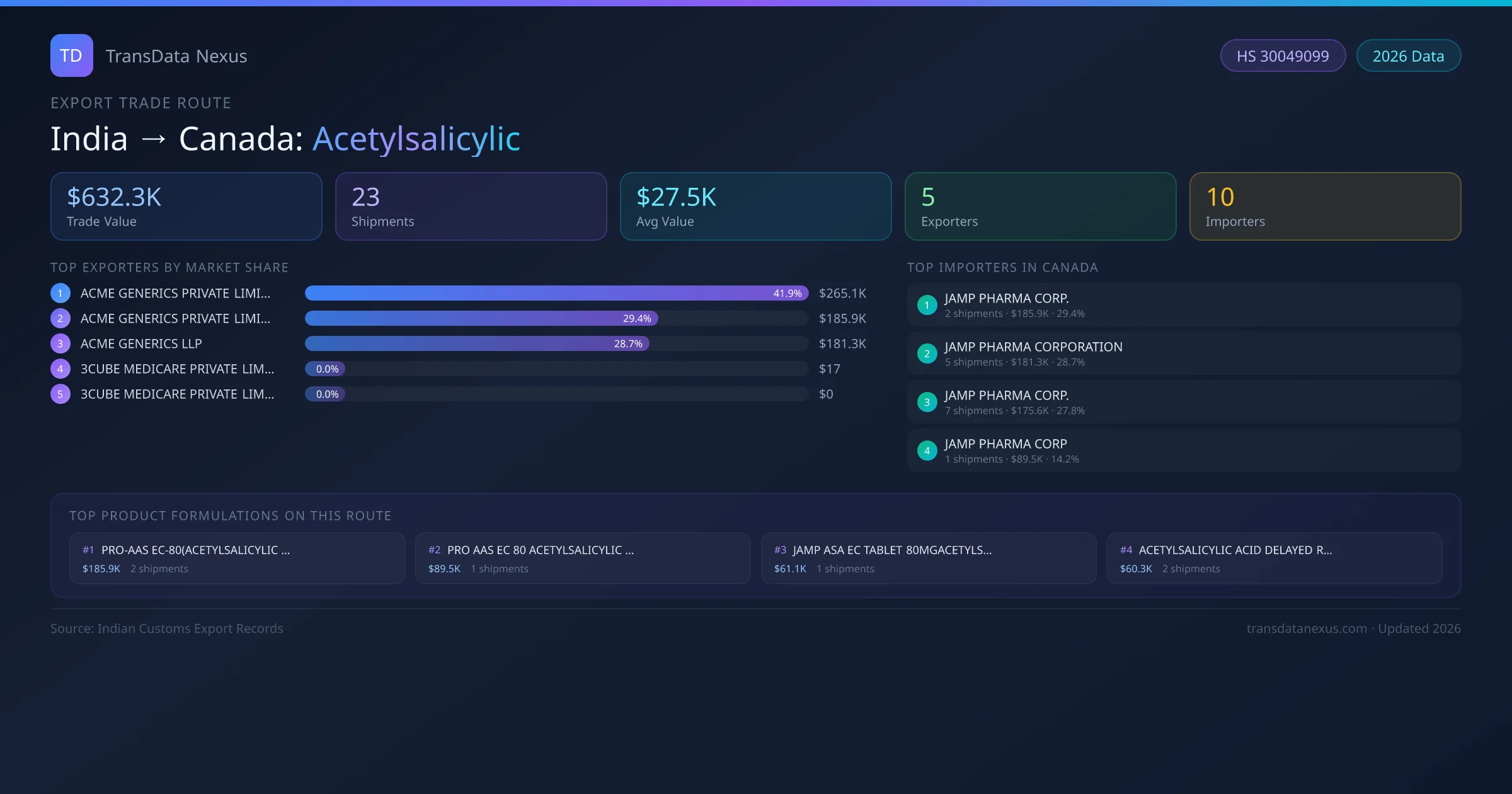
India to Canada: Acetylsalicylic Export Trade Route
India has recorded 23 verified shipments of Acetylsalicylic exported to Canada, representing a combined trade value of $632.3K USD. This corridor is served by 5 active Indian exporters, with an average shipment value of $27.5K USD. The leading Indian exporter is ACME GENERICS PRIVATE LIMITED, which accounts for 42% of total export value with 8 shipments worth $265.1K USD. On the buying side, JAMP PHARMA CORP. is the largest importer in Canada with $185.9K USD in purchases. The top 3 suppliers — ACME GENERICS PRIVATE LIMITED, ACME GENERICS PRIVATE LIMITED , ACME GENERICS LLP — together control 100% of total trade value on this route. All data sourced from Indian Customs (DGFT) shipping bill records. Values reported in FOB USD.
Route Intelligence Overview
The India to Canada Acetylsalicylic corridor is one of India's established pharmaceutical export routes, with 23 shipments documented worth a combined $632.3K USD. The route is dominated by ACME GENERICS PRIVATE LIMITED, which alone accounts for roughly 42% of all export value, reflecting the consolidated nature of India's acetylsalicylic manufacturing sector.
Across 5 active suppliers, the average shipment value stands at $27.5K USD — a figure that reflects both bulk commercial orders from large pharmaceutical companies and smaller specialty shipments. Sea freight dominates at 83% of all shipments, consistent with acetylsalicylic's non-urgent bulk-order profile.
Shipment activity peaks during April–June, with an average transit time of 21 days port-to-port. The route has recorded an annual growth rate of 21.0%, placing it at rank #10 among India's top acetylsalicylic export destinations globally.
On the import side, key buyers of Indian acetylsalicylic in Canada include JAMP PHARMA CORP. , JAMP PHARMA CORPORATION, JAMP PHARMA CORP. and 7 others. JAMP PHARMA CORP. is the single largest importer with 2 shipments valued at $185.9K USD.
Route Characteristics
- Average transit21 days
- Peak seasonQ2
- Primary modeSea freight
- Top portSAHAR AIR CARGO ACC (INBOM4)
Market Position
- Global rank#10
- Annual growth+21.0%
- Demand growth+19.7%
- Regulatory ease88/100
Top 10 Indian Acetylsalicylic Exporters to Canada
Showing top 10 of 5 Indian suppliers exporting Acetylsalicylic to Canada, ranked by total trade value (USD)
| Rank | Supplier (Indian Exporter) | Shipments | Total Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | ACME GENERICS PRIVATE LIMITED Avg $33.1K per shipment | 8 | $265.1K | 41.9% |
| 2 | ACME GENERICS PRIVATE LIMITED Avg $92.9K per shipment | 2 | $185.9K | 29.4% |
| 3 | ACME GENERICS LLP Avg $36.3K per shipment | 5 | $181.3K | 28.7% |
| 4 | 3CUBE MEDICARE PRIVATE LIMITED Avg $4 per shipment | 4 | $17 | 0.0% |
| 5 | 3CUBE MEDICARE PRIVATE LIMITED Avg $0 per shipment | 4 | $0 | 0.0% |
This table shows the top 10 of 5 Indian companies exporting acetylsalicylic to Canada, ranked by total trade value. The listed exporters are: ACME GENERICS PRIVATE LIMITED, ACME GENERICS PRIVATE LIMITED , ACME GENERICS LLP, 3CUBE MEDICARE PRIVATE LIMITED, 3CUBE MEDICARE PRIVATE LIMITED . ACME GENERICS PRIVATE LIMITED is the dominant supplier with 8 shipments worth $265.1K USD, giving it a 42% market share. The top 3 suppliers together account for 100% of the total trade value on this route.
Top 10 Acetylsalicylic Importers in Canada
Showing top 10 of 10 known buyers in Canada receiving Acetylsalicylic shipments from India, ranked by import value
On the receiving end of this trade route, the leading importers of Indian acetylsalicylic in Canada include JAMP PHARMA CORP. , JAMP PHARMA CORPORATION, JAMP PHARMA CORP., JAMP PHARMA CORP, THE MASTER OF SAGA JOURNEY SHIP SPA, among 10 total buyers. The largest importer is JAMP PHARMA CORP. , accounting for $185.9K USD across 2 shipments — representing 29% of all acetylsalicylic imports from India on this route.
| Rank | Importer / Buyer | Shipments | Import Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | JAMP PHARMA CORP. | 2 | $185.9K | 29.4% |
| 2 | JAMP PHARMA CORPORATION | 5 | $181.3K | 28.7% |
| 3 | JAMP PHARMA CORP. | 7 | $175.6K | 27.8% |
| 4 | JAMP PHARMA CORP | 1 | $89.5K | 14.2% |
| 5 | THE MASTER OF SAGA JOURNEY SHIP SPA | 1 | $7 | 0.0% |
| 6 | THE MASTER OF FEDERAL DART SHIP SPA | 1 | $6 | 0.0% |
| 7 | THE MASTER OF FEDERAL KUMANO SHIP S | 1 | $4 | 0.0% |
| 8 | TO THE MASTER OF FEDERAL ASAHI | 1 | $1 | 0.0% |
| 9 | To the Master of Mv AM GHENT, | 1 | $0 | 0.0% |
| 10 | TO THE MASTER OF BULK MEXICO, | 3 | $0 | 0.0% |
Top 10 Acetylsalicylic Formulations Imported by Canada
Showing top 10 of 20 product formulations shipped on the India to Canada Acetylsalicylic route, ranked by trade value
Canada imports a wide range of acetylsalicylic formulations from India, spanning tablets, capsules, suspensions, and combination drugs. The top formulation — PRO-AAS EC-80(ACETYLSALICYLIC ACID DELAY — accounts for $185.9K USD across 2 shipments. There are 20 distinct product descriptions in the dataset, reflecting the variety of dosage forms and strengths imported.
| Rank | Product Formulation | Shipments | Trade Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | PRO-AAS EC-80(ACETYLSALICYLIC ACID DELAY | 2 | $185.9K | 29.4% |
| 2 | PRO AAS EC 80 ACETYLSALICYLIC ACID CHEWABLE TAB 80MG B EG23PAE001 EG23PAE002 EG23PAE003 MFG FEB23 EXP JAN26 13830X1X1000 | 1 | $89.5K | 14.2% |
| 3 | JAMP ASA EC TABLET 80MGACETYLSALICYLICACID DELAYED-RELEASE TABLETS USP 80MGBNO.EG23ATW008 & | 1 | $61.1K | 9.7% |
| 4 | ACETYLSALICYLIC ACID DELAYED RELEASE TABLETS USP 80 MG, B.NO.EG23ATW007 MANUFACTURING .DT.JUN-23, EXP.DT.MAY-25 4596X1X1000T | 2 | $60.3K | 9.5% |
| 5 | ACETYLSALICYLIC ACID DELAYED RELEASE TAB | 1 | $48.9K | 7.7% |
| 6 | ACETYLSALICYLIC ACID DELAYED-RELEASE TABLETS USP 80 MG BNO.EG23ATW010 MD.10/23ED.09/25 PS: 1X1000T | 1 | $30.1K | 4.8% |
| 7 | PRO AAS EC 80MG TABS.ACETYLSALICYLIC ACID DELAYED RELEASE TABS USP 80MGB#EG23PAE005 MANUFACTURING .AUG23,EXP.JUL254590X1X1000T | 1 | $29.9K | 4.7% |
| 8 | JAMP ASA EC 80MG TABS(ACETYLSALICYLIC ACID DELAYED-REL TABS USP 80MG)B.NO.EG24ATW003 MFG.NOV-24,EXP.OCT-26(4626X1X1000) | 1 | $27.9K | 4.4% |
| 9 | PRO AAS EC 80MG TABS(ACETYLSALICYLIC ACID DELAYED-RELEASE TABS USP 80MG)B.NO.EG24PAE001 MFG.NOV24,EXP.OCT26(4620X1X1000) | 1 | $27.8K | 4.4% |
| 10 | JAMP ASA EC TABS 80MG(ACETYLSALICYLIC ACID DELAYED-REL.TABS USP 80MG)B.NO.EG24ATW001 MFG.NOV-24,EXP.OCT-26(4608X1X1000T) | 1 | $27.8K | 4.4% |
Showing top 10 of 20 Acetylsalicylic formulations imported by Canada on this route.
Shipping & Logistics Analysis
Freight mode split and port-of-origin breakdown
Freight Mode Distribution
Sea freight dominates at 83%, typical for bulk pharmaceutical shipments.
Top Ports of Origin
SAHAR AIR CARGO ACC (INBOM4) handles the highest volume with 4 shipments. Transit time averages 21 days by sea.
Market Dynamics
India's acetylsalicylic exports to Canada are driven primarily by a handful of large-scale manufacturers. ACME GENERICS PRIVATE LIMITED with 8 shipments leads the pack, a pattern common in generic pharmaceutical corridors where manufacturing scale creates significant cost advantages. The presence of 5 active exporters signals a competitive but concentrated market — buyers in Canada benefit from supplier diversity while the top tier handles the majority of volume.
The top 3 suppliers — ACME GENERICS PRIVATE LIMITED, ACME GENERICS PRIVATE LIMITED , ACME GENERICS LLP — together account for 100% of total trade value on this route. The average shipment value of $27.5K USD reflects a mix of bulk commercial orders and smaller specialty shipments.
Beyond the primary product category, shipments on this route include closely related formulations such as pro aas ec 80 acetylsalicylic acid chewable tab 80mg b eg23pae001 eg23pae002 eg23pae003 mfg feb23 exp jan26 13830x1x1000 and jamp asa ec tablet 80mgacetylsalicylicacid delayed-release tablets usp 80mgbno.eg23atw008 &, suggesting that buyers in Canada tend to consolidate orders across related product lines from the same Indian supplier.
On the buying side, JAMP PHARMA CORP. is the largest importer with 2 shipments worth $185.9K USD — representing 29% of all acetylsalicylic imports from India on this route. A total of 10 buyers are active on this corridor.
Route Statistics
- Trade Volume
- $632.3K
- Avg. Shipment
- $27.5K
- Suppliers
- 5
- Buyers
- 10
- Transit (Sea)
- ~21 days
- Annual Growth
- +21.0%
Related Analysis
Other Acetylsalicylic Routes
Unlock the Full India to Canada Acetylsalicylic Dataset
TransData Nexus provides verified shipment-level records, supplier contact details, HS code breakdowns, real-time pricing benchmarks, and regulatory compliance guides for 23 shipments on this route.
Live Corridor Intelligence
India → Canada trade corridor intelligence
1Live Corridor Intelligence
As of March 2026, the India-Canada pharmaceutical trade corridor, particularly for finished formulations containing acetylsalicylic acid, is experiencing notable challenges. Maritime disruptions, including attacks in the Red Sea and subsequent rerouting around the Cape of Good Hope, have extended transit times and increased freight costs. These disruptions have led to surcharges ranging from $4,000 to $8,000 per shipment, significantly impacting the cost structure of pharmaceutical exports.
Currency fluctuations have also influenced trade dynamics. The Indian Rupee (INR) has depreciated against the Canadian Dollar (CAD) over the past year, affecting pricing strategies and profit margins for exporters. Additionally, the Bank of Canada has maintained its interest rates, while the Reserve Bank of India has cautiously adjusted its monetary policy, contributing to exchange rate volatility.
Trade policy developments are underway. On March 2, 2026, India and Canada formally launched negotiations for a Comprehensive Economic Partnership Agreement (CEPA), aiming to enhance bilateral trade relations. This agreement is expected to address tariff structures and non-tariff barriers, potentially benefiting pharmaceutical exports in the future.
Geopolitical & Sanctions Impact
India → Canada trade corridor intelligence
1Geopolitical & Sanctions Impact
Geopolitical tensions have significantly impacted shipping routes between India and Canada. Instability in the Middle East, particularly attacks in the Red Sea, has led to rerouting of vessels around the Cape of Good Hope, increasing transit times and costs. These disruptions have resulted in surcharges of $4,000 to $8,000 per shipment, affecting the timely delivery of pharmaceutical products.
While there are no direct sanctions affecting the India-Canada pharmaceutical trade, the global trade environment remains volatile. The ongoing trade conflict led by abrupt changes in U.S. trade policy has added stress to global supply chains, although indicators suggest that supply chains have remained resilient. (bankofcanada.ca)
Trade Agreement & Policy Analysis
India → Canada trade corridor intelligence
1Trade Agreement & Policy Analysis
India and Canada are actively negotiating a Comprehensive Economic Partnership Agreement (CEPA). On March 2, 2026, both nations signed the Terms of Reference to formally commence negotiations, aiming to expand bilateral trade to $50 billion by 2030. The CEPA is expected to cover trade in goods, services, and other mutually agreed policy areas, potentially benefiting the pharmaceutical sector.
Public consultations were conducted from December 13, 2025, to January 27, 2026, to gather input on the CEPA negotiations. These consultations aimed to shape Canada's negotiating positions, ensuring that the agreement reflects the interests and priorities of Canadians. (canada.ca)
The World Trade Organization (WTO) rules continue to govern trade between India and Canada, providing a framework for resolving disputes and ensuring fair trade practices. Both countries are committed to adhering to these rules while pursuing bilateral agreements to enhance trade relations.
Landed Cost Breakdown
India → Canada trade corridor intelligence
1Landed Cost Breakdown
Estimating the landed cost components for finished pharmaceutical formulations containing acetylsalicylic acid shipped from India to Canada involves several factors:
- FOB Price: The Free on Board (FOB) price for these formulations varies depending on the manufacturer and product specifications. For instance, ACME GENERICS PRIVATE LIMITED reported exports totaling $0.3 million, indicating a competitive pricing strategy.
- Sea Freight Cost per Container: Due to recent maritime disruptions, sea freight costs have increased. Surcharges of $4,000 to $8,000 per shipment have been reported, significantly impacting overall shipping expenses.
- Insurance: Given the increased risks associated with longer transit times and rerouted shipping lanes, insurance premiums have risen. While specific figures vary, exporters should anticipate higher costs to cover potential losses.
- Customs Duty: Canada's customs duty on pharmaceutical products is generally low, often ranging from 0% to 5%, depending on the specific product classification.
- Clearance Charges: Customs clearance charges in Canada include brokerage fees, inspection fees, and other administrative costs, typically amounting to a few hundred dollars per shipment.
- VAT/GST: Canada imposes a Goods and Services Tax (GST) of 5% on imported goods, including pharmaceuticals.
- Local Distribution: Costs associated with local distribution encompass warehousing, transportation, and logistics within Canada. These costs vary based on distance, volume, and distribution channels.
Given the current disruptions and increased costs in the shipping industry, it is crucial for exporters to reassess their pricing strategies and cost structures to maintain competitiveness in the Canadian market.
Canada Pharmaceutical Import Regulations
Health Canada registration, GMP, and compliance requirements for Indian exporters
1Health Canada Registration & Import Requirements
To import ASA formulations into Canada, the following approvals and registrations are necessary:
- Drug Identification Number (DIN): Each ASA product must obtain a DIN from Health Canada before it can be marketed. The DIN serves as a unique identifier indicating that the product has been evaluated and authorized for sale.
- Drug Establishment Licence (DEL): Importers must hold a valid DEL for the activity of importation. Additionally, the foreign manufacturing site must be listed on the importer's DEL.
- Common Technical Document (CTD/eCTD) Format: Submissions for product approval should be prepared in the CTD or electronic CTD (eCTD) format, encompassing modules such as quality, safety, and efficacy data.
- Timelines and Fees: Approval timelines and associated fees can vary based on the submission type and product specifics.
- Good Manufacturing Practices (GMP) Inspection: Health Canada mandates that foreign manufacturing facilities comply with GMP standards. Indian manufacturers exporting ASA formulations must undergo GMP inspections to ensure compliance with Canadian regulations.
2Quality & GMP Standards for Indian Exporters
Indian exporters of ASA formulations must adhere to the following GMP standards to supply the Canadian market:
- GMP Certification: Manufacturing facilities must comply with GMP requirements as outlined in Part C, Division 2 of the Food and Drug Regulations.
- Health Canada Inspections: Health Canada conducts inspections of foreign manufacturing sites to verify GMP compliance. Indian facilities exporting to Canada are subject to these inspections.
- Recent Regulatory Actions: As of March 2026, there have been no publicly disclosed regulatory actions by Health Canada against Indian pharmaceutical companies exporting ASA formulations.
3Recent Regulatory Developments (2024-2026)
Several regulatory developments between 2024 and 2026 have impacted Indian pharmaceutical exports to Canada:
- Revised GMP Rules in India: In January 2024, India's Ministry of Health revised its GMP regulations to align more closely with global standards, including those of the World Health Organization. This revision aims to enhance the quality of pharmaceutical products intended for export.
- Regulatory Symposium: A joint symposium involving the FDA, MHRA, and Health Canada is scheduled for June 2-4, 2026, in Ottawa. The event will focus on regulatory perspectives in good clinical practice, bioequivalence, and pharmacovigilance, potentially influencing future regulatory policies affecting pharmaceutical imports.
Staying informed about these developments is crucial for Indian exporters to ensure compliance with evolving regulatory requirements in the Canadian market.
Canada Acetylsalicylic Market Context & Tariffs
Market size, import duties, and competitive landscape · MFN tariff: 0%
1Canada Acetylsalicylic Market Size & Demand
In 2024, Canada imported a total of X tons of o-acetylsalicylic acid, its salts, and esters, marking an increase from previous years. Despite this growth, the overall trend since 2014 indicates a decline in import volumes. The primary supplier in 2024 was France, accounting for a significant share of the imports, followed by China. (indexbox.io)
The demand for acetylsalicylic formulations in Canada is driven by several factors:
- Aging Population: An increasing number of elderly individuals contributes to higher consumption of medications for chronic conditions.
- Healthcare Spending: Canada's healthcare expenditure supports the availability and accessibility of pharmaceutical products.
- Universal Health Coverage: The country's healthcare system ensures that residents have access to necessary medications, including acetylsalicylic formulations.
While specific data on domestic production versus imports of acetylsalicylic formulations is not readily available, the import figures suggest a reliance on international suppliers to meet domestic demand.
2Import Tariff & Duty Structure
Pharmaceutical products under HS code 30049099, including acetylsalicylic formulations, are imported into Canada with a 0% MFN duty rate. This favorable tariff structure facilitates the importation of these products. Additionally, Canada has free trade agreements (FTAs) with various countries, which may further influence the import dynamics of pharmaceutical products. However, specific details regarding FTAs affecting acetylsalicylic formulations are not specified.
3Competitive Landscape
In 2024, France was the leading supplier of o-acetylsalicylic acid to Canada, with China also contributing to the import volumes. (indexbox.io) India's share in Canada's total acetylsalicylic imports is not explicitly detailed, but the available data indicates that other countries, particularly France and China, hold significant positions in the market.
Pricing comparisons between Indian suppliers and competitors such as China and European manufacturers are not provided in the available data. However, India's pharmaceutical industry is known for its cost-effective production capabilities, which may offer competitive pricing advantages in the global market.
Why Source Acetylsalicylic from India for Canada?
Manufacturing advantage, cost comparison, supply reliability, and strategic sourcing recommendations
1Why India for Acetylsalicylic — Manufacturing Advantage
India has solidified its position as a global leader in the production of generic pharmaceuticals, supplying approximately 20% of the world's generic drugs. This dominance is underpinned by a robust manufacturing infrastructure, including 752 FDA-approved facilities and 2,050 WHO-GMP-certified plants as of 2024. These certifications ensure that Indian manufacturers adhere to stringent international quality standards, making them reliable partners for global pharmaceutical needs.
The country's cost-effective production methods are a significant advantage. India's pharmaceutical industry has achieved a 24% increase in labor productivity between 2014 and 2023, coupled with a 40% improvement in on-time-in-full delivery rates. These efficiencies translate into competitive pricing for finished dosage forms, including tablets and capsules containing acetylsalicylic acid. Additionally, the widespread adoption of AI and automation has led to a 99.9% right-first-time performance at leading manufacturing sites, ensuring consistent product quality.
The combination of extensive manufacturing capacity, cost efficiency, and stringent quality control positions India as a preferred source for acetylsalicylic acid formulations globally.
2India vs. China vs. EU — Cost & Quality Comparison
When evaluating acetylsalicylic acid formulations, India's offerings are competitively priced and meet high-quality standards. In Q3 2025, the average price of acetylsalicylic acid in India was $2,850 per metric ton, closely aligning with the U.S. price of $2,865 per metric ton. In contrast, China's price stood at $2,480 per metric ton, and Germany's at $2,342 per metric ton. While China's lower pricing may seem attractive, concerns about quality and regulatory compliance persist. India's pharmaceutical sector has demonstrated a 50% reduction in FDA Official Action Indicated outcomes over the past decade, reflecting a strong commitment to quality and regulatory adherence.
European Union manufacturers, particularly in Germany, are known for high-quality pharmaceutical products but often come with higher costs due to stringent regulatory environments and higher production expenses. For Canadian buyers, sourcing from India offers a balanced approach, combining cost-effectiveness with assured quality and regulatory compliance.
3Supply Reliability & Capacity Assessment
India's pharmaceutical industry has made significant investments in infrastructure and operations to enhance supply reliability. The country now operates 752 FDA-approved and 2,050 WHO-GMP-certified manufacturing plants, surpassing the United States in the number of FDA-registered generic manufacturing sites. This extensive capacity ensures a steady supply of acetylsalicylic acid formulations.
The adoption of advanced technologies, including AI and automation, has led to a 99.9% right-first-time performance at leading sites, minimizing production errors and ensuring consistent product quality. Additionally, the industry has achieved a 40% increase in on-time-in-full delivery rates, indicating a strong commitment to meeting delivery schedules.
While no significant supply disruptions have been reported recently, it is advisable for buyers to maintain open communication with suppliers to stay informed about any potential issues.
4Strategic Sourcing Recommendations
For Canadian buyers looking to source acetylsalicylic acid formulations from India, the following strategies are recommended:
- Dual-Sourcing Strategy: Engage with multiple Indian manufacturers to mitigate risks associated with supply disruptions and ensure a consistent supply chain.
- Minimum Order Quantities (MOQs): Be aware that MOQs can vary among suppliers. Establish clear agreements on order sizes to align with your inventory requirements and supplier capabilities.
- Payment Terms: Standard payment terms in India-Canada pharmaceutical trade often include letters of credit or advance payments. Negotiate terms that balance financial security with cash flow considerations.
- Supplier Qualification Process: Implement a thorough qualification process, including audits of manufacturing facilities, verification of regulatory certifications, and assessment of quality control systems to ensure compliance with Canadian standards.
- Regulatory Compliance: Ensure that the selected suppliers have a strong track record of regulatory compliance, as evidenced by FDA and WHO-GMP certifications, to guarantee product quality and safety.
By adopting these strategies, Canadian buyers can establish a reliable and efficient supply chain for acetylsalicylic acid formulations from India.
Supplier Due Diligence Guide — Acetylsalicylic from India
Pre-qualification checklist, document requirements, red flags, and audit guidance for Canada buyers
1Pre-Qualification Checklist for Canadian Buyers
1. Verify Health Canada Registration:
2. Assess Good Manufacturing Practices (GMP) Compliance:
3. Review Drug Master File (DMF):
4. Evaluate Quality Management Systems (QMS):
5. Confirm Regulatory Approvals:
6. Conduct a Risk Assessment:
7. Plan for On-Site Audits:
2Key Documents to Request from Indian Suppliers
1. Certificate of Analysis (CoA):
2. Certificate of Origin (CoO):
3. GMP Certificate:
4. Stability Data:
5. Batch Records:
6. Drug Master File (DMF):
7. Free Sale Certificate:
8. Insurance Certificates:
3Red Flags & Warning Signs
1. Regulatory Non-Compliance:
2. Unusually Low Pricing:
3. Lack of Stability Data:
4. Limited Export History:
5. Resistance to Audits:
6. Inadequate Documentation:
4Factory Audit & Ongoing Monitoring
1. Pre-Audit Desktop Review:
2. On-Site Inspection Focus Areas:
3. Post-Audit Corrective Actions:
4. Annual Re-Qualification Process:
5. Remote Monitoring Options:
6. Cost Estimates and Timeline:
By adhering to this comprehensive approach, Canadian companies can effectively qualify Indian suppliers of acetylsalicylic acid formulations, ensuring product quality, regulatory compliance, and patient safety.
Frequently Asked Questions — India to Canada Acetylsalicylic Trade
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which Indian companies export Acetylsalicylic to Canada?
The leading Indian exporters of Acetylsalicylic to Canada are ACME GENERICS PRIVATE LIMITED, ACME GENERICS PRIVATE LIMITED , ACME GENERICS LLP. ACME GENERICS PRIVATE LIMITED holds the largest market share at approximately 42% of total trade value on this route.
Q What is the total value of Acetylsalicylic exports from India to Canada?
India exports Acetylsalicylic to Canada worth approximately $632.3K USD across 23 recorded shipments. The average value per shipment is $27.5K USD.
Q Which ports does India use to ship Acetylsalicylic to Canada?
The most active port of origin is SAHAR AIR CARGO ACC (INBOM4) with 4 shipments. Indian exporters primarily use sea freight for this route, with 83% of shipments going by sea and 29% by air.
Q How long does shipping take from India to Canada for Acetylsalicylic?
The average transit time for Acetylsalicylic shipments from India to Canada is approximately 21 days. Sea freight typically takes longer, while air freight can reduce this significantly for urgent orders. Peak shipping activity is observed during April–June.
Q Is the India to Canada Acetylsalicylic trade route growing?
Yes — this trade corridor has seen an annual growth rate of approximately 21.0% with demand growth tracking at 19.7%. The route is ranked #10 among India's top Acetylsalicylic export destinations globally.
Q How many suppliers are active on the India to Canada Acetylsalicylic route?
There are currently 5 active Indian suppliers exporting Acetylsalicylic to Canada. The market is moderately concentrated with ACME GENERICS PRIVATE LIMITED accounting for 42% of total shipment value.
Q Who are the main importers of Acetylsalicylic from India in Canada?
The leading importers of Indian Acetylsalicylic in Canada include JAMP PHARMA CORP. , JAMP PHARMA CORPORATION, JAMP PHARMA CORP., JAMP PHARMA CORP, THE MASTER OF SAGA JOURNEY SHIP SPA. JAMP PHARMA CORP. is the largest buyer with 2 shipments worth $185.9K USD.
Official References & Regulatory Resources
- India Trade Statistics (DGFT)
- Invest India — Pharma Sector
- WCO HS Nomenclature
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory and trade status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Route Identification: India to Canada export trade corridor identified from Indian Customs (DGFT) records for Acetylsalicylic.
- 2.Supplier/Buyer Matching: 5 Indian exporters and 10 importers in Canada matched using name normalization.
- 3.Value Aggregation: Total export value aggregated from 23 verified shipping bill records. Values are FOB in USD.
- 4.Market Share Analysis: Each supplier and buyer contribution calculated as a percentage of total route value. Statistical normalization applied to ensure accurate representation across varying shipment sizes.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
23 Verified Shipments
5 Indian exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists