World Health Organization
Pharmaceutical Importer · Pakistan · Medical Devices & Diagnostics Focus · $4.4M Total Trade · DGFT Verified
World Health Organization is a pharmaceutical importer based in Pakistan with a total trade value of $4.4M across 4 products in 2 therapeutic categories. Based on 200 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. World Health Organization sources from 46 verified Indian suppliers, with Sandoz Private Limited accounting for 16.4% of imports.
World Health Organization — Import Portfolio & Supplier Network
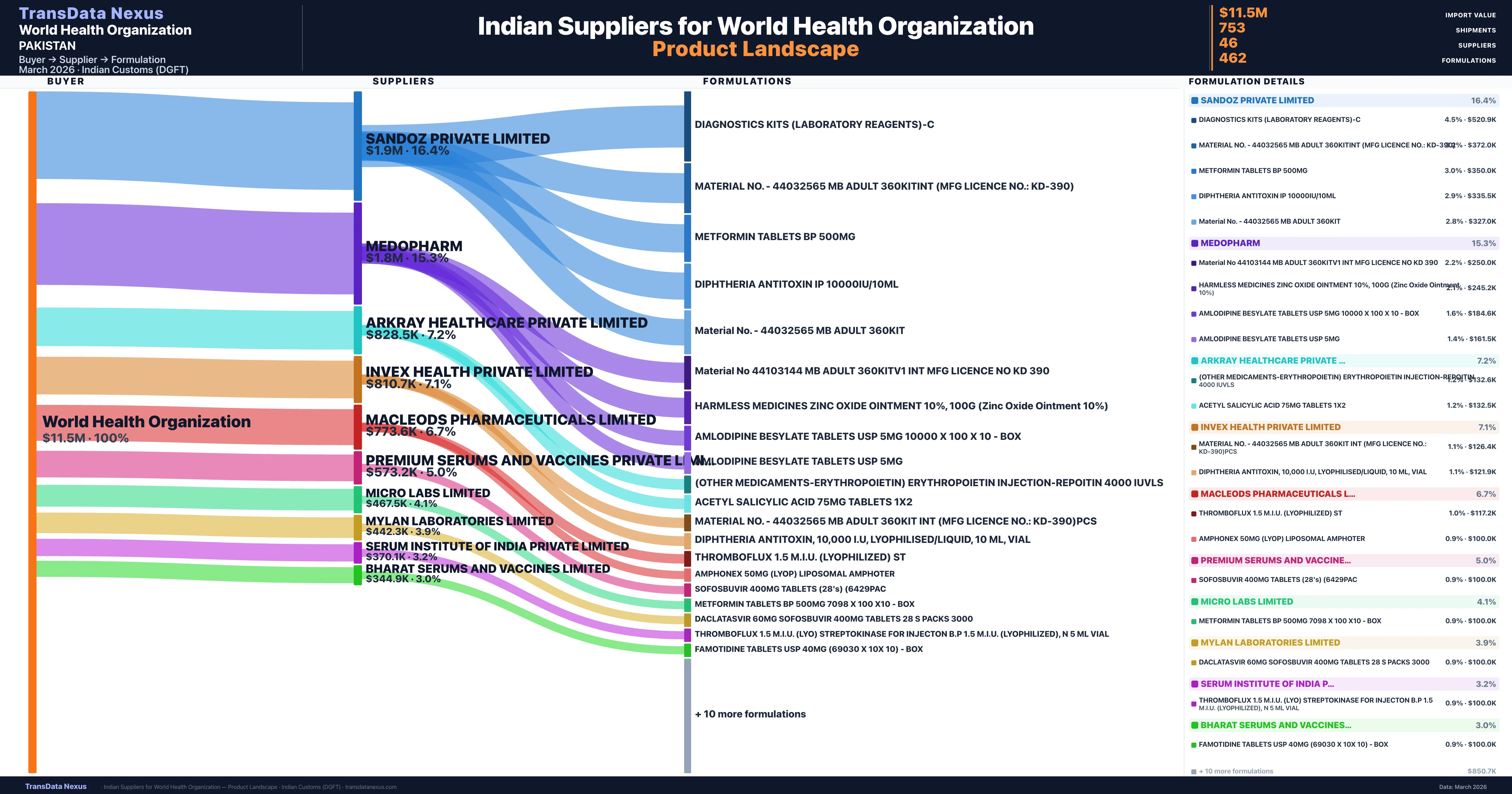
Who Are the Verified Indian Suppliers to World Health Organization?
Customs-verified supplier relationships from Indian DGFT records
World Health Organization sources from 46 verified Indian suppliers across 462 distinct formulations. The supply base is diversified across 46 suppliers, reducing single-source dependency risk.
What Formulations Does World Health Organization Import?
| Formulation | Value | Ships |
|---|---|---|
| Diagnostics kits (laboratory reagents | $520.9K | 26 |
| Material no. - 44032565 mb adult 360kitint (mfg licence no.: kd-390) | $372.0K | 10 |
| Metformin tablets BP 500MG | $350.0K | 7 |
| Diphtheria antitoxin IP 10000iu/10ML | $335.5K | 17 |
| Material no. - 44032565 mb adult | $327.0K | 8 |
| Material no 44103144 mb adult 360kitv1 int mfg licence no kd | $250.0K | 5 |
| Harmless medicines zinc oxide ointment 10%, 100g (zinc oxide ointment 10%) | $245.2K | 5 |
| Amlodipine besylate tablets USP 5MG 10000 x 100 x 10 - | $184.6K | 5 |
| Amlodipine besylate tablets USP 5MG | $161.5K | 5 |
| (other medicaments-erythropoietin) erythropoietin injection-repoitin 4000 | $132.6K | 3 |
| Acetyl salicylic acid 75MG tablets | $132.5K | 6 |
| Material no. - 44032565 mb adult 360kit int (mfg licence no.: kd-390 | $126.4K | 4 |
| Diphtheria antitoxin, 10,000 i.u, lyophilised/liquid, 10 ML | $121.9K | 3 |
| Thromboflux 1.5 m.i.u. (lyophilized) | $117.2K | 5 |
| Amphonex 50MG (lyop) liposomal | $100.0K | 2 |
World Health Organization imports 462 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does World Health Organization Import?
Top Products by Import Value
World Health Organization Therapeutic Categories — 2 Specializations
World Health Organization imports across 2 therapeutic categories, with Medical Devices & Diagnostics (89.8%), Other (10.2%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Medical Devices & Diagnostics
3 products · 89.8% · $4.0M
Other
1 products · 10.2% · $450.0K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Kit | Medical Devices & Diagnostics | $2.6M | 107 | 0.6% | 12 |
| 2 | Diagnostic | Medical Devices & Diagnostics | $691.4K | 42 | 3.8% | 4 |
| 3 | Reagent | Medical Devices & Diagnostics | $691.4K | 42 | 5.3% | 3 |
| 4 | Antitoxin | Other | $450.0K | 9 | 3.4% | 5 |
World Health Organization imports 4 pharmaceutical products across 2 categories into Pakistan totaling $4.4M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for World Health Organization.
Request DemoWorld Health Organization — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. In Pakistan, the WHO operates as a governmental procurement entity, focusing on the acquisition of pharmaceutical products to support public health initiatives. Unlike commercial entities, the WHO's role is not to distribute or retail pharmaceuticals but to procure and supply essential medicines and medical supplies to the Pakistani government and affiliated health organizations. This procurement is integral to Pakistan's healthcare system, ensuring the availability of necessary medical products for public health programs.
2Distribution Network
The WHO's distribution network in Pakistan is extensive, encompassing multiple warehouses strategically located across the country to facilitate efficient storage and distribution of medical supplies. These facilities are equipped with advanced logistics capabilities, including temperature-controlled storage for sensitive pharmaceuticals, to maintain product integrity. The WHO's geographic coverage spans urban and rural areas, ensuring equitable access to medical supplies nationwide. Additionally, the organization coordinates with international suppliers and partners to source and deliver medical products, leveraging global logistics networks to meet Pakistan's healthcare needs.
3Industry Role
Within Pakistan's pharmaceutical supply chain, the WHO functions primarily as a governmental procurement agency. It is not a wholesaler, distributor, or retailer but plays a pivotal role in sourcing and supplying essential medicines and medical supplies to the government and public health institutions. This role is crucial for the implementation of national health programs and the provision of medical care to the population. By procuring pharmaceuticals, the WHO ensures that the government has access to necessary medical products, thereby supporting the overall healthcare infrastructure.
Supplier Relationship Intelligence — World Health Organization
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The WHO's sourcing strategy exhibits a high degree of concentration, with a total import value of $4.4 million USD from India across 200 shipments, encompassing four products in two therapeutic categories. This focused procurement approach suggests a strategic choice to streamline supply chains and maintain consistency in product quality. The top five products imported—Kit, Diagnostic, Reagent, and Antitoxin—account for 100% of the WHO's pharmaceutical imports from India, indicating a specialized and targeted sourcing strategy. This concentration may enhance operational efficiency but also poses risks related to supply chain disruptions.
2Supply Chain Resilience
The WHO's supply chain resilience is closely tied to its sourcing concentration. With a portfolio concentrated entirely on four products from a limited number of suppliers, the organization may face challenges in mitigating supply chain disruptions. The reliance on a select group of suppliers necessitates robust contingency planning and the development of alternative sourcing strategies to ensure continuity of supply. Additionally, the WHO's dependence on Indian suppliers for these critical products underscores the importance of maintaining strong relationships and ensuring compliance with international quality standards to mitigate potential risks.
3Strategic Implications
The WHO's concentrated sourcing pattern positions it to negotiate favorable terms with its suppliers, potentially securing better pricing and consistent product quality. However, this strategy also exposes the organization to risks associated with supply chain disruptions, such as geopolitical tensions or logistical challenges. For Indian exporters, the WHO's focused procurement presents an opportunity to establish long-term partnerships, provided they can meet the stringent quality and regulatory standards required by the WHO. Diversifying the supplier base could further enhance the WHO's supply chain resilience and reduce dependency on a limited number of sources.
Importing Pharmaceuticals into Pakistan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Pakistan
1Regulatory Authority & Framework
In Pakistan, the Drug Regulatory Authority of Pakistan (DRAP) is the primary regulatory body overseeing the pharmaceutical sector. Established under the DRAP Act of 2012, DRAP is responsible for ensuring the quality, safety, and efficacy of therapeutic goods, including pharmaceuticals and medical devices. The regulatory framework governing pharmaceutical imports is primarily based on the Drugs Act of 1976, which outlines the procedures for the import, export, and distribution of drugs. Additionally, the Therapeutic Goods (Import and Export) Rules, 2026, provide detailed guidelines on the import and export procedures for therapeutic goods, including the requirements for obtaining necessary licenses and approvals from DRAP. (dra.gov.pk)
2Import Licensing & GMP
Importers of pharmaceutical products into Pakistan must obtain a Drug Import License (D.I.L.) from DRAP. The application process involves submitting the required documentation through DRAP's online portal, with a typical processing timeline of five days. For the import of finished drugs for commercial purposes, an Import Clearance Certificate is also required, which can be obtained by submitting an application with the necessary documents. Regarding Good Manufacturing Practice (GMP) certification, DRAP recognizes certifications from various international bodies, including the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their suppliers hold valid GMP certifications from recognized authorities to comply with DRAP's standards. (dra.gov.pk)
3Quality & Labeling
Imported pharmaceutical products must adhere to DRAP's quality standards, which include batch testing and stability requirements to ensure product safety and efficacy. Labeling requirements stipulate that all imported packaged medicines display the name and prescription material in accordance with the Drugs (Labeling and Packaging) Rules, 1986. Additionally, labeling must be in the local language, and products must comply with halal certification requirements as specified by the Ministry of National Health Services, Regulations, and Coordination. Serialization mandates may also apply to facilitate traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
In January 2026, Pakistan imposed immediate restrictions on the import of Trichloromethane (chloroform), allowing conditional imports solely for the pharmaceutical sector. This measure requires pharmaceutical companies to obtain a No Objection Certificate (NOC) from DRAP before importing chloroform, aiming to enhance regulatory oversight and prevent misuse. The restriction underscores DRAP's commitment to ensuring that imported chemicals meet national quality standards and are used appropriately within the pharmaceutical industry.
World Health Organization — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
The WHO's product category focus on medical devices and diagnostics, comprising 89.8% of its imports, reflects a strategic emphasis on essential diagnostic tools and medical equipment critical for public health initiatives. The remaining 10.2% of imports fall under the 'Other' category, indicating a diversified approach to sourcing additional medical supplies. This product strategy aligns with the WHO's mission to support comprehensive healthcare services by ensuring the availability of necessary diagnostic and medical equipment.
2Sourcing Profile
The WHO's sourcing strategy is characterized by a focus on finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, imported from India. This approach suggests a preference for ready-to-use products that meet international quality standards, facilitating efficient distribution and utilization within Pakistan's healthcare system. The reliance on Indian suppliers indicates a strategic choice to leverage established manufacturing capabilities and regulatory compliance within India's pharmaceutical industry.
3Market Positioning
Based on its product mix, the WHO serves the public sector of Pakistan's pharmaceutical market, supplying essential medicines and medical supplies to government health programs and institutions. This positioning underscores the WHO's role in supporting public health infrastructure and ensuring the availability of critical medical products for the population.
Seller's Guide — How to Become a Supplier to World Health Organization
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with the WHO, provided they can meet the stringent quality and regulatory standards set by DRAP. Gaps in the WHO's current sourcing may exist in areas such as specialized medical devices or emerging pharmaceutical products not currently covered by existing suppliers. Indian exporters can explore these opportunities by ensuring compliance with DRAP's requirements and demonstrating the quality and efficacy of their products.
2Requirements & Qualifications
Indian exporters seeking to supply the WHO and the Pakistani market must obtain a Drug Import License from DRAP and ensure that their products are registered and approved for import. Suppliers must hold valid GMP certifications from recognized authorities, such as WHO or PIC/S, and comply with DRAP's labeling and packaging requirements, including local language labeling and halal certification. Additionally, products must meet DRAP's quality standards, including batch testing and stability requirements.
3How to Approach
To establish a relationship with the WHO, Indian exporters should initiate contact with DRAP to understand the specific requirements for product registration and import licensing. Participating in tenders and responding to procurement notices issued by the WHO can provide opportunities for collaboration. Developing a comprehensive regulatory filing strategy, including the submission of necessary documentation and certifications, is essential. Timelines for approval can vary; therefore, maintaining open communication with DRAP and the WHO is crucial to navigate the regulatory process effectively.
Frequently Asked Questions — World Health Organization
What products does World Health Organization import from India?
World Health Organization imports 4 pharmaceutical products across 2 categories. Top imports: Kit ($2.6M), Diagnostic ($691.4K), Reagent ($691.4K), Antitoxin ($450.0K).
Who supplies pharmaceuticals to World Health Organization from India?
World Health Organization sources from 46 verified Indian suppliers. The primary supplier is Sandoz Private Limited (16.4% of imports, $1.9M).
What is World Health Organization's total pharmaceutical import value?
World Health Organization's total pharmaceutical import value from India is $4.4M, based on 200 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does World Health Organization focus on?
World Health Organization imports across 2 categories. The largest: Medical Devices & Diagnostics (89.8%), Other (10.2%).
Get Full World Health Organization Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: World Health Organization identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as World Health Organization's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 200 individual customs records matching World Health Organization.
- 5.Supplier Verification: World Health Organization sources from 46 verified Indian suppliers across 462 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.