Zuhoor Elham Ltd
Pharmaceutical Importer · Pakistan · Respiratory Focus · $1.1M Total Trade · DGFT Verified
Zuhoor Elham Ltd is a pharmaceutical importer based in Pakistan with a total trade value of $1.1M across 5 products in 4 therapeutic categories. Based on 23 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Zuhoor Elham Ltd sources from 1 verified Indian supplier, with Macleods Pharmaceuticals Limited accounting for 100.0% of imports.
Zuhoor Elham Ltd — Import Portfolio & Supplier Network
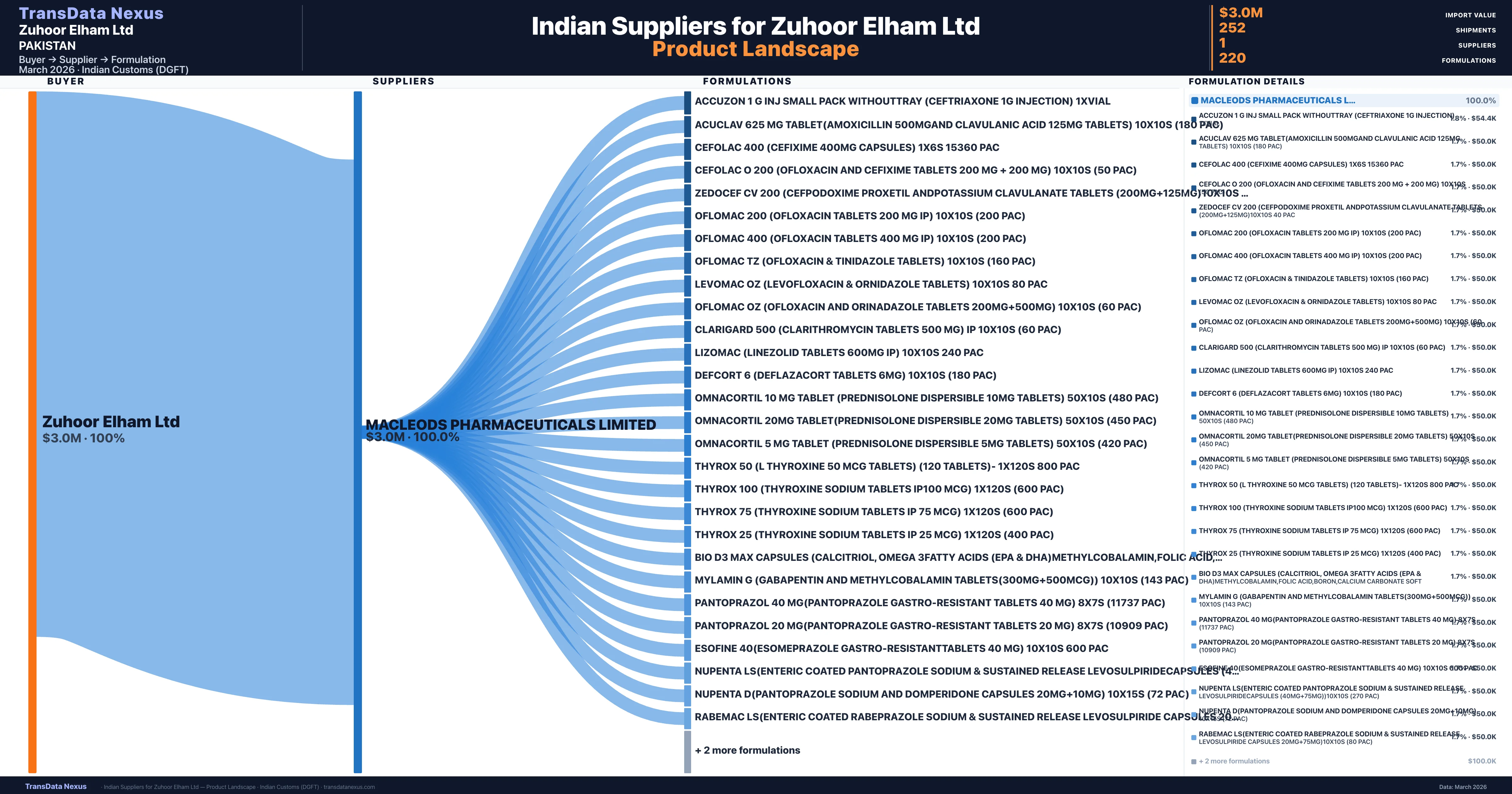
Who Are the Verified Indian Suppliers to Zuhoor Elham Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Macleods Pharmaceuticals Limited | $3.0M | 252 | 100.0% |
Zuhoor Elham Ltd sources from 1 verified Indian supplier across 220 distinct formulations. The sourcing is highly concentrated — Macleods Pharmaceuticals Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Zuhoor Elham Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Accuzon 1 g INJ small Pack withouttray (ceftriaxone 1g injection) | $54.4K | 3 |
| Acuclav 625 MG tablet(amoxicillin 500mgand clavulanic acid 125MG tablets) 10x10s (180 pac) | $50.0K | 1 |
| Cefolac 400 (cefixime 400MG capsules) 1x6s 15360 | $50.0K | 1 |
| Cefolac o 200 (ofloxacin and cefixime tablets 200 MG + 200 MG) 10x10s (50 pac) | $50.0K | 1 |
| Zedocef cv 200 (cefpodoxime proxetil andpotassium clavulanate tablets (200MG+125MG)10x10s 40 | $50.0K | 1 |
| Oflomac 200 (ofloxacin tablets 200 MG IP) 10x10s (200 pac) | $50.0K | 1 |
| Oflomac 400 (ofloxacin tablets 400 MG IP) 10x10s (200 pac) | $50.0K | 1 |
| Oflomac tz (ofloxacin & tinidazole tablets) 10x10s (160 pac) | $50.0K | 1 |
| Levomac oz (levofloxacin & ornidazole tablets) 10x10s 80 | $50.0K | 1 |
| Oflomac oz (ofloxacin and orinadazole tablets 200MG+500MG) 10x10s (60 pac) | $50.0K | 1 |
| Clarigard 500 (clarithromycin tablets 500 MG) IP 10x10s (60 pac) | $50.0K | 1 |
| Lizomac (linezolid tablets 600MG IP) 10x10s 240 | $50.0K | 1 |
| Defcort 6 (deflazacort tablets 6MG) 10x10s (180 pac) | $50.0K | 1 |
| Omnacortil 10 MG tablet (prednisolone dispersible 10MG tablets) 50x10s (480 pac) | $50.0K | 1 |
| Omnacortil 20MG tablet(prednisolone dispersible 20MG tablets) 50x10s (450 pac) | $50.0K | 1 |
Zuhoor Elham Ltd imports 220 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Zuhoor Elham Ltd Import?
Top Products by Import Value
Zuhoor Elham Ltd Therapeutic Categories — 4 Specializations
Zuhoor Elham Ltd imports across 4 therapeutic categories, with Respiratory (40.6%), Advanced Antibiotics (22.5%), Gastrointestinal (18.9%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Respiratory
1 products · 40.6% · $450.0K
Advanced Antibiotics
1 products · 22.5% · $250.0K
Gastrointestinal
1 products · 18.9% · $209.4K
Nutritional Supplements
2 products · 18.0% · $200.0K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Prednisolone | Respiratory | $450.0K | 9 | 0.5% | 4 |
| 2 | Linezolid | Advanced Antibiotics | $250.0K | 5 | 0.7% | 18 |
| 3 | Domperidone | Gastrointestinal | $209.4K | 5 | 0.7% | 18 |
| 4 | Folic | Nutritional Supplements | $100.0K | 2 | 0.2% | 6 |
| 5 | Omega | Nutritional Supplements | $100.0K | 2 | 0.5% | 5 |
Zuhoor Elham Ltd imports 5 pharmaceutical products across 4 categories into Pakistan totaling $1.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Zuhoor Elham Ltd.
Request DemoZuhoor Elham Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Zuhoor Elham Ltd is a pharmaceutical importer based in Pakistan, specializing in the procurement of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company plays a pivotal role in Pakistan's pharmaceutical distribution network, ensuring the availability of essential medications across various therapeutic categories. While specific details about its parent company and exact headquarters location are not publicly disclosed, Zuhoor Elham Ltd's operations are integral to the healthcare supply chain in Pakistan.
2Distribution Network
Zuhoor Elham Ltd's distribution network encompasses multiple warehouse locations strategically situated to facilitate efficient logistics and timely delivery of pharmaceutical products throughout Pakistan. The company's logistics capabilities are robust, enabling it to manage a diverse portfolio of imported medications. Geographically, Zuhoor Elham Ltd's reach extends across Pakistan, ensuring that both urban and rural areas have access to essential pharmaceutical products.
3Industry Role
In Pakistan's pharmaceutical supply chain, Zuhoor Elham Ltd functions primarily as a wholesaler and importer. By sourcing finished pharmaceutical formulations from international suppliers, the company bridges the gap between global manufacturers and the local market, ensuring the availability of a wide range of medications to meet the country's healthcare needs.
Supplier Relationship Intelligence — Zuhoor Elham Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Zuhoor Elham Ltd demonstrates a high degree of supplier concentration, sourcing the majority of its pharmaceutical imports from a single supplier, MACLEODS PHARMACEUTICALS LIMITED. This single-source dependency could pose risks related to supply chain disruptions, such as production delays or quality control issues. However, the consistent volume of shipments and the substantial total import value suggest a stable and well-established relationship between Zuhoor Elham Ltd and MACLEODS PHARMACEUTICALS LIMITED. The company's portfolio concentration, with the top five products accounting for 100% of imports, further indicates a focused sourcing strategy.
2Supply Chain Resilience
The resilience of Zuhoor Elham Ltd's supply chain is closely tied to its reliance on MACLEODS PHARMACEUTICALS LIMITED. The absence of backup suppliers and the limited diversity in imported formulations may expose the company to risks if disruptions occur with this primary supplier. Additionally, the concentration of shipments through specific ports, such as Nhava Sheva, could impact the supply chain's flexibility. Ensuring that MACLEODS PHARMACEUTICALS LIMITED adheres to international regulatory standards and maintains robust quality control measures is crucial for mitigating potential supply chain vulnerabilities.
3Strategic Implications
Zuhoor Elham Ltd's sourcing pattern, characterized by a concentrated supplier base and a focused product portfolio, positions the company to leverage strong relationships with its primary supplier. This strategy may lead to favorable terms and consistent product quality. For Indian exporters, understanding Zuhoor Elham Ltd's specific product requirements and quality standards is essential to explore opportunities for becoming alternative suppliers, thereby diversifying the company's supply chain and reducing potential risks associated with single-source dependency.
Importing Pharmaceuticals into Pakistan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Pakistan
1Regulatory Authority & Framework
The Drug Regulatory Authority of Pakistan (DRAP) is the primary regulatory body overseeing the pharmaceutical sector in Pakistan. Established under the DRAP Act of 2012, DRAP is responsible for ensuring the quality, safety, and efficacy of therapeutic goods, including pharmaceuticals. The Drugs Act of 1976 provides the legal framework for the regulation of drugs in Pakistan, encompassing aspects such as manufacturing, import, export, and distribution. The Import and Export of Therapeutic Goods Rules, 2026, further delineate the procedures and requirements for importing and exporting pharmaceutical products, ensuring compliance with national standards. (dra.gov.pk)
2Import Licensing & GMP
Importers of pharmaceutical products in Pakistan must obtain a Drug Import License (D.I.L.) from DRAP. The application process involves submitting requisite documents through DRAP's online portal, with a typical processing timeline of five days. Additionally, an Import Clearance Certificate is required for each shipment of finished drugs, ensuring that imported products meet the prescribed standards of quality, safety, and efficacy. Compliance with Good Manufacturing Practices (GMP) is mandatory for both domestic and international manufacturers. DRAP recognizes GMP certifications from reputable authorities, including the European Union (EU), World Health Organization (WHO), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S), facilitating the importation of quality pharmaceutical products. (dra.gov.pk)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality and compliance with DRAP's standards. Stability requirements are enforced to ensure that products maintain their efficacy and safety throughout their shelf life. Labeling must be in accordance with the Drugs (Labeling and Packaging) Rules, 1986, which stipulate that all imported packaged medicines display the name and prescription material in the local language. Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
In March 2026, DRAP published the draft Therapeutic Goods (Import and Export) Rules, 2026, for public consultation. These proposed rules aim to streamline the import and export procedures for therapeutic goods, including pharmaceuticals, and are expected to come into force upon finalization. The draft rules emphasize compliance with international standards and aim to enhance the efficiency of regulatory processes. (dra.gov.pk)
Zuhoor Elham Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Zuhoor Elham Ltd's product strategy focuses on therapeutic areas with high demand in Pakistan, including respiratory, advanced antibiotics, and gastrointestinal categories. The company's imports of prednisolone, linezolid, domperidone, folic acid, and omega-3 supplements align with these therapeutic areas, indicating a strategic approach to meet the healthcare needs of the population. The substantial import values of these products reflect their critical importance in the local market.
2Sourcing Profile
Zuhoor Elham Ltd's sourcing strategy is centered on generic pharmaceutical products, primarily sourced from India. The company's preference for finished pharmaceutical formulations, as opposed to raw active pharmaceutical ingredients (APIs), suggests a focus on ready-to-market products that can be swiftly distributed within Pakistan. India's established pharmaceutical manufacturing capabilities and competitive pricing make it a favorable sourcing destination for Zuhoor Elham Ltd.
3Market Positioning
Based on its product mix, Zuhoor Elham Ltd serves a broad segment of the Pakistani pharmaceutical market, including retail pharmacies, hospitals, and wholesale distribution channels. The company's focus on essential medications across various therapeutic categories positions it as a key player in meeting the diverse healthcare needs of the population.
Seller's Guide — How to Become a Supplier to Zuhoor Elham Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Zuhoor Elham Ltd, especially in therapeutic areas where the company seeks to diversify its product offerings. Identifying gaps in the current sourcing strategy, such as the need for additional suppliers or alternative formulations, can provide avenues for new entrants to establish partnerships. However, any new supplier must meet DRAP's regulatory requirements and quality standards to be considered a viable partner.
2Requirements & Qualifications
Indian exporters aiming to supply pharmaceutical products to Zuhoor Elham Ltd and the broader Pakistani market must obtain GMP certifications recognized by DRAP, such as those from the EU, WHO, or PIC/S. Additionally, compliance with DRAP's import licensing requirements, including obtaining a Drug Import License and Import Clearance Certificates, is mandatory. Adherence to labeling and packaging regulations, as outlined in the Drugs (Labeling and Packaging) Rules, 1986, is also essential. (dra.gov.pk)
3How to Approach
To establish a relationship with Zuhoor Elham Ltd, Indian exporters should first ensure compliance with DRAP's regulatory requirements, including obtaining the necessary certifications and licenses. Engaging in direct communication with Zuhoor Elham Ltd to understand their specific product needs and quality expectations is crucial. Participating in relevant tenders and industry events can provide opportunities to showcase products and build rapport. Developing a clear regulatory filing strategy and understanding the expected timelines for approval processes will facilitate smoother collaboration.
Frequently Asked Questions — Zuhoor Elham Ltd
What products does Zuhoor Elham Ltd import from India?
Zuhoor Elham Ltd imports 5 pharmaceutical products across 4 categories. Top imports: Prednisolone ($450.0K), Linezolid ($250.0K), Domperidone ($209.4K), Folic ($100.0K), Omega ($100.0K).
Who supplies pharmaceuticals to Zuhoor Elham Ltd from India?
Zuhoor Elham Ltd sources from 1 verified Indian suppliers. The primary supplier is Macleods Pharmaceuticals Limited (100.0% of imports, $3.0M).
What is Zuhoor Elham Ltd's total pharmaceutical import value?
Zuhoor Elham Ltd's total pharmaceutical import value from India is $1.1M, based on 23 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Zuhoor Elham Ltd focus on?
Zuhoor Elham Ltd imports across 4 categories. The largest: Respiratory (40.6%), Advanced Antibiotics (22.5%), Gastrointestinal (18.9%).
Get Full Zuhoor Elham Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Zuhoor Elham Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Zuhoor Elham Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 23 individual customs records matching Zuhoor Elham Ltd.
- 5.Supplier Verification: Zuhoor Elham Ltd sources from 1 verified Indian suppliers across 220 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.