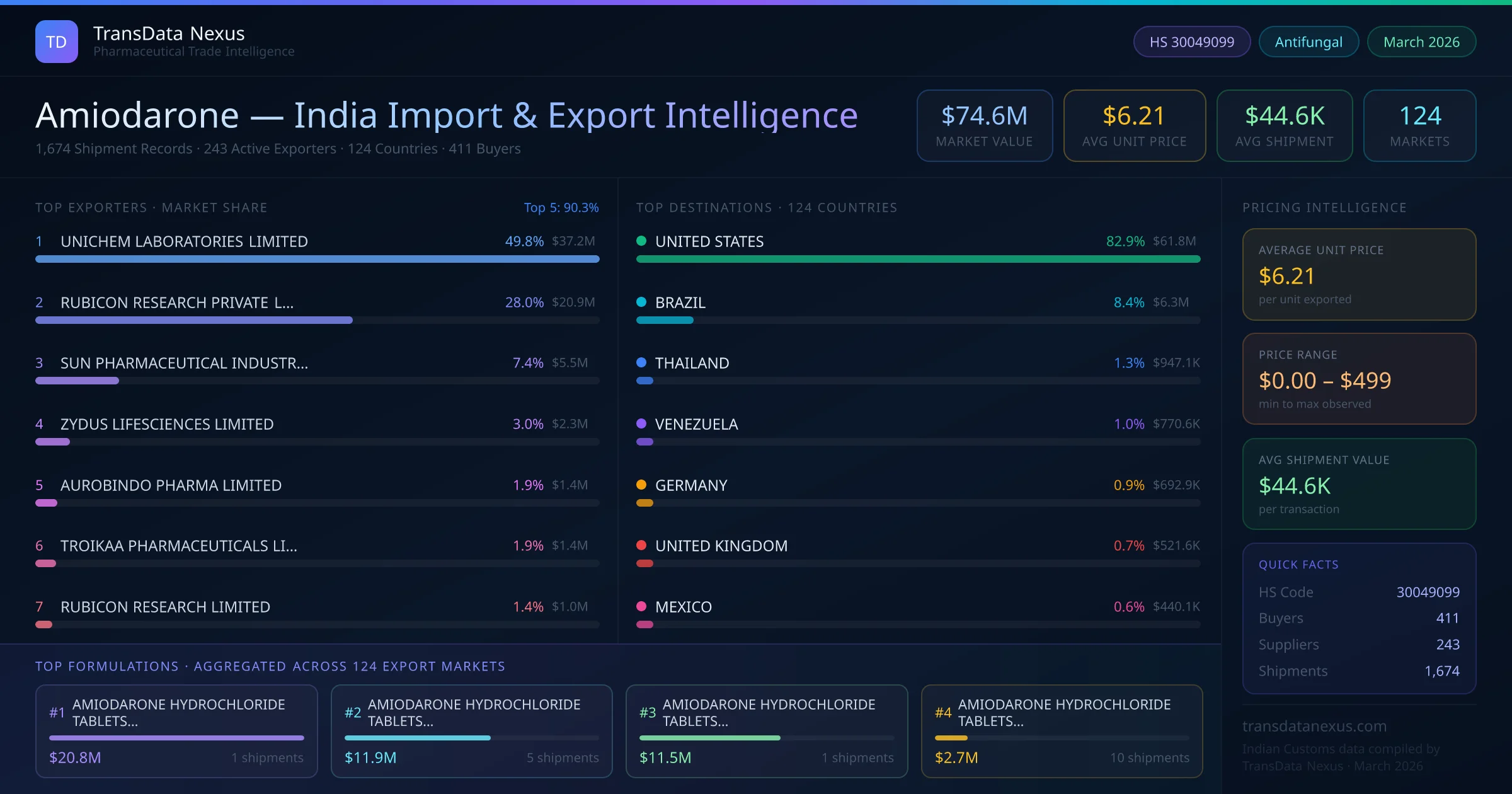
Importing finished pharmaceutical formulations containing Amiodarone into India requires compliance with the Drugs and Cosmetics Act, 1940, and the Drugs and Cosmetics Rules, 1945. The Central Drugs Standard Control Organization (CDSCO) mandates that both the manufacturing site and the product be registered prior to import. An Import Registration Certificate is necessary for each product intended for import. The application process involves submitting Form 40 or 41 to the CDSCO, along with the requisite fee and supporting documents, including a Certificate of Pharmaceutical Product (CoPP) and a Free Sale Certificate from the country of origin. The timeline for obtaining registration can vary, but it typically ranges from 6 to 12 months, depending on the completeness of the application and the regulatory review process. For Amiodarone formulations under HS Code 30049099, specific requirements include stability data demonstrating compliance with ICH guidelines, a CoA, and evidence of Good Manufacturing Practice (GMP) compliance. Additionally, a No Objection Certificate (NOC) from the manufacturer may be required to confirm that the product is not patented or under exclusive marketing rights in India.