Trans Sped AG - SMI
Pharmaceutical Importer · Switzerland · Antimalarial & Antiparasitic Focus · $263.5K Total Trade · DGFT Verified
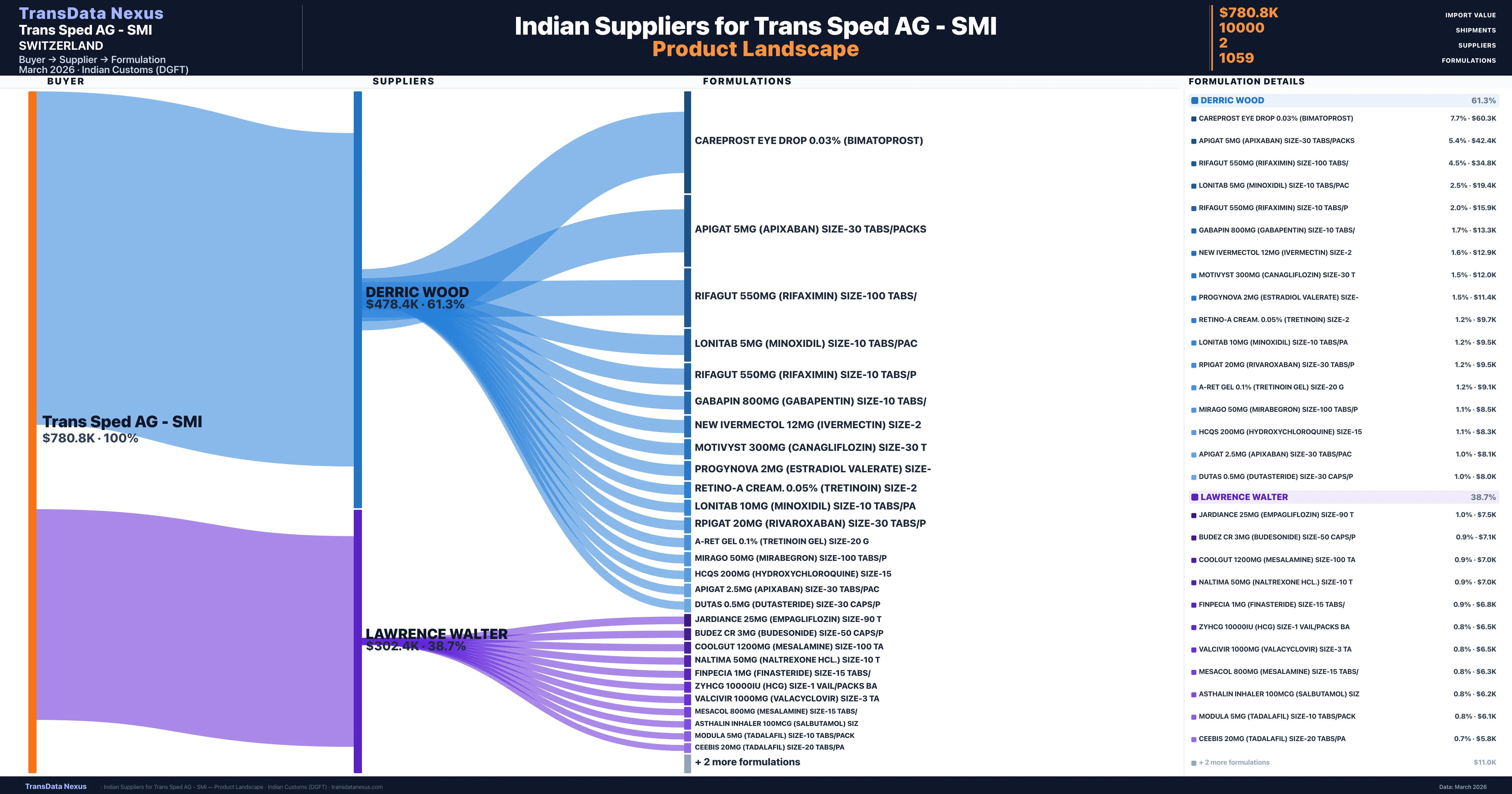
Trans Sped AG - SMI is a pharmaceutical importer based in Switzerland with a total trade value of $263.5K across 3 products in 3 therapeutic categories. Based on 2,170 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Trans Sped AG - SMI sources from 2 verified Indian suppliers, with Derric Wood accounting for 61.3% of imports.
Trans Sped AG - SMI — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Trans Sped AG - SMI?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Derric Wood | $478.4K | 7,152 | 61.3% |
| Lawrence Walter | $302.4K | 2,848 | 38.7% |
Trans Sped AG - SMI sources from 2 verified Indian suppliers across 1,059 distinct formulations. Total import value: $780.8K across 10,000 shipments.
What Formulations Does Trans Sped AG - SMI Import?
| Formulation | Value | Ships |
|---|---|---|
| Careprost eye drop 0.03% (bimatoprost) | $60.3K | 23 |
| Apigat 5MG (apixaban) size-30 | $42.4K | 25 |
| Rifagut 550MG (rifaximin) size-100 | $34.8K | 20 |
| Lonitab 5MG (minoxidil) size-10 | $19.4K | 26 |
| Rifagut 550MG (rifaximin) size-10 | $15.9K | 24 |
| Gabapin 800MG (gabapentin) size-10 | $13.3K | 25 |
| New ivermectol 12MG (ivermectin) | $12.9K | 23 |
| Motivyst 300MG (canagliflozin) size-30 | $12.0K | 20 |
| Progynova 2MG (estradiol valerate) | $11.4K | 30 |
| Retino-a cream. 0.05% (tretