Waymade PLC
Pharmaceutical Importer · United Kingdom · CNS & Psychiatric Focus · $4.2M Total Trade · DGFT Verified
Waymade PLC is a pharmaceutical importer based in United Kingdom with a total trade value of $4.2M across 2 products in 2 therapeutic categories. Based on 121 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Waymade PLC sources from 10 verified Indian suppliers, with Apothecon Pharmaceuticals Private Limited accounting for 34.0% of imports.
Waymade PLC — Import Portfolio & Supplier Network
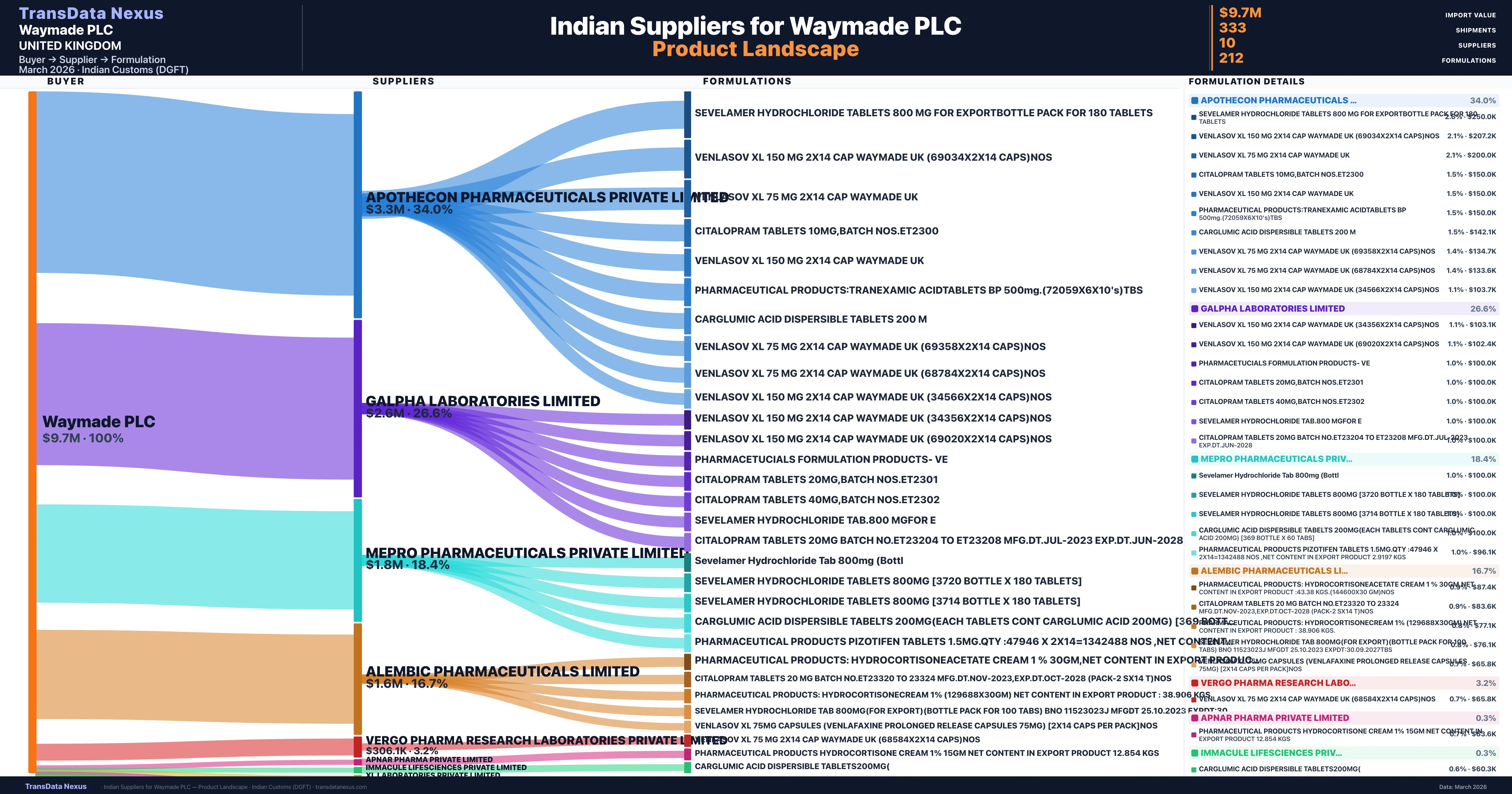
Who Are the Verified Indian Suppliers to Waymade PLC?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Apothecon Pharmaceuticals Private Limited | $3.3M | 83 | 34.0% |
| Galpha Laboratories Limited | $2.6M | 64 | 26.6% |
| Mepro Pharmaceuticals Private Limited | $1.8M | 78 | 18.4% |
| Alembic Pharmaceuticals Limited | $1.6M | 65 | 16.7% |
| Vergo Pharma Research Laboratories Private Limited | $306.1K | 27 | 3.2% |
| Apnar Pharma Private Limited | $32.9K | 10 | 0.3% |
| Immacule Lifesciences Private Limited | $29.3K | 1 | 0.3% |
| Xl Laboratories Private Limited | $28.3K | 1 | 0.3% |
| Usv Private Limited | $22.4K | 1 | 0.2% |
| Sharon Bio-medicine Limited | $135 | 3 | 0.0% |
Waymade PLC sources from 10 verified Indian suppliers across 212 distinct formulations. The supply base is diversified across 10 suppliers, reducing single-source dependency risk.
What Formulations Does Waymade PLC Import?
| Formulation | Value | Ships |
|---|---|---|
| Sevelamer hydrochloride tablets 800 MG for exportbottle Pack for 180 | $250.0K | 5 |
| Venlasov xl 150 MG 2x14 cap waymade uk (69034x2x14 CAPS | $207.2K | 6 |
| Venlasov xl 75 MG 2x14 cap waymade | $200.0K | 4 |
| Citalopram tablets 10MG,batch | $150.0K | 3 |
| Venlasov xl 150 MG 2x14 cap waymade | $150.0K | 3 |
| Pharmaceutical products:tranexamic acidtablets BP 500MG.(72059x6x10's | $150.0K | 3 |
| Carglumic acid dispersible tablets 200 | $142.1K | 4 |
| Venlasov xl 75 MG 2x14 cap waymade uk (69358x2x14 CAPS | $134.7K | 6 |
| Venlasov xl 75 MG 2x14 cap waymade uk (68784x2x14 CAPS | $133.6K | 6 |
| Venlasov xl 150 MG 2x14 cap waymade uk (34566x2x14 CAPS | $103.7K | 6 |
| Venlasov xl 150 MG 2x14 cap waymade uk (34356x2x14 CAPS | $103.1K | 6 |
| Venlasov xl 150 MG 2x14 cap waymade uk (69020x2x14 CAPS | $102.4K | 3 |
| Pharmacetucials formulation products- | $100.0K | 2 |
| Citalopram tablets 20MG,batch | $100.0K | 2 |
| Citalopram tablets 40MG,batch | $100.0K | 2 |
Waymade PLC imports 212 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Waymade PLC Import?
Top Products by Import Value
Waymade PLC Therapeutic Categories — 2 Specializations
Waymade PLC imports across 2 therapeutic categories, with CNS & Psychiatric (75.5%), Corticosteroids (24.5%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
CNS & Psychiatric
1 products · 75.5% · $3.2M
Corticosteroids
1 products · 24.5% · $1.0M
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Citalopram | CNS & Psychiatric | $3.2M | 64 | 1.3% | 12 |
| 2 | Hydrocortisone | Corticosteroids | $1.0M | 57 | 1.6% | 6 |
Waymade PLC imports 2 pharmaceutical products across 2 categories into United Kingdom totaling $4.2M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Waymade PLC.
Request DemoWaymade PLC — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Waymade PLC is a UK-based specialty pharmaceutical company specializing in the development, licensing, and commercialization of generic and niche medicines. Established in 1984 by brothers Vijay and Bhikhu Patel, the company has evolved from a single chemist's shop in Essex into a significant player in the pharmaceutical industry. Headquartered at Monarch House, Miles Gray Road, Basildon, Essex, SS14 3RW, United Kingdom, Waymade PLC operates under the SIC code 46460, which pertains to the wholesale of pharmaceutical goods. (find-and-update.company-information.service.gov.uk)
The company has a history of substantial growth, with a turnover exceeding £300 million at its peak. In 2003, Waymade PLC expanded its operations by establishing Amdipharm, an international pharmaceutical company that acquired rights to sell products in over 100 countries. Amdipharm was acquired by a private equity group in October 2012, with the Patel brothers maintaining some interest in the expanded group. In November 2013, Waymade PLC formed Atnahs Pharma, a company focused on the in-licensing and sale of branded pharmaceuticals in 27 countries.
2Distribution Network
Waymade PLC's distribution network is centered around its headquarters in Basildon, Essex. The company holds a Wholesale Distribution Authorisation (WDA) from the UK's Medicines and Healthcare products Regulatory Agency (MHRA), which permits the wholesale distribution of medicinal products for human use. This authorisation is valid as of January 14, 2026, and is associated with their site at Sovereign House, Miles Gray Road, Basildon, SS14 3FR, United Kingdom. (cms.mhra.gov.uk)
While specific details about additional warehouse locations and logistics capabilities are not publicly disclosed, Waymade PLC's authorisation and operational history suggest a well-established infrastructure capable of supporting its distribution activities across the United Kingdom.
3Industry Role
Waymade PLC plays a multifaceted role in the UK's pharmaceutical supply chain. As a wholesaler, the company supplies a range of pharmaceutical products, including branded and generic medicines, to retail pharmacies, hospitals, and wholesalers. Additionally, Waymade PLC engages in the licensing and commercialization of generic and niche medicines, indicating its involvement in both the wholesale distribution and the in-licensing of pharmaceutical products.
This dual role positions Waymade PLC as a key intermediary in the pharmaceutical supply chain, bridging the gap between manufacturers and end-users within the United Kingdom.
Supplier Relationship Intelligence — Waymade PLC
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Waymade PLC's sourcing strategy exhibits a high degree of concentration, with a total import value from India amounting to $4.2 million USD across 121 shipments. The company's portfolio is heavily concentrated in two therapeutic categories: Central Nervous System (CNS) & Psychiatric disorders, accounting for 75.5% of imports, and Corticosteroids, comprising 24.5%. Within these categories, the top five products imported are Citalopram ($3.2 million, 1.3% share) and Hydrocortisone ($1.0 million, 1.6% share).
The significant reliance on a limited number of products and suppliers suggests a strategic focus on specific therapeutic areas. However, this concentration also introduces potential risks, such as supply chain disruptions or regulatory changes affecting these products. The stability of Waymade PLC's relationships with its Indian suppliers, including APOTHECON PHARMACEUTICALS PRIVATE LIMITED, GALPHA LABORATORIES LIMITED, and MEPRO PHARMACEUTICALS PRIVATE LIMITED, is crucial to mitigate these risks.
2Supply Chain Resilience
Waymade PLC's supply chain resilience is influenced by its concentrated sourcing strategy. The company's reliance on a select group of Indian suppliers for its primary products necessitates robust relationships and contingency planning. The diversity of formulations imported—212 unique formulations—indicates a degree of flexibility in product offerings. However, the heavy dependence on specific suppliers for key products like Citalopram and Hydrocortisone underscores the importance of maintaining strong, reliable partnerships.
To enhance supply chain resilience, Waymade PLC may consider diversifying its supplier base and exploring alternative sourcing options. Additionally, evaluating the regulatory compliance and manufacturing standards of its suppliers can further strengthen the supply chain's robustness.
3Strategic Implications
Waymade PLC's concentrated sourcing pattern positions the company as a specialized distributor within the UK's pharmaceutical market, focusing on specific therapeutic areas. This strategy allows for deep expertise and potentially stronger relationships with healthcare providers in these niches. For Indian exporters, understanding Waymade PLC's focus on CNS & Psychiatric and Corticosteroid products presents an opportunity to align their offerings with the company's needs.
However, the concentration also means that any disruptions in these product lines could significantly impact Waymade PLC's operations. Indian exporters seeking to become alternative suppliers should ensure they can meet the quality, regulatory, and logistical standards required by Waymade PLC to effectively integrate into their supply chain.
Importing Pharmaceuticals into United Kingdom — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Kingdom
1Regulatory Authority & Framework
In the United Kingdom, the Medicines and Healthcare products Regulatory Agency (MHRA) serves as the primary regulatory authority overseeing the safety, efficacy, and quality of medicines and medical devices. MHRA operates under the Human Medicines Regulations 2012, which implement European Union directives and establish the legal framework for the marketing and distribution of medicinal products in the UK.
For Indian pharmaceutical companies exporting to the UK, obtaining a Marketing Authorisation (MA) from the MHRA is essential. The MA process involves submitting comprehensive data on the product's quality, safety, and efficacy, as well as compliance with Good Manufacturing Practice (GMP) standards. The MHRA evaluates these submissions to ensure that only safe and effective medicines are available to UK patients.
2Import Licensing & GMP
Import licensing in the UK is governed by the Human Medicines Regulations 2012. Companies wishing to import medicinal products must hold a Wholesale Distribution Authorisation (WDA) issued by the MHRA. This authorisation permits the wholesale distribution of medicinal products for human use and is subject to compliance with GMP standards.
GMP certification is a critical requirement for pharmaceutical imports into the UK. The MHRA recognizes GMP certificates from countries with established regulatory frameworks, such as the European Union (EU GMP), the World Health Organization (WHO GMP), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian exporters must ensure that their manufacturing facilities are certified by one of these recognized bodies to meet UK import standards.
3Quality & Labeling
Pharmaceutical products imported into the UK must undergo batch testing to verify their quality, safety, and efficacy. This testing ensures that each batch meets the required standards before reaching the market.
Stability requirements are also stringent, with products needing to demonstrate consistent quality over their intended shelf life. Labeling must be in English and include essential information such as the product name, active ingredients, dosage instructions, and storage conditions. Serialization mandates may apply, requiring unique identifiers on packaging to enhance traceability and prevent counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, the UK has implemented several regulatory changes affecting pharmaceutical imports. These include updates to the Human Medicines Regulations 2012, aligning with new European Union directives and international standards. Changes have also been made to the WDA requirements, with stricter compliance measures and enhanced oversight to ensure product safety and quality.
Additionally, the MHRA has introduced more rigorous post-market surveillance and reporting requirements for imported medicines, aiming to strengthen pharmacovigilance and protect public health. Indian exporters should stay informed about these regulatory updates to ensure continued compliance and uninterrupted market access.
Waymade PLC — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Waymade PLC's focus on CNS & Psychiatric and Corticosteroid products aligns with the company's strategic emphasis on specialty medicines. The demand for these therapeutic areas is driven by the prevalence of mental health conditions and inflammatory disorders, which require effective and accessible treatments. By concentrating on these segments, Waymade PLC can leverage its expertise to meet specific patient needs and differentiate itself in the competitive pharmaceutical market.
The company's sourcing strategy, heavily reliant on specific products and suppliers, indicates a targeted approach to product development and market positioning. This focus allows Waymade PLC to build strong relationships with healthcare providers and patients within these therapeutic areas.
2Sourcing Profile
Waymade PLC's sourcing strategy is characterized by a concentrated approach, primarily importing finished pharmaceutical formulations from India. The company's portfolio is heavily weighted towards CNS & Psychiatric and Corticosteroid products, with a significant reliance on a select group of Indian suppliers. This strategy suggests a preference for established suppliers capable of meeting specific quality and regulatory standards.
India's robust pharmaceutical manufacturing sector, with its adherence to international GMP standards, aligns well with Waymade PLC's sourcing requirements. The country's capacity to produce a diverse range of formulations supports Waymade PLC's focus on product variety and quality.
Frequently Asked Questions — Waymade PLC
What products does Waymade PLC import from India?
Waymade PLC imports 2 pharmaceutical products across 2 categories. Top imports: Citalopram ($3.2M), Hydrocortisone ($1.0M).
Who supplies pharmaceuticals to Waymade PLC from India?
Waymade PLC sources from 10 verified Indian suppliers. The primary supplier is Apothecon Pharmaceuticals Private Limited (34.0% of imports, $3.3M).
What is Waymade PLC's total pharmaceutical import value?
Waymade PLC's total pharmaceutical import value from India is $4.2M, based on 121 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Waymade PLC focus on?
Waymade PLC imports across 2 categories. The largest: CNS & Psychiatric (75.5%), Corticosteroids (24.5%).
Get Full Waymade PLC Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Waymade PLC identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Waymade PLC's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 121 individual customs records matching Waymade PLC.
- 5.Supplier Verification: Waymade PLC sources from 10 verified Indian suppliers across 212 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.