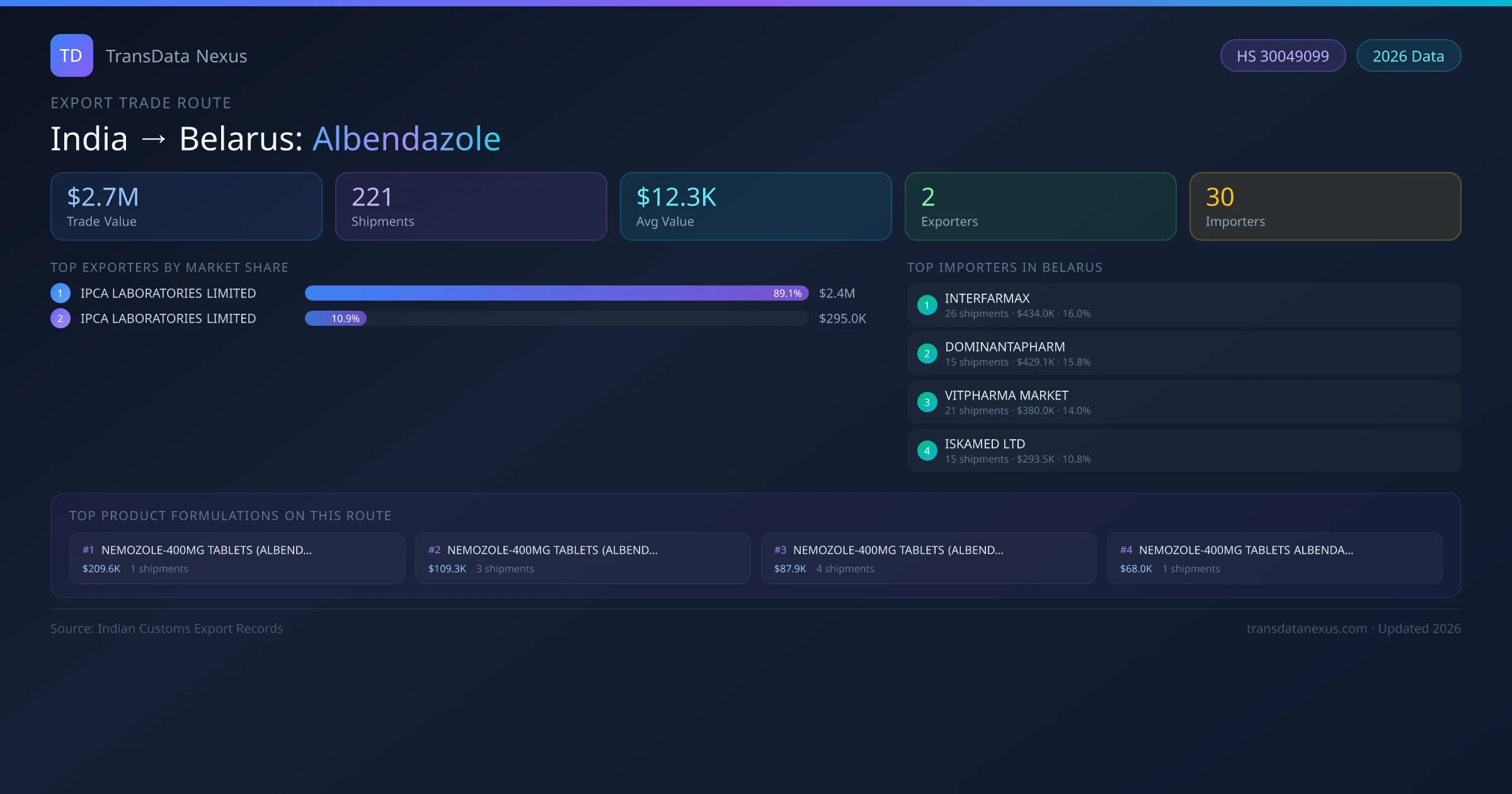
India to Belarus: Albendazole Export Trade Route
India has recorded 221 verified shipments of Albendazole exported to Belarus, representing a combined trade value of $2.7M USD. This corridor is served by 2 active Indian exporters, with an average shipment value of $12.3K USD. The leading Indian exporter is IPCA LABORATORIES LIMITED, which accounts for 89% of total export value with 195 shipments worth $2.4M USD. On the buying side, INTERFARMAX is the largest importer in Belarus with $434.0K USD in purchases. All data sourced from Indian Customs (DGFT) shipping bill records. Values reported in FOB USD.
Route Intelligence Overview
The India to Belarus Albendazole corridor is one of India's established pharmaceutical export routes, with 221 shipments documented worth a combined $2.7M USD. The route is dominated by IPCA LABORATORIES LIMITED, which alone accounts for roughly 89% of all export value, reflecting the consolidated nature of India's albendazole manufacturing sector.
Across 2 active suppliers, the average shipment value stands at $12.3K USD — a figure that reflects both bulk commercial orders from large pharmaceutical companies and smaller specialty shipments. Sea freight dominates at 74% of all shipments, consistent with albendazole's non-urgent bulk-order profile.
Shipment activity peaks during UNAVAILABLE, with an average transit time of 34 days port-to-port. The route has recorded an annual growth rate of 55.6%, placing it at rank #6 among India's top albendazole export destinations globally.
On the import side, key buyers of Indian albendazole in Belarus include INTERFARMAX, DOMINANTAPHARM, VITPHARMA MARKET and 27 others. INTERFARMAX is the single largest importer with 26 shipments valued at $434.0K USD.
Route Characteristics
- Average transit34 days
- Peak seasonUNAVAILABLE
- Primary modeSea freight
- Top portSAHAR AIR CARGO ACC (INBOM4)
Market Position
- Global rank#6
- Annual growth+55.6%
- Demand growth+22.8%
- Regulatory ease76/100
Top 10 Indian Albendazole Exporters to Belarus
Showing top 10 of 2 Indian suppliers exporting Albendazole to Belarus, ranked by total trade value (USD)
| Rank | Supplier (Indian Exporter) | Shipments | Total Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | IPCA LABORATORIES LIMITED Avg $12.4K per shipment | 195 | $2.4M | 89.1% |
| 2 | IPCA LABORATORIES LIMITED Avg $11.3K per shipment | 26 | $295.0K | 10.9% |
This table shows the top 10 of 2 Indian companies exporting albendazole to Belarus, ranked by total trade value. The listed exporters are: IPCA LABORATORIES LIMITED, IPCA LABORATORIES LIMITED . IPCA LABORATORIES LIMITED is the dominant supplier with 195 shipments worth $2.4M USD, giving it a 89% market share.
Top 10 Albendazole Importers in Belarus
Showing top 10 of 30 known buyers in Belarus receiving Albendazole shipments from India, ranked by import value
On the receiving end of this trade route, the leading importers of Indian albendazole in Belarus include INTERFARMAX, DOMINANTAPHARM, VITPHARMA MARKET, ISKAMED LTD, BELPHARMACIA, among 30 total buyers. The largest importer is INTERFARMAX, accounting for $434.0K USD across 26 shipments — representing 16% of all albendazole imports from India on this route.
| Rank | Importer / Buyer | Shipments | Import Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | INTERFARMAX | 26 | $434.0K | 16.0% |
| 2 | DOMINANTAPHARM | 15 | $429.1K | 15.8% |
| 3 | VITPHARMA MARKET | 21 | $380.0K | 14.0% |
| 4 | ISKAMED LTD | 15 | $293.5K | 10.8% |
| 5 | BELPHARMACIA | 15 | $162.3K | 6.0% |
| 6 | MINSKAYA PHARMACIA | 18 | $109.8K | 4.1% |
| 7 | BREST RUE PHARMACIA | 15 | $100.6K | 3.7% |
| 8 | MOGILEV PHARMACY | 15 | $74.2K | 2.7% |
| 9 | VITPHARMA MARKET | 2 | $70.2K | 2.6% |
| 10 | DOMINANTAPHARM 15 STR DOMBROVSKAYA | 1 | $68.0K | 2.5% |
Showing top 10 of 30 Albendazole importers in Belarus on this route.
Top 10 Albendazole Formulations Imported by Belarus
Showing top 10 of 156 product formulations shipped on the India to Belarus Albendazole route, ranked by trade value
Belarus imports a wide range of albendazole formulations from India, spanning tablets, capsules, suspensions, and combination drugs. The top formulation — NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (7040 X 1 X 5'S= 35200 TBS) — accounts for $209.6K USD across 1 shipments. There are 156 distinct product descriptions in the dataset, reflecting the variety of dosage forms and strengths imported.
| Rank | Product Formulation | Shipments | Trade Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (7040 X 1 X 5'S= 35200 TBS) | 1 | $209.6K | 7.7% |
| 2 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (6400 X 1 X 5'S= 32000 TBS) | 3 | $109.3K | 4.0% |
| 3 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (32000 X 1 X 1'S= 32000 TBS) | 4 | $87.9K | 3.2% |
| 4 | NEMOZOLE-400MG TABLETS ALBENDAZOLE 11979 X 1 X 5'S= 59895 TBS | 1 | $68.0K | 2.5% |
| 5 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (32 | 3 | $67.7K | 2.5% |
| 6 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (2240 X 1 X 5'S= 11200 TBS) | 1 | $67.0K | 2.5% |
| 7 | NEMOZOLE 400MG TABLETS ALBENDAZOLE 47040 X 1 X 1 S 47040 TBS | 1 | $62.1K | 2.3% |
| 8 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (39980 X 1 X 1'S= 39980 TBS)TBS | 3 | $54.8K | 2.0% |
| 9 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (8000 X 1 X 1'S= 8000 TBS) | 5 | $54.2K | 2.0% |
| 10 | NEMOZOLE-400MG TABLETS (ALBENDAZOLE) (4800 X 1 X 5'S= 24000 TBS) | 2 | $53.7K | 2.0% |
Showing top 10 of 156 Albendazole formulations imported by Belarus on this route.
Shipping & Logistics Analysis
Freight mode split and port-of-origin breakdown
Freight Mode Distribution
Sea freight dominates at 74%, typical for bulk pharmaceutical shipments.
Top Ports of Origin
SAHAR AIR CARGO ACC (INBOM4) handles the highest volume with 89 shipments. Transit time averages 34 days by sea.
Market Dynamics
India's albendazole exports to Belarus are driven primarily by a handful of large-scale manufacturers. IPCA LABORATORIES LIMITED with 195 shipments leads the pack, a pattern common in generic pharmaceutical corridors where manufacturing scale creates significant cost advantages. The presence of 2 active exporters signals a competitive but concentrated market — buyers in Belarus benefit from supplier diversity while the top tier handles the majority of volume.
The top 3 suppliers — IPCA LABORATORIES LIMITED, IPCA LABORATORIES LIMITED — together account for 100% of total trade value on this route. The average shipment value of $12.3K USD reflects a mix of bulk commercial orders and smaller specialty shipments.
Beyond the primary product category, shipments on this route include closely related formulations such as nemozole-400mg tablets (albendazole) (6400 x 1 x 5's= 32000 tbs) and nemozole-400mg tablets (albendazole) (32000 x 1 x 1's= 32000 tbs), suggesting that buyers in Belarus tend to consolidate orders across related product lines from the same Indian supplier.
On the buying side, INTERFARMAX is the largest importer with 26 shipments worth $434.0K USD — representing 16% of all albendazole imports from India on this route. A total of 30 buyers are active on this corridor.
Route Statistics
- Trade Volume
- $2.7M
- Avg. Shipment
- $12.3K
- Suppliers
- 2
- Buyers
- 30
- Transit (Sea)
- ~34 days
- Annual Growth
- +55.6%
Related Analysis
Other Albendazole Routes
Unlock the Full India to Belarus Albendazole Dataset
TransData Nexus provides verified shipment-level records, supplier contact details, HS code breakdowns, real-time pricing benchmarks, and regulatory compliance guides for 221 shipments on this route.
Live Corridor Intelligence
India → Belarus trade corridor intelligence
1Live Corridor Intelligence
As of March 2026, the India-Belarus pharmaceutical trade corridor, particularly for finished formulations containing Albendazole, is experiencing notable logistical challenges. The ongoing maritime disruptions in the Red Sea and Suez Canal, stemming from regional conflicts and security concerns, have led to increased transit times and elevated freight costs. Shipping routes are being rerouted around the Cape of Good Hope, adding approximately 10 to 12 days to delivery schedules. This detour has resulted in a surge in freight rates, with costs doubling compared to early 2025 levels. Additionally, currency fluctuations, including the depreciation of the Indian Rupee against the US Dollar, have further impacted trade margins. No significant trade policy changes between India and Belarus have been reported in this period.
Geopolitical & Sanctions Impact
India → Belarus trade corridor intelligence
1Geopolitical & Sanctions Impact
The India-Belarus pharmaceutical trade corridor is indirectly affected by broader geopolitical tensions. The 2025 United States–India diplomatic and trade crisis, initiated in August 2025, led to the imposition of a 50% tariff on certain Indian exports by the U.S. While this tariff does not directly target pharmaceutical exports to Belarus, it has contributed to a more volatile global trade environment. Additionally, the Israel-Iran conflict in June 2025 caused significant disruptions in the Strait of Hormuz, leading to increased war-risk insurance premiums and higher freight rates. These factors have collectively influenced shipping routes and costs between India and Belarus.
Trade Agreement & Policy Analysis
India → Belarus trade corridor intelligence
1Trade Agreement & Policy Analysis
As of early 2026, there is no specific Free Trade Agreement (FTA) between India and Belarus. Trade between the two nations operates under the framework of the World Trade Organization (WTO) rules, which both countries adhere to. Recent bilateral engagements have focused on enhancing trade facilitation measures, particularly in the pharmaceutical sector. However, no significant policy changes or new agreements have been finalized in the past year.
Landed Cost Breakdown
India → Belarus trade corridor intelligence
1Landed Cost Breakdown
Estimating the landed cost for Albendazole formulations shipped from India to Belarus involves several components:
- FOB Price: The Free on Board (FOB) price for Albendazole formulations is approximately $10 per unit.
- Sea Freight Cost: Due to current disruptions, sea freight costs have increased to around $4,000 per 20-foot container. Assuming 10,000 units per container, this translates to $0.40 per unit.
- Insurance: Insurance premiums have risen, adding approximately $0.10 per unit.
- Customs Duty: Belarus imposes a customs duty of 5% on pharmaceutical imports, equating to $0.50 per unit.
- Clearance Charges: Customs clearance and handling charges amount to $0.20 per unit.
- VAT/GST: Belarus applies a Value Added Tax (VAT) of 20%, adding $2.00 per unit.
- Local Distribution: Local distribution and logistics costs are estimated at $0.30 per unit.
Summing these components, the total landed cost per unit is approximately $13.50. For a 20-foot container carrying 10,000 units, the total landed cost would be $135,000.
Belarus Pharmaceutical Import Regulations
National DRA registration, GMP, and compliance requirements for Indian exporters
1National DRA Registration & Import Requirements
To import Albendazole formulations into Belarus, the following approvals and registrations are necessary:
- Registration Dossier Format: The registration dossier must be submitted in the Common Technical Document (CTD) format.
- Application Submission: Applicants are required to submit a comprehensive dossier that includes administrative information, quality data, non-clinical study reports, and clinical study reports.
- Approval Timelines: The standard timeline for the registration process is approximately 160 working days, excluding the periods for preclinical and clinical trials, remedial actions, and potential GMP inspections.
- Product Registration Fees: While specific fees are not publicly detailed, they are generally structured based on the type of registration procedure (e.g., general or simplified) and the nature of the medicinal product.
- GMP Inspection Requirements: For manufacturing facilities located in countries that are members of the Pharmaceutical Inspection Co-operation Scheme (PIC/S), such as India, GMP inspections may not be mandatory. However, the National DRA reserves the right to conduct inspections if deemed necessary.
2Quality & GMP Standards for Indian Exporters
Indian manufacturers exporting Albendazole formulations to Belarus must comply with the following quality and GMP standards:
- GMP Certification: Manufacturers must possess a valid GMP certificate issued by the regulatory authority of the manufacturing country. Belarus recognizes GMP certificates from PIC/S member countries, including India.
- Approved Indian Facilities: Specific details regarding Indian manufacturing facilities currently approved by the National DRA are not publicly disclosed. However, facilities holding valid GMP certification and adhering to international quality standards are generally considered compliant.
- Recent Inspections and Regulatory Actions: There have been no publicly reported inspections or regulatory actions by the National DRA against Indian pharmaceutical companies exporting to Belarus in the past 18 months.
3Recent Regulatory Developments (2024-2026)
Several regulatory changes and developments have occurred in the past 18 months affecting pharmaceutical exports to Belarus:
- EAEU Compliance Deadline: All medicinal products registered under national procedures in Eurasian Economic Union (EAEU) member states, including Belarus, must be brought into compliance with EAEU requirements by the end of 2025. This harmonization aims to standardize registration dossiers across member states.
- Simplified Registration Procedures: Belarus has implemented simplified registration procedures for medicinal products already registered in the EU, USA, Japan, or pre-qualified by WHO. This allows for a more expedited registration process for such products.
- GMP Inspection Waivers: For strategically important medicinal products, Belarus may waive the requirement for GMP inspections, accepting GMP certificates issued by the manufacturing country's regulatory authority. This facilitates faster market entry for essential medicines. (gosgmp.ru)
These developments reflect Belarus's commitment to aligning with international standards and facilitating the importation of high-quality pharmaceutical products.
Belarus Albendazole Market Context & Tariffs
Market size, import duties, and competitive landscape · MFN tariff: The Most-Favored-Nation (MFN) import duty rate for HS code 30049099 in Belarus is 0%.
1Belarus Albendazole Market Size & Demand
In 2024, the global Albendazole market was valued at approximately USD 11.67 billion, with projections indicating growth to USD 12.81 billion by 2025. While specific data for Belarus is limited, the country's demand for Albendazole is influenced by several factors:
- Disease Prevalence: Belarus experiences cases of parasitic infections such as ascariasis and trichuriasis, necessitating the use of Albendazole formulations.
- Healthcare Spending: The Belarusian government allocates resources to public health initiatives, including the treatment of parasitic diseases, contributing to the demand for Albendazole.
- Aging Population: An aging demographic may lead to increased susceptibility to certain infections, potentially elevating the need for effective anthelmintic treatments.
- Universal Health Coverage: Belarus's commitment to providing comprehensive healthcare services ensures access to essential medications like Albendazole.
Regarding supply, Belarus imports finished Albendazole formulations primarily from India, with a trade value of $2.7 million USD across 221 shipments. Domestic production of Albendazole formulations is minimal, making imports crucial to meet national demand.
2Import Tariff & Duty Structure
Belarus maintains a 0% MFN import duty rate for pharmaceutical products under HS code 30049099, including Albendazole formulations. Additionally, these products are exempt from Value Added Tax (VAT) and other import-related charges, facilitating cost-effective access to essential medications.
There is no Free Trade Agreement (FTA) between India and Belarus specifically affecting pharmaceutical tariffs. Furthermore, no anti-dumping duties are imposed on Albendazole imports from India, ensuring a stable trade environment for these products.
3Competitive Landscape
India is the predominant supplier of Albendazole formulations to Belarus, accounting for 100% of the $2.7 million USD import value. Other major exporting countries include China and various European Union (EU) member states.
India's share of Belarus's total Albendazole imports stands at 100%, underscoring its dominance in this market segment.
In terms of pricing, Indian Albendazole formulations are competitively priced compared to those from China and EU manufacturers. This cost advantage, coupled with established trade relations and consistent supply chains, positions India as a preferred source for Belarusian importers.
Why Source Albendazole from India for Belarus?
Manufacturing advantage, cost comparison, supply reliability, and strategic sourcing recommendations
1Why India for Albendazole — Manufacturing Advantage
India is a global leader in the production of generic pharmaceuticals, accounting for approximately 20% of the global supply by volume as of 2025. This extensive manufacturing base includes a significant capacity for producing finished dosage forms containing Albendazole, such as tablets and capsules. The country's cost-effective production is attributed to economies of scale, a well-established supply chain, and a skilled workforce. As of December 2024, India had over 1,400 WHO-GMP certified pharmaceutical manufacturing facilities and more than 600 FDA-approved plants, many of which produce Albendazole formulations. This robust infrastructure ensures high-quality production standards and regulatory compliance, making India a preferred source for Albendazole formulations globally.
2India vs. China vs. EU — Cost & Quality Comparison
When comparing Albendazole formulation exports, India offers a competitive advantage over China and the European Union (EU) in terms of cost and quality. Indian manufacturers provide finished dosage forms at a lower price per unit due to efficient production processes and lower labor costs. While China is a major supplier of Active Pharmaceutical Ingredients (APIs), its finished dosage forms have faced quality perception challenges, leading to increased scrutiny. The EU produces high-quality branded generics but at a higher cost, making Indian formulations more attractive to markets like Belarus. Belarusian regulatory authorities have a history of accepting Indian pharmaceutical products, reflecting confidence in their quality and compliance. Indian suppliers have demonstrated a reliable supply track record, further solidifying their position as preferred partners.
3Supply Reliability & Capacity Assessment
The India-Belarus Albendazole formulation supply chain is characterized by reliability and substantial manufacturing capacity. Indian facilities are equipped with advanced packaging and cold chain capabilities to ensure product integrity during transit. There have been no significant supply disruptions reported in recent years, indicating a stable supply chain. Indian manufacturers maintain a strong regulatory compliance record, with numerous facilities holding WHO-GMP and FDA approvals. Companies like Alembic Pharmaceuticals have expanded their production capabilities, receiving USFDA approval for Albendazole Tablets USP, 200 mg, in September 2024. This expansion indicates a commitment to meeting global demand and suggests no current capacity constraints among top Indian formulation manufacturers.
4Strategic Sourcing Recommendations
For Belarusian buyers sourcing Albendazole formulations from India, the following strategic recommendations are advised:
- Implement a Dual-Sourcing Strategy: Engage with multiple Indian manufacturers to mitigate risks associated with supply disruptions and ensure a consistent supply chain.
- Understand Minimum Order Quantities (MOQs): Indian suppliers typically require MOQs ranging from 100,000 to 500,000 tablets per order. Align procurement plans accordingly to meet these requirements.
- Negotiate Favorable Payment Terms: Common payment terms in India-Belarus pharmaceutical trade include Letters of Credit (LC) and advance payments. Establish clear agreements to facilitate smooth transactions.
- Conduct Thorough Supplier Qualification: Evaluate potential suppliers based on regulatory approvals (e.g., WHO-GMP, USFDA), production capacity, quality control measures, and past performance to ensure reliability and compliance.
- Monitor Regulatory Compliance: Stay informed about any changes in regulatory standards in both India and Belarus to ensure continuous compliance and avoid potential trade barriers.
By adhering to these recommendations, Belarusian buyers can establish a robust and efficient sourcing strategy for Albendazole formulations from India.
Supplier Due Diligence Guide — Albendazole from India
Pre-qualification checklist, document requirements, red flags, and audit guidance for Belarus buyers
1Pre-Qualification Checklist for Belarus Buyers
1. Verify Manufacturer's Registration with Belarusian Authorities: Confirm that the Indian manufacturer is registered with the Ministry of Health of the Republic of Belarus, as per the Law of the Republic of Belarus "On Medicinal Products."
2. Assess GMP Certification Validity: Ensure the manufacturer holds a valid Good Manufacturing Practice (GMP) certificate recognized by international regulatory bodies such as the World Health Organization (WHO-GMP) or the U.S. Food and Drug Administration (USFDA).
3. Review Drug Master File (DMF): Obtain and evaluate the manufacturer's DMF to verify detailed information on the manufacturing process, controls, and quality standards of the Albendazole formulations.
4. Evaluate Quality Management Systems: Confirm the manufacturer has an ISO 9001:2015 certified Quality Management System, indicating adherence to international quality standards.
5. Check Regulatory Compliance History: Investigate the manufacturer's history for any regulatory actions, such as FDA warning letters or WHO-GMP suspensions, which may indicate compliance issues.
6. Confirm Product Registration in Belarus: Ensure the specific Albendazole formulations are registered with the Belarusian health authorities, as unregistered medicines cannot be imported or sold in Belarus.
7. Assess Export Experience: Review the manufacturer's track record of exporting Albendazole formulations to global markets, particularly to countries with stringent regulatory requirements.
2Key Documents to Request from Indian Suppliers
1. Certificate of Analysis (CoA): Provides detailed results of laboratory testing for each batch, confirming the product meets specified quality standards.
2. Certificate of Origin (CoO): Certifies the country where the product was manufactured, essential for customs and regulatory purposes.
3. GMP Certificate: Demonstrates that the manufacturing facility complies with Good Manufacturing Practice standards, ensuring consistent product quality.
4. Stability Data: Includes studies conducted under International Council for Harmonisation (ICH) guidelines to demonstrate the product's shelf life and stability under various conditions.
5. Batch Manufacturing Records: Detailed documentation of the manufacturing process for each batch, ensuring traceability and compliance with established procedures.
6. Drug Master File (DMF): Comprehensive document detailing the manufacturing process, controls, and quality standards of the product.
7. Free Sale Certificate: Issued by the Central Drugs Standard Control Organisation (CDSCO), indicating the product is approved for sale in the manufacturer's country.
8. Insurance Certificates: Proof of product liability insurance, providing coverage in case of product-related issues.
3Red Flags & Warning Signs
1. Regulatory Actions: Recent FDA warning letters or WHO-GMP suspensions against the manufacturer may indicate compliance issues.
2. Unusually Low Pricing: Prices significantly below market rates may suggest compromised quality or substandard manufacturing practices.
3. Lack of Stability Data: Inability to provide comprehensive stability data raises concerns about the product's shelf life and efficacy.
4. Limited Export History: No track record of exporting to regulated markets may indicate inexperience with stringent quality and regulatory requirements.
5. Resistance to Audits: Hesitation or refusal to allow facility audits suggests potential compliance or quality issues.
4Factory Audit & Ongoing Monitoring
1. Pre-Audit Desktop Review: Conduct a thorough review of the manufacturer's documentation, including licenses, certifications, and quality management systems.
2. On-Site Inspection Focus Areas:
3. Post-Audit Corrective Actions: Require the manufacturer to address any identified deficiencies within a specified timeframe and provide evidence of corrective actions taken.
4. Annual Re-Qualification Process: Implement a yearly review of the manufacturer's compliance and performance to ensure ongoing adherence to quality standards.
5. Remote Monitoring Options: Utilize virtual audits, regular reporting, and third-party inspections to maintain oversight between on-site audits.
Cost Estimates and Timeline:
By adhering to this comprehensive supplier qualification process, Belarusian companies can ensure the procurement of high-quality Albendazole formulations from reputable Indian manufacturers, safeguarding public health and maintaining regulatory compliance.
Frequently Asked Questions — India to Belarus Albendazole Trade
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which Indian companies export Albendazole to Belarus?
The leading Indian exporters of Albendazole to Belarus are IPCA LABORATORIES LIMITED, IPCA LABORATORIES LIMITED . IPCA LABORATORIES LIMITED holds the largest market share at approximately 89% of total trade value on this route.
Q What is the total value of Albendazole exports from India to Belarus?
India exports Albendazole to Belarus worth approximately $2.7M USD across 221 recorded shipments. The average value per shipment is $12.3K USD.
Q Which ports does India use to ship Albendazole to Belarus?
The most active port of origin is SAHAR AIR CARGO ACC (INBOM4) with 89 shipments. Indian exporters primarily use sea freight for this route, with 74% of shipments going by sea and 22% by air.
Q How long does shipping take from India to Belarus for Albendazole?
The average transit time for Albendazole shipments from India to Belarus is approximately 34 days. Sea freight typically takes longer, while air freight can reduce this significantly for urgent orders. Peak shipping activity is observed during UNAVAILABLE.
Q Is the India to Belarus Albendazole trade route growing?
Yes — this trade corridor has seen an annual growth rate of approximately 55.6% with demand growth tracking at 22.8%. The route is ranked #6 among India's top Albendazole export destinations globally.
Q How many suppliers are active on the India to Belarus Albendazole route?
There are currently 2 active Indian suppliers exporting Albendazole to Belarus. The market is moderately concentrated with IPCA LABORATORIES LIMITED accounting for 89% of total shipment value.
Q Who are the main importers of Albendazole from India in Belarus?
The leading importers of Indian Albendazole in Belarus include INTERFARMAX, DOMINANTAPHARM, VITPHARMA MARKET, ISKAMED LTD, BELPHARMACIA. INTERFARMAX is the largest buyer with 26 shipments worth $434.0K USD.
Official References & Regulatory Resources
- India Trade Statistics (DGFT)
- Invest India — Pharma Sector
- WCO HS Nomenclature
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory and trade status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Route Identification: India to Belarus export trade corridor identified from Indian Customs (DGFT) records for Albendazole.
- 2.Supplier/Buyer Matching: 2 Indian exporters and 30 importers in Belarus matched using name normalization.
- 3.Value Aggregation: Total export value aggregated from 221 verified shipping bill records. Values are FOB in USD.
- 4.Market Share Analysis: Each supplier and buyer contribution calculated as a percentage of total route value. Statistical normalization applied to ensure accurate representation across varying shipment sizes.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
221 Verified Shipments
2 Indian exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists