Loading...
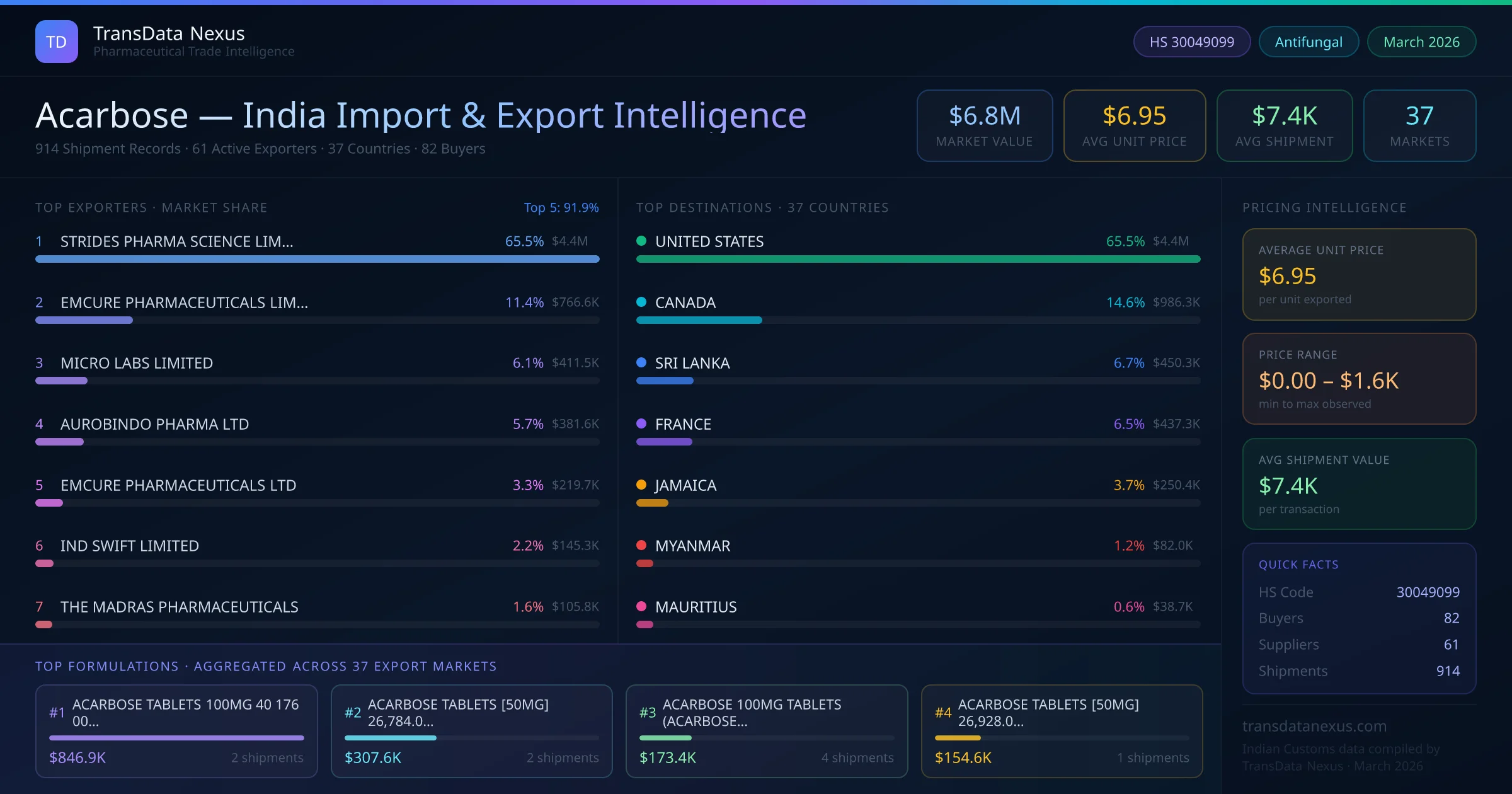
Acarbose
Who Supplies Acarbose to India from GERMANY
India's acarbose imports from GERMANY total $99 across 1 shipments from 1 foreign suppliers. MERCKLE GMBH leads with $99 in import value; the top 5 suppliers together control 100.0% of this origin. Leading Indian buyers include TEVAPHARM INDIA PRIVATE LIMITED. This corridor reflects India's pharmaceutical import demand for acarbose — a concentrated sourcing relationship with select suppliers from GERMANY.
Top foreign suppliers (GERMANY) — Ranked by import value
MERCKLE GMBH is the leading Acarbose supplier from GERMANY to India, with import value of $99 across 1 shipments. The top 5 suppliers — MERCKLE GMBH — collectively account for 100.0% of total import value from this origin.
Top foreign suppliers (GERMANY)
Ranked by import value (USD) · Indian Customs (DGFT) data
| # | Supplier | Value (USD) | Shipments | Share |
|---|---|---|---|---|
| 1 | MERCKLE GMBH | $99 | 1 | 100.0% |
Top Indian buyers (importing from GERMANY)
Ranked by import value (USD)
| # | Buyer | Value (USD) | Shipments | Share |
|---|---|---|---|---|
| 1 | TEVAPHARM INDIA PRIVATE LIMITED | $99 | 1 | 100.0% |
Import Statistics
- Import Value
- $99
- Shipments
- 1
- Foreign Suppliers
- 1
- Indian Buyers
- 1
- Avg. Shipment
- $99
Other Acarbose Import Origins
Live Corridor Intelligence
GERMANY → India trade corridor intelligence
1Live Corridor Intelligence
As of March 2026, the Germany-to-India pharmaceutical import corridor is experiencing significant disruptions due to escalating geopolitical tensions in West Asia, particularly around the Strait of Hormuz. Major shipping lines have suspended or rerouted vessels to avoid the Persian Gulf, leading to extended transit times and increased freight costs. For instance, Europe-bound shipments from India are now taking the longer route around the Cape of Good Hope, adding 10–20 days to transit times and raising freight rates by 40–50% on key India–Europe routes.
These disruptions have led to a doubling of freight charges for both imports and exports, with surcharges ranging from $4,000 to $8,000 per shipment. Additionally, airspace closures in the UAE, Qatar, Kuwait, Israel, Bahrain, and Iraq have restricted major cargo hubs, forcing longer and more expensive alternative routes.
The Indian government has responded by temporarily relaxing import procedures at domestic ports to facilitate the unloading of export shipments that were forced to return due to these disruptions. This includes simplified processes for handling containers that could not reach overseas destinations.
Currency fluctuations have also impacted the corridor. The Indian Rupee (INR) has depreciated against the Euro (EUR) over the past year, increasing the cost of imports from Germany. As of March 2026, the exchange rate stands at approximately 1 EUR to 90 INR, compared to 1 EUR to 85 INR in March 2025.
India's Import Policy & Self-Reliance Push
GERMANY → India trade corridor intelligence
1India's Import Policy & Self-Reliance Push
India's Production Linked Incentive (PLI) scheme, initiated in March 2020, aims to bolster domestic manufacturing of critical Key Starting Materials (KSMs), Drug Intermediates (DIs), and Active Pharmaceutical Ingredients (APIs). While the primary focus is on reducing dependency on imported raw materials, the scheme indirectly influences the import of finished pharmaceutical formulations, including those containing Acarbose. By enhancing domestic production capabilities, India seeks to decrease reliance on imported formulations. The PLI scheme offers financial incentives based on sales of domestically produced critical APIs, thereby encouraging local manufacturing and potentially reducing the need for imported finished formulations.
In July 2025, the Indian government proposed introducing a Minimum Import Price (MIP) on select critical pharmaceutical raw materials to protect domestic manufacturers from low-cost imports, particularly from China. This measure aims to encourage local production and reduce import dependency. (glottis.global)
Bilateral Trade Relations
GERMANY → India trade corridor intelligence
1Bilateral Trade Relations
India and Germany maintain a robust trade relationship, particularly in the pharmaceutical sector. Both countries have engaged in discussions to enhance bilateral trade, including negotiations on mutual recognition of Good Manufacturing Practices (GMP) to facilitate smoother pharmaceutical trade. While no formal Free Trade Agreement (FTA) has been concluded as of March 2026, ongoing dialogues aim to strengthen cooperation and reduce trade barriers in the pharmaceutical industry.
Landed Cost Estimate (Germany to India)
GERMANY → India trade corridor intelligence
1Landed Cost Estimate (Germany to India)
Estimating the landed cost for Acarbose formulations imported from Germany to India involves several components:
- FOB Price from Germany: Assuming a Free on Board (FOB) price of $100 per unit.
- Sea Freight Cost: Due to current disruptions, sea freight costs have increased by approximately 50%. If the previous cost was $10 per unit, it is now $15.
- Marine Insurance: Typically around 0.5% of the CIF (Cost, Insurance, and Freight) value. For a CIF value of $115 ($100 FOB + $15 freight), insurance would be $0.575.
- Basic Customs Duty (BCD): For HS Code 30049099, the BCD is 10% of the CIF value, amounting to $11.50.
- Social Welfare Surcharge (SWS): Calculated at 10% of the BCD, which is $1.15.
- Integrated Goods and Services Tax (IGST): Assuming an IGST rate of 12%, applied to the sum of CIF value, BCD, and SWS: 12% of ($115 + $11.50 + $1.15) = $15.90.
- Port Handling and CHA Charges: Estimated at $5 per unit.
Total Landed Cost per Unit:
- FOB Price: $100.00
- Sea Freight: $15.00
- Marine Insurance: $0.575
- CIF Value: $115.575
- BCD: $11.50
- SWS: $1.15
- IGST: $15.90
- Port Handling and CHA Charges: $5.00
- Total Landed Cost: $149.125
This estimate reflects the increased costs due to current shipping disruptions and currency fluctuations. Actual costs may vary based on specific contractual terms and prevailing market conditions.
India's Import Regulations for Acarbose from GERMANY
CDSCO registration, import licensing, and quality testing requirements
1India Import License & CDSCO Registration
To import Acarbose formulations into India, the following approvals are required:
1. Registration Certificate (Form 41): The foreign manufacturer must obtain a Registration Certificate for the drug and the manufacturing site. This involves submitting Form 40 to the Central Drugs Standard Control Organization (CDSCO), along with detailed information about the manufacturer, product, and compliance with Good Manufacturing Practices (GMP).
2. Import License (Form 10): An Indian entity, such as a pharmaceutical company or distributor, must apply for an Import License by submitting Form 8 to the CDSCO. This application should include the Registration Certificate and other relevant documents.
3. No Objection Certificate (NOC): In certain cases, an NOC from the Drug Controller General of India (DCGI) may be required, especially if the product is a new drug or has specific regulatory considerations.
The timeline for obtaining these approvals can vary, typically ranging from several months to over a year, depending on the completeness of the application and the specific requirements for Acarbose formulations under HS Code 30049099.
2Quality Testing & Batch Certification for Imports
Quality control measures for imported Acarbose formulations include:
1. Mandatory Testing: Each batch must undergo testing at CDSCO-approved laboratories to verify compliance with prescribed standards.
2. Certificate of Analysis (CoA): A CoA from the manufacturer, detailing the batch's quality parameters, must accompany each shipment.
3. Stability Data: Stability studies should be conducted under conditions specific to India's climate (ICH Zone IV) to ensure the product's efficacy and safety throughout its shelf life.
4. Pharmacopoeial Standards: Imported formulations must comply with the Indian Pharmacopoeia standards. If the product adheres to other pharmacopoeias like USP, BP, or EP, equivalence must be demonstrated.
5. Port of Entry Inspection: Upon arrival, customs drug inspectors may conduct inspections to ensure compliance with regulatory requirements before the product is released into the market.
3Recent Import Policy Changes (2024-2026)
Between 2024 and 2026, several policy changes have impacted the import of pharmaceutical formulations into India:
1. Minimum Import Price (MIP): In July 2025, the Indian government introduced an MIP on certain pharmaceutical raw materials to protect the domestic industry from low-cost imports. While this primarily targets raw materials, it may indirectly affect the cost structure of imported finished formulations.
2. Production-Linked Incentive (PLI) Scheme: The PLI scheme, aimed at boosting domestic manufacturing, has led to increased scrutiny of imported finished formulations to encourage local production.
3. Bilateral Agreements: As of March 2026, there have been no specific bilateral agreements between India and Germany affecting the import of pharmaceutical products.
Importers should stay informed about these regulatory changes to ensure compliance and adapt their import strategies accordingly.
India's Acarbose Import Market & Tariffs
Market demand, customs duty structure, and competitive landscape
1India's Acarbose Import Demand
India's importation of finished pharmaceutical formulations containing Acarbose from Germany is minimal, with a recorded import value of $99 USD from a single shipment. This limited import activity suggests that domestic production capacity for Acarbose formulations is sufficient to meet the country's demand. Acarbose is primarily used in the management of type 2 diabetes mellitus, a condition with a significant prevalence in India. The domestic pharmaceutical industry appears capable of producing the necessary Acarbose formulations, reducing the need for imports.
2India's Import Duty & Customs Structure
Under HS Code 30049099, which covers various pharmaceutical products, India's import duty structure is as follows:
- Basic Customs Duty (BCD): 10%
- Social Welfare Surcharge (SWS): 10% of the BCD
- Integrated Goods and Services Tax (IGST): 12%
Calculating the total landed duty:
- 1BCD: 10% of the assessable value
- 2SWS: 10% of BCD (i.e., 1% of the assessable value)
- 3IGST: 12% of the sum of the assessable value, BCD, and SWS
This results in a cumulative duty impact of approximately 23.536% on the assessable value of the imported goods. No specific exemptions or concessional duties under Free Trade Agreements (FTAs) with Germany are currently applicable to this HS code.
3Germany as a Source — Competitive Position
Germany's role as a supplier of Acarbose formulations to India is minimal, with only one recorded shipment valued at $99 USD. This suggests that Germany's competitive advantage in this sector is limited. The Indian pharmaceutical industry, known for its robust manufacturing capabilities and cost-effective production, likely fulfills the majority of domestic demand for Acarbose formulations. Consequently, imports from Germany and other countries remain negligible.
Why India Imports Acarbose from GERMANY
Import rationale, competitive comparison, supply chain risk, and procurement strategy
1Why India Imports Acarbose from Germany
India's importation of finished pharmaceutical formulations containing acarbose from Germany is primarily driven by the need for specialized dosage forms and high-quality standards that may not be widely available domestically. German manufacturers, such as MERCKLE GMBH, are known for producing advanced formulations, including modified-release tablets, which offer improved patient compliance and therapeutic efficacy. These specialized products often require sophisticated manufacturing technologies and stringent quality control measures, ensuring consistent bioavailability and stability. Additionally, certain acarbose formulations may be under patent protection or produced under technology licenses held by German companies, limiting their production to specific manufacturers. Consequently, Indian pharmaceutical companies and healthcare providers import these specialized formulations to meet the therapeutic needs of patients requiring specific acarbose products not readily manufactured within India.
2Germany vs. Other Origins — Comparison
When comparing Germany to other countries as a source of finished acarbose formulations, several factors come into play:
- Quality Perception and Regulatory Compliance: German pharmaceutical products are globally recognized for their high quality and adherence to stringent regulatory standards. The European Medicines Agency (EMA) enforces rigorous guidelines, ensuring that products meet safety and efficacy requirements. This reputation provides confidence to importers regarding product reliability.
- Supply Reliability: German manufacturers have established robust supply chains, ensuring consistent availability of pharmaceutical products. This reliability is crucial for maintaining uninterrupted treatment regimens for patients.
- Price Considerations: While German products may come at a premium compared to generic formulations from countries like China, the cost is often justified by the superior quality, advanced formulations, and regulatory compliance. In contrast, Chinese manufacturers may offer more cost-effective generic versions, but concerns regarding quality control and regulatory adherence can arise.
Germany's unique advantage lies in its ability to provide high-quality, specialized acarbose formulations that meet stringent regulatory standards, offering assurance to healthcare providers and patients alike.
3Supply Chain Risk for Indian Importers
Relying on Germany as a primary source for acarbose formulations presents several supply chain risks for Indian importers:
- Single-Source Dependency: Dependence on a single country or manufacturer increases vulnerability to supply disruptions. Any production issues or regulatory actions affecting the German supplier can lead to shortages.
- Currency Fluctuations: Exchange rate volatility between the Euro and the Indian Rupee can impact procurement costs, affecting pricing strategies and profit margins.
- Regulatory Changes: Modifications in German or European Union pharmaceutical regulations can affect production processes, compliance requirements, and export policies, potentially disrupting supply chains.
- Quality Incidents: Although rare, any quality control issues or product recalls involving German-manufactured acarbose can lead to supply interruptions and necessitate finding alternative sources.
- Shipping Disruptions: Global events, such as pandemics or geopolitical tensions, can disrupt international shipping routes, leading to delays or increased transportation costs.
To date, there have been no significant reported supply shortages of acarbose formulations imported from Germany into India. However, the potential risks underscore the importance of proactive supply chain management strategies.
4Procurement Recommendations for Indian Buyers
To mitigate the aforementioned risks and ensure a stable supply of acarbose formulations, Indian pharmaceutical companies should consider the following strategies:
1. Dual-Sourcing Strategy: Establish relationships with multiple suppliers from different regions to reduce dependency on a single source. This approach enhances supply chain resilience and provides alternatives in case of disruptions.
2. Inventory Planning: Maintain adequate buffer stock to cushion against potential supply delays. Regularly review inventory levels and adjust procurement schedules based on demand forecasts and supplier lead times.
3. Flexible Payment Terms: Negotiate payment terms that accommodate potential currency fluctuations and economic uncertainties. Options such as Letters of Credit (LC), Documents Against Acceptance (DA), or Documents Against Payment (DP) can provide financial flexibility.
4. Minimum Order Quantities (MOQs): Collaborate with suppliers to determine MOQs that balance cost-effectiveness with inventory management needs. Understanding supplier production constraints can aid in planning and negotiations.
5. Supplier Qualification Process: Implement a rigorous supplier evaluation and qualification process, including audits and quality assessments, to ensure compliance with regulatory standards and product specifications. Regular performance reviews can help maintain quality and reliability.
By adopting these strategies, Indian pharmaceutical companies can enhance their procurement processes, mitigate supply chain risks, and ensure a consistent supply of high-quality acarbose formulations to meet patient needs.
Import Due Diligence — Acarbose from GERMANY
Import license checklist, document requirements, quality testing, and compliance
1Import License & Registration Checklist
To legally import Acarbose formulations into India, follow these steps:
1. Obtain a Directorate General of Foreign Trade (DGFT) Import-Export Code (IEC):
2. Apply for Registration Certificate (Form 41):
3. Obtain Drug Import License (Form 10):
4. Secure No Objection Certificate (NOC) from the Drug Controller:
5. Appoint a Licensed Customs Broker:
6. Request Advance Ruling for HS Code Classification:
2Documents Required from Germany Supplier
Before importing Acarbose formulations, ensure the German supplier provides the following documents:
1. Certificate of Analysis (CoA):
2. Certificate of Pharmaceutical Product (CoPP):
3. GMP Certificate from German Regulatory Authority:
4. Drug Master File (DMF):
5. Stability Data (ICH Zone IVa/IVb for India):
6. Free Sale Certificate:
7. Manufacturer Authorization Letter:
3Quality Assurance & Testing at Indian Ports
Upon arrival of Acarbose formulations at an Indian port, the following quality assurance procedures are conducted:
1. Inspection by CDSCO Port Drug Inspector:
2. Mandatory Batch Testing at Government Laboratories:
3. Sample Collection Procedure:
4. Testing Timelines:
5. Actions on Batch Failure:
6. Common Quality Issues with Germany-Origin Pharmaceuticals:
4Ongoing Compliance & Audit Requirements
To maintain compliance after importation, Indian importers should:
1. Annual CDSCO Renewal:
2. Periodic Supplier Audits in Germany:
3. Change Control Notifications:
4. Pharmacovigilance Reporting:
5. Post-Import Quality Monitoring:
6. Maintaining Import Records:
By meticulously following these guidelines, Indian importers can ensure the lawful and efficient importation of Acarbose formulations from Germany, maintaining the highest standards of pharmaceutical quality and compliance.
Frequently Asked Questions — Acarbose Imports from GERMANY
Answers based on Indian Customs (DGFT) import records compiled by TransData Nexus
QWho are the top Acarbose suppliers from GERMANY to India?
The top Acarbose suppliers from GERMANY to India include MERCKLE GMBH. The leading supplier is MERCKLE GMBH with import value of $99 USD across 1 shipments. India imported Acarbose worth $99 USD from GERMANY in total across 1 shipments.
QHow much Acarbose does India import from GERMANY?
India imported Acarbose worth $99 USD from GERMANY across 1 shipments. Data is from Indian Customs (DGFT) records. Values are in USD.
QWho are the main Indian buyers of Acarbose from GERMANY?
The main Indian buyers of Acarbose sourced from GERMANY include TEVAPHARM INDIA PRIVATE LIMITED. The largest buyer is TEVAPHARM INDIA PRIVATE LIMITED with $99 in imports across 1 shipments.
QWhat is the total value of Acarbose imports from GERMANY to India?
The total value of Acarbose imports from GERMANY to India is $99 USD, across 1 shipments and 1 foreign suppliers. Data source: Indian Customs (DGFT).
Official References & Regulatory Resources
- India Trade Statistics (DGFT)
- Invest India — Pharma Sector
- WCO HS Nomenclature
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory and trade status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Route Identification: GERMANY to India pharmaceutical import trade corridor identified from Indian Customs (DGFT) shipping bill records for Acarbose.
- 2.Supplier/Buyer Matching: 1 foreign suppliers from GERMANY and 1 Indian buyers identified using company name normalization across customs records.
- 3.Value Aggregation: Total import value of $99 aggregated from 1 verified shipping bill records. All values reported in FOB USD as recorded by Indian Customs.
- 4.Market Share Analysis: Each supplier and buyer contribution calculated as a percentage of total route value. Statistical normalization applied to ensure accurate representation across varying shipment sizes.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
1 Verified Shipments
1 suppliers, 1 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists