Who Buys Adaptogen from India — 5 Importers Behind a $6.2K Market
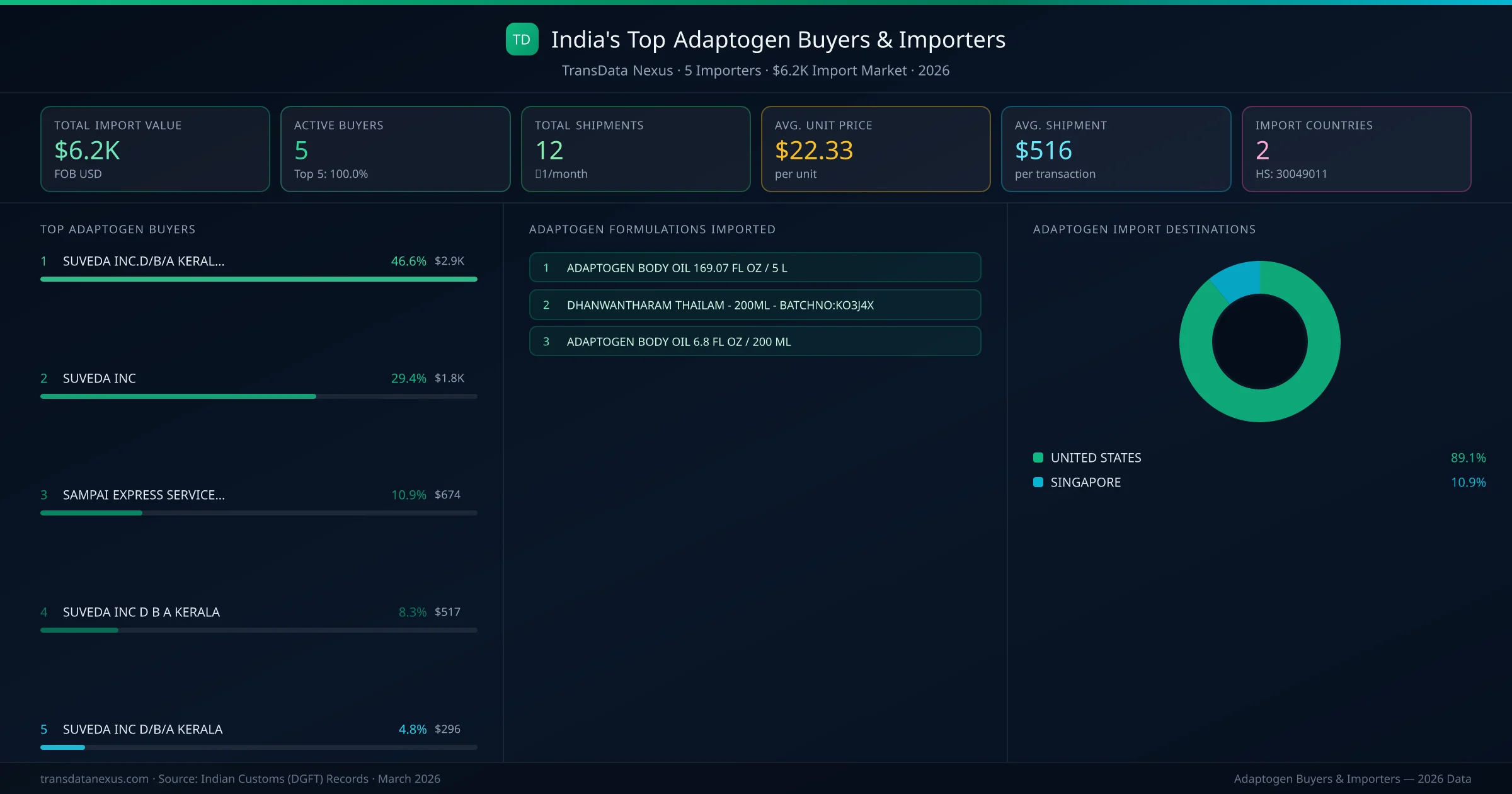
India's adaptogen import market is served by 5 active buyers who collectively imported $6.2K across 12 shipments. SUVEDA INC.D/B/A KERALA AYURVEDA (UNITED STATES) leads with a 46.6% market share, followed by SUVEDA INC. The top 5 buyers together control 76.0% of total import value, reflecting a concentrated buyer landscape.
Top Adaptogen Importers — Ranked by Import Value
SUVEDA INC.D/B/A KERALA AYURVEDA (UNITED STATES) is the leading adaptogen importer from India, holding a 46.6% share of the $6.2K market across 12 shipments from 5 buyers. The top 5 buyers — SUVEDA INC.D/B/A KERALA AYURVEDA (UNITED STATES), SUVEDA INC (UNITED STATES) — collectively control 76.0% of total import value.
Top Adaptogen Buyers & Importers
Ranked by import value · 5 active buyers · Indian Customs (DGFT) data
| # | Buyer & Formulations | Value | Suppliers | Share |
|---|---|---|---|---|
| 1 | SUVEDA INC.D/B/A KERALA AYURVEDAUNITED STATES ADAPTOGEN BODY OIL 169.07 FL OZ / 5 L | $2.9K | 5 | 46.6% |
| 2 | SUVEDA INCUNITED STATES ADAPTOGEN BODY OIL 169.07 FL OZ / 5 L | $1.8K | 5 | 29.4% |
Related Analysis
Market Analysis: Top Importing Countries & Importers
Comprehensive geographic market intelligence dashboard
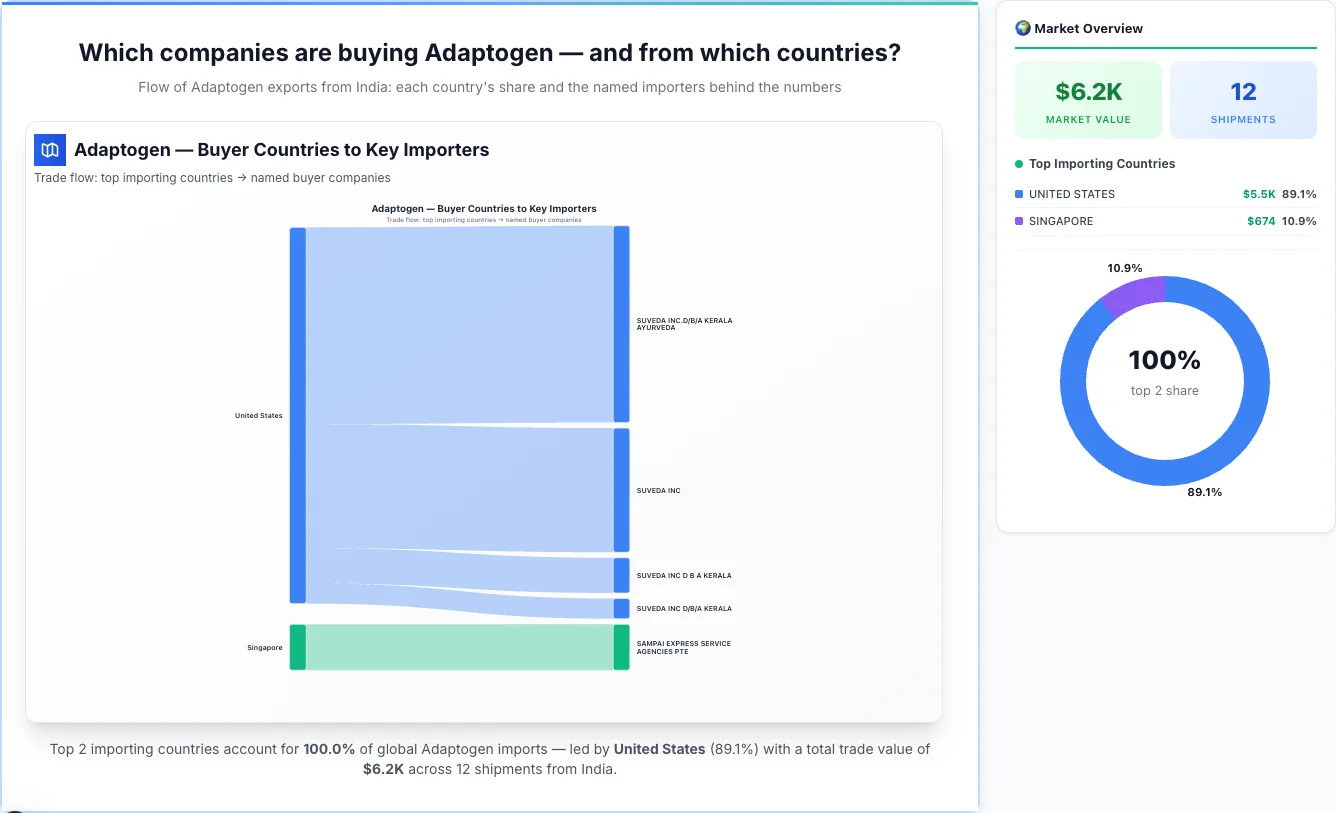
Which companies are buying Adaptogen — and from which countries?
Flow of Adaptogen exports from India: each country's share and the named importers behind the numbers
Adaptogen — Buyer Countries to Key Importers
Trade flow: top importing countries → named buyer companies
Market Analysis: Top Importing Countries & Their Importers
1United States
United States emerges as the first largest importing country, achieving a total trade value of $5.5K through 9 shipments. This represents a market share of 89.1% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: United States has a diverse importer base, with their top importers including:
• SUVEDA INC.D/B/A KERALA AYURVEDA - $2.9K (52.3% of country's total business)
• SUVEDA INC - $1.8K (33.0% of country's total business)
Key Insight: United States demonstrates strong market positioning with an average shipment value of $614, indicating premium pricing strategy compared to the market average of $516.
United States is a key market for Adaptogen imports, representing 89.1% of total trade value.
2Singapore
Singapore emerges as the second largest importing country, achieving a total trade value of $674 through 0 shipments. This represents a market share of 10.9% of the total market activity, demonstrating their significant presence in the industry.
Key Insight: Singapore demonstrates strong market positioning with an average shipment value of $674, indicating premium pricing strategy compared to the market average of $516.
Singapore is a key market for Adaptogen imports, representing 10.9% of total trade value.
Buyer Segment Analysis
TransData Nexus Adaptogen buyer market intelligence
1Buyer Segment Analysis
Based on TransData Nexus data from Indian Customs/DGFT (2022-2026), the buyer landscape for Adaptogen imports from India comprises five buyers across two countries, with a repeat buyer rate of 40.0%. The top buyers include SUVEDA INC.D/B/A KERALA AYURVEDA and SUVEDA INC, both based in the United States.
Distributors: Entities like SUVEDA INC.D/B/A KERALA AYURVEDA and SUVEDA INC likely function as distributors. Distributors typically purchase large quantities of products to supply to various retailers, healthcare providers, or directly to consumers. Their buying behavior often involves regular, bulk orders to maintain a steady supply chain and meet market demand.
Retail Chains: While specific retail chains are not identified in the provided data, such entities would purchase Adaptogen products to stock their shelves for direct consumer sales. Their order patterns are usually consistent, aligning with consumer demand and seasonal trends.
Re-exporters: Given that Singapore accounts for 10.9% of the imports, some buyers may act as re-exporters, importing Adaptogen products to distribute them to other markets. Re-exporters often place orders based on the demands of their target export markets, which can lead to variable order sizes and frequencies.
Country-Specific Import Regulations
TransData Nexus Adaptogen buyer market intelligence
1Country-Specific Import Regulations
United States:
1. Drug Regulatory Authority Name: U.S. Food and Drug Administration (FDA).
2. Import Registration Process for Adaptogen: Adaptogens are often marketed as dietary supplements in the U.S. Importers must ensure that these products comply with the Dietary Supplement Health and Education Act (DSHEA). This includes ensuring product safety, proper labeling, and that no unapproved health claims are made. The FDA monitors dietary supplements post-market and can take action if products are found to be unsafe or misbranded.
3. Estimated Timeline for Registration: As dietary supplements do not require pre-market approval by the FDA, there is no formal registration timeline. However, importers must adhere to FDA regulations, and any violations can lead to enforcement actions.
4. Import Tariff/Duty for HS 30049011: Under the Singapore Free Trade Agreement (SGFTA), which went into effect on January 1, 2004, and provided for complete duty phase-out by January 1, 2013, goods under this agreement are free of merchandise processing fees.
Singapore:
1. Drug Regulatory Authority Name: Health Sciences Authority (HSA).
2. Import Registration Process for Adaptogen: In Singapore, Adaptogen products may be classified as health supplements or traditional medicines. Health supplements are not subject to approvals and licensing by HSA for their importation, manufacture, and sale. However, dealers must ensure that their products are not harmful or unsafe and conform to safety and quality standards. Traditional medicines are also not subject to approvals and licensing by HSA for their importation, manufacture, and sale. Dealers have the obligation to ensure that their products are not harmful or unsafe and that they conform with the necessary safety and quality standards. (hsa.gov.sg)
3. Estimated Timeline for Registration: Since health supplements and traditional medicines are not subject to pre-market approval, there is no formal registration timeline. However, dealers must ensure compliance with HSA guidelines before supplying these products in Singapore.
4. Import Tariff/Duty for HS 30049011: Import duties in Singapore vary based on the product's classification and origin. Specific tariff rates for HS Code 30049011 should be verified with Singapore Customs or a customs broker.
Demand Drivers & Market Opportunity
TransData Nexus Adaptogen buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Adaptogen products in importing countries like the United States and Singapore is influenced by several factors:
1. Health and Wellness Trends: There is a growing consumer interest in natural and herbal products that promote overall well-being and stress management. Adaptogens are marketed for their purported ability to help the body resist stressors, aligning with this trend.
2. Aging Population: Both countries have aging populations seeking preventive health measures and natural remedies to maintain health, potentially increasing the demand for Adaptogen products.
3. Regulatory Environment: In the United States, the FDA has issued warning letters to manufacturers making unapproved health claims about Adaptogen products, indicating a need for compliance with regulatory standards. In Singapore, while health supplements and traditional medicines are not subject to pre-market approval, dealers must ensure their products meet safety and quality standards. (hsa.gov.sg)
Given the presence of five buyers across two countries and a repeat buyer rate of 40.0%, there is a demonstrated market interest in Adaptogen products. However, market opportunities are contingent upon compliance with regulatory requirements and the ability to substantiate product claims with scientific evidence.
Common Questions — Adaptogen Buyers & Importers
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
QWho is the largest adaptogen buyer importing from India?
Based on import volume and value, SUVEDA INC.D/B/A KERALA AYURVEDA (UNITED STATES) leads with $2.9K in imports and a 46.6% market share — the highest of any single adaptogen importer. SUVEDA INC (UNITED STATES) and () are the next largest buyers.
QHow many companies buy adaptogen from India?
There are 5 active adaptogen buyers importing from India, with a combined market of $6.2K across 12 shipments to 2 countries. The top 5 buyers hold 76.0% of total import value, while the remaining 0 buyers handle the other 24.0%.
QWhich countries import the most adaptogen from India?
The top importing countries for adaptogen from India are United States (89.1%), Singapore (10.9%). These markets represent the largest demand centres for Indian pharmaceutical exports of adaptogen, with buyers ranging from government procurement agencies to private pharmaceutical distributors.
QWhat is the average import order value for adaptogen from India?
The average import transaction value for adaptogen from India is $516, with an average unit price of $22.33 per unit. Order sizes vary significantly based on buyer type — government tenders tend to be larger while private distributors place more frequent smaller orders.
Buyer Segment Analysis
TransData Nexus Adaptogen buyer market intelligence
1Buyer Segment Analysis
Based on TransData Nexus data from Indian Customs/DGFT (2022-2026), the buyer landscape for Adaptogen imports from India comprises five buyers across two countries, with a repeat buyer rate of 40.0%. The top buyers include SUVEDA INC.D/B/A KERALA AYURVEDA and SUVEDA INC, both based in the United States.
Distributors: Entities like SUVEDA INC.D/B/A KERALA AYURVEDA and SUVEDA INC likely function as distributors. Distributors typically purchase large quantities of products to supply to various retailers, healthcare providers, or directly to consumers. Their buying behavior often involves regular, bulk orders to maintain a steady supply chain and meet market demand.
Retail Chains: While specific retail chains are not identified in the provided data, such entities would purchase Adaptogen products to stock their shelves for direct consumer sales. Their order patterns are usually consistent, aligning with consumer demand and seasonal trends.
Re-exporters: Given that Singapore accounts for 10.9% of the imports, some buyers may act as re-exporters, importing Adaptogen products to distribute them to other markets. Re-exporters often place orders based on the demands of their target export markets, which can lead to variable order sizes and frequencies.
Country-Specific Import Regulations
TransData Nexus Adaptogen buyer market intelligence
1Country-Specific Import Regulations
United States:
1. Drug Regulatory Authority Name: U.S. Food and Drug Administration (FDA).
2. Import Registration Process for Adaptogen: Adaptogens are often marketed as dietary supplements in the U.S. Importers must ensure that these products comply with the Dietary Supplement Health and Education Act (DSHEA). This includes ensuring product safety, proper labeling, and that no unapproved health claims are made. The FDA monitors dietary supplements post-market and can take action if products are found to be unsafe or misbranded.
3. Estimated Timeline for Registration: As dietary supplements do not require pre-market approval by the FDA, there is no formal registration timeline. However, importers must adhere to FDA regulations, and any violations can lead to enforcement actions.
4. Import Tariff/Duty for HS 30049011: Under the Singapore Free Trade Agreement (SGFTA), which went into effect on January 1, 2004, and provided for complete duty phase-out by January 1, 2013, goods under this agreement are free of merchandise processing fees.
Singapore:
1. Drug Regulatory Authority Name: Health Sciences Authority (HSA).
2. Import Registration Process for Adaptogen: In Singapore, Adaptogen products may be classified as health supplements or traditional medicines. Health supplements are not subject to approvals and licensing by HSA for their importation, manufacture, and sale. However, dealers must ensure that their products are not harmful or unsafe and conform to safety and quality standards. Traditional medicines are also not subject to approvals and licensing by HSA for their importation, manufacture, and sale. Dealers have the obligation to ensure that their products are not harmful or unsafe and that they conform with the necessary safety and quality standards. (hsa.gov.sg)
3. Estimated Timeline for Registration: Since health supplements and traditional medicines are not subject to pre-market approval, there is no formal registration timeline. However, dealers must ensure compliance with HSA guidelines before supplying these products in Singapore.
4. Import Tariff/Duty for HS 30049011: Import duties in Singapore vary based on the product's classification and origin. Specific tariff rates for HS Code 30049011 should be verified with Singapore Customs or a customs broker.
Demand Drivers & Market Opportunity
TransData Nexus Adaptogen buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Adaptogen products in importing countries like the United States and Singapore is influenced by several factors:
1. Health and Wellness Trends: There is a growing consumer interest in natural and herbal products that promote overall well-being and stress management. Adaptogens are marketed for their purported ability to help the body resist stressors, aligning with this trend.
2. Aging Population: Both countries have aging populations seeking preventive health measures and natural remedies to maintain health, potentially increasing the demand for Adaptogen products.
3. Regulatory Environment: In the United States, the FDA has issued warning letters to manufacturers making unapproved health claims about Adaptogen products, indicating a need for compliance with regulatory standards. In Singapore, while health supplements and traditional medicines are not subject to pre-market approval, dealers must ensure their products meet safety and quality standards. (hsa.gov.sg)
Given the presence of five buyers across two countries and a repeat buyer rate of 40.0%, there is a demonstrated market interest in Adaptogen products. However, market opportunities are contingent upon compliance with regulatory requirements and the ability to substantiate product claims with scientific evidence.
Official References & Regulatory Resources
- European Medicines Agency
- WHO Essential Medicines
- Invest India — Pharma Sector
- India Trade Statistics (DGFT)
Verify import regulations and drug registration requirements with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Identification: 5 global importers of Adaptogen identified from consignee fields in DGFT shipping bill records.
- 2.Import Value Analysis: Total import value aggregated from 12 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Geographic Distribution: Buyer imports distributed across 2 destination countries with market share per buyer calculated from capped values.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
12 Verified Shipments
5 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists