Undp South Sudan
Pharmaceutical Importer · South Sudan · Advanced Antibiotics Focus · $1.3M Total Trade · DGFT Verified
Undp South Sudan is a pharmaceutical importer based in South Sudan with a total trade value of $1.3M across 2 products in 1 therapeutic categories. Based on 32 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Undp South Sudan sources from 9 verified Indian suppliers, with Cipla Limited accounting for 55.8% of imports.
Undp South Sudan — Import Portfolio & Supplier Network
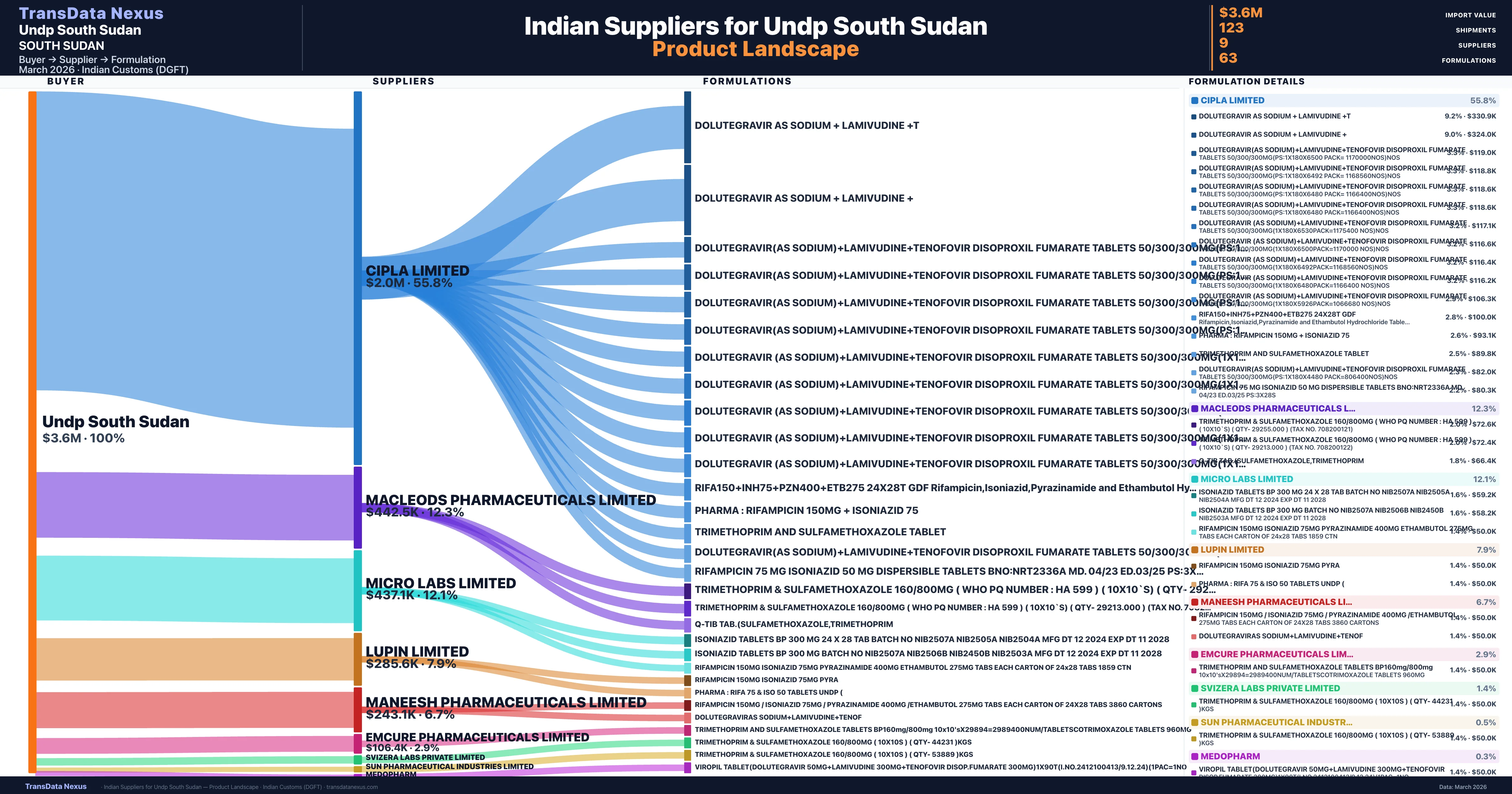
Who Are the Verified Indian Suppliers to Undp South Sudan?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $2.0M | 68 | 55.8% |
| Macleods Pharmaceuticals Limited | $442.5K | 19 | 12.3% |
| Micro Labs Limited | $437.1K | 15 | 12.1% |
| Lupin Limited | $285.6K | 8 | 7.9% |
| Maneesh Pharmaceuticals Limited | $243.1K | 5 | 6.7% |
| Emcure Pharmaceuticals Limited | $106.4K | 4 | 2.9% |
| Svizera Labs Private Limited | $50.0K | 1 | 1.4% |
| Sun Pharmaceutical Industries Limited | $18.1K | 2 | 0.5% |
| Medopharm | $11.7K | 1 | 0.3% |
Undp South Sudan sources from 9 verified Indian suppliers across 63 distinct formulations. The supply base is diversified across 9 suppliers, reducing single-source dependency risk.
What Formulations Does Undp South Sudan Import?
| Formulation | Value | Ships |
|---|---|---|
| Dolutegravir as sodium + lamivudine | $330.9K | 7 |
| Dolutegravir as sodium + lamivudine | $324.0K | 7 |
| Dolutegravir(as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(ps:1x180x6500 Pack= 1170000nos | $119.0K | 3 |
| Dolutegravir(as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(ps:1x180x6492 Pack= 1168560nos | $118.8K | 3 |
| Dolutegravir(as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(ps:1x180x6480 Pack= 1166400nos | $118.6K | 3 |
| Dolutegravir(as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(ps:1x180x6480 Pack=1166400nos | $118.6K | 3 |
| Dolutegravir (as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(1x180x6530pack=1175400 nos | $117.1K | 3 |
| Dolutegravir (as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(1x180x6500pack=1170000 nos | $116.6K | 3 |
| Dolutegravir (as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(1x180x6492pack=1168560nos | $116.4K | 3 |
| Dolutegravir (as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(1x180x6480pack=1166400 nos | $116.2K | 3 |
| Dolutegravir (as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(1x180x5926pack=1066680 nos | $106.3K | 3 |
| Rifa150+inh75+pzn400+etb275 24x28t gdf rifampicin,isoniazid,pyrazinamide and ethambutol hydrochloride tablet | $100.0K | 2 |
| Pharma : rifampicin 150MG + isoniazid | $93.1K | 2 |
| Trimethoprim and sulfamethoxazole | $89.8K | 2 |
| Dolutegravir(as sodium)+lamivudine+tenofovir disoproxil fumarate tablets 50/300/300MG(ps:1x180x4480 Pack=806400nos | $82.0K | 3 |
Undp South Sudan imports 63 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Undp South Sudan Import?
Top Products by Import Value
Undp South Sudan Therapeutic Categories — 1 Specializations
Undp South Sudan imports across 1 therapeutic categories, with Advanced Antibiotics (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
2 products · 100.0% · $1.3M
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Sulfamethoxazole | Advanced Antibiotics | $636.3K | 16 | 3.2% | 5 |
| 2 | Trimethoprim | Advanced Antibiotics | $636.3K | 16 | 2.4% | 5 |
Undp South Sudan imports 2 pharmaceutical products across 1 categories into South Sudan totaling $1.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Undp South Sudan.
Request DemoUndp South Sudan — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Undp South Sudan is a procurement entity operating under the United Nations Development Programme (UNDP) in South Sudan. As part of the UNDP's mission, it is responsible for sourcing and importing pharmaceutical products to support the country's healthcare system. The organization plays a pivotal role in ensuring the availability of essential medicines and medical supplies, particularly for programs targeting diseases such as HIV/AIDS and tuberculosis. By managing the procurement process, Undp South Sudan contributes to the effective delivery of healthcare services across the nation.
2Distribution Network
Undp South Sudan's distribution network is centered around key facilities, notably the Riverside Warehouse in Gumbo, Juba. This warehouse serves as a critical hub for storing and managing pharmaceutical supplies, including HIV/AIDS and tuberculosis medications. The facility employs advanced inventory management systems, such as mSupply, to monitor stock levels, manage orders, and ensure the safe storage of medicines. Additionally, the warehouse is equipped with cold chain storage to maintain the efficacy of temperature-sensitive products. Through this infrastructure, Undp South Sudan ensures the timely and efficient distribution of medical supplies to health facilities across the country.
3Industry Role
In South Sudan's pharmaceutical supply chain, Undp South Sudan functions as a primary wholesaler and distributor. It is responsible for the procurement, storage, and distribution of essential medicines and medical supplies to various health facilities nationwide. By managing these operations, Undp South Sudan ensures that healthcare providers have access to the necessary pharmaceuticals to deliver effective patient care. Its role is crucial in maintaining the continuity of treatment programs and supporting the overall health infrastructure in the country.
Supplier Relationship Intelligence — Undp South Sudan
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Undp South Sudan's procurement strategy exhibits a high degree of concentration, with a significant portion of its pharmaceutical imports sourced from India. The total import value from India amounts to $1.3 million USD, encompassing 32 shipments and 63 unique formulations. This concentration indicates a strategic choice to leverage India's established pharmaceutical manufacturing capabilities and cost-effectiveness. The consistent shipment data suggests a stable and reliable relationship between Undp South Sudan and its Indian suppliers, which is essential for maintaining a steady supply of essential medicines.
2Supply Chain Resilience
The resilience of Undp South Sudan's supply chain is bolstered by its diversified supplier base, comprising nine unique Indian pharmaceutical companies. This diversity mitigates the risks associated with over-reliance on a single supplier and enhances the stability of the supply chain. The inclusion of multiple suppliers also provides flexibility in sourcing and the ability to adapt to changing market conditions. However, the supply chain's resilience is contingent upon the reliability and compliance of these suppliers with international quality standards, as well as the robustness of the logistics infrastructure facilitating the importation process.
3Strategic Implications
Undp South Sudan's sourcing pattern, characterized by a concentrated focus on Indian suppliers, positions it to benefit from competitive pricing and a consistent supply of quality pharmaceuticals. For Indian exporters, this presents an opportunity to strengthen existing partnerships and explore avenues for expanding their product offerings to meet the evolving needs of the South Sudanese market. Additionally, the established procurement processes and quality standards adhered to by Undp South Sudan provide a framework for Indian exporters to align their products and services, thereby enhancing their competitiveness in the market.
Importing Pharmaceuticals into South Sudan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for South Sudan
1Regulatory Authority & Framework
The South Sudan Drug and Food Control Authority (SSDFCA) is the primary regulatory body overseeing the quality, safety, and efficacy of pharmaceuticals within the country. Established under the Drug and Food Control Authority Act, 2012, the SSDFCA is responsible for regulating the importation, manufacture, distribution, advertisement, and use of pharmaceutical products. The authority collaborates with the Ministry of Health, Ministry of Commerce, Industry and Investment, and other relevant agencies to enforce its mandate, ensuring that all pharmaceutical products meet national and international standards.
2Import Licensing & GMP
Importers of pharmaceutical products into South Sudan are required to obtain a License B from the SSDFCA, which authorizes them to import, wholesale, and distribute pharmaceutical products. Applicants must demonstrate financial capacity, provide suitable premises, and employ qualified professionals, such as pharmacists, to meet the regulatory requirements. Additionally, the SSDFCA mandates that imported pharmaceutical products comply with recognized Good Manufacturing Practice (GMP) standards, such as those from the European Union (EU GMP), World Health Organization (WHO GMP), or Pharmaceutical Inspection Co-operation Scheme (PIC/S). This ensures that imported medicines are of high quality and safe for public use.
3Quality & Labeling
Imported pharmaceutical products must adhere to specific quality and labeling standards set by the SSDFCA. All regulated products are required to be labeled with their generic or international non-proprietary names, in addition to the proprietary name, to ensure clarity and prevent confusion. The labeling must also include tamperproof and indelible imprints of manufacturing and expiry dates, batch or lot numbers, and other information as prescribed by regulations. These requirements are designed to ensure the safety, efficacy, and traceability of pharmaceutical products within South Sudan. (extranet.who.int)
4Recent Regulatory Changes
Between 2024 and 2026, South Sudan has implemented several policy changes affecting pharmaceutical imports. Notably, in July 2025, Gavi, the Vaccine Alliance, provided $2.6 million to strengthen South Sudan's supply chain system. This funding supports the expansion of cold-chain capacity and dry storage at key facilities, enhancing the country's ability to manage and distribute essential medicines effectively. Such initiatives reflect a commitment to improving the pharmaceutical supply chain infrastructure and ensuring the availability of quality medicines to the population.
Undp South Sudan — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Undp South Sudan's focus on importing advanced antibiotics, specifically sulfamethoxazole and trimethoprim, indicates a strategic approach to addressing prevalent infectious diseases within the country. The substantial import values of these products, each amounting to $636,000 USD, underscore their critical role in the national healthcare strategy. The decision to concentrate on these therapeutic areas is likely driven by the high incidence of bacterial infections and the need for effective antimicrobial treatments to improve public health outcomes.
2Sourcing Profile
Undp South Sudan's sourcing strategy emphasizes the procurement of generic pharmaceutical formulations, primarily from India. This approach leverages India's robust pharmaceutical manufacturing sector, known for producing high-quality generics at competitive prices. The focus on finished pharmaceutical formulations, rather than raw active pharmaceutical ingredients (APIs), aligns with the immediate needs of the South Sudanese healthcare system, ensuring the availability of ready-to-use medicines for patients.
3Market Positioning
By importing and distributing essential medicines, Undp South Sudan serves a critical role in the healthcare sector, positioning itself as a key supplier to government health facilities, hospitals, and clinics. Its operations support the public health infrastructure, ensuring that healthcare providers have access to necessary pharmaceuticals to deliver effective patient care. This positioning underscores Undp South Sudan's commitment to enhancing the health and well-being of the South Sudanese population.
Seller's Guide — How to Become a Supplier to Undp South Sudan
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Undp South Sudan, particularly those offering high-quality generic formulations that meet international GMP standards. The existing concentration on a limited number of products suggests potential gaps in the product portfolio, which new suppliers could fill. Additionally, the ongoing strengthening of the pharmaceutical supply chain in South Sudan presents an opportunity for suppliers to establish long-term partnerships and contribute to the country's health sector development.
2Requirements & Qualifications
Indian exporters seeking to supply Undp South Sudan must ensure that their products comply with recognized GMP standards, such as EU GMP, WHO GMP, or PIC/S. Additionally, they must adhere to the labeling requirements set by the SSDFCA, including the use of generic or international non-proprietary names and the inclusion of manufacturing and expiry dates, batch numbers, and other prescribed information. Establishing a partnership with Undp South Sudan also requires a thorough understanding of the regulatory framework governing pharmaceutical imports into South Sudan and a commitment to maintaining high-quality standards.
3How to Approach
To establish a relationship with Undp South Sudan, Indian exporters should begin by ensuring their products meet the necessary GMP standards and labeling requirements. Engaging with the SSDFCA to obtain the required import licenses and approvals is a crucial step. Participating in UNDP procurement processes, which may include responding to Requests for Proposals (RFPs) or Invitations to Bid (ITBs), is essential. Building a strong understanding of the local market dynamics and demonstrating a commitment to quality and reliability will enhance the prospects of successful collaboration with Undp South Sudan.
Frequently Asked Questions — Undp South Sudan
What products does Undp South Sudan import from India?
Undp South Sudan imports 2 pharmaceutical products across 1 categories. Top imports: Sulfamethoxazole ($636.3K), Trimethoprim ($636.3K).
Who supplies pharmaceuticals to Undp South Sudan from India?
Undp South Sudan sources from 9 verified Indian suppliers. The primary supplier is Cipla Limited (55.8% of imports, $2.0M).
What is Undp South Sudan's total pharmaceutical import value?
Undp South Sudan's total pharmaceutical import value from India is $1.3M, based on 32 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Undp South Sudan focus on?
Undp South Sudan imports across 1 categories. The largest: Advanced Antibiotics (100.0%).
Get Full Undp South Sudan Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Undp South Sudan identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Undp South Sudan's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 32 individual customs records matching Undp South Sudan.
- 5.Supplier Verification: Undp South Sudan sources from 9 verified Indian suppliers across 63 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.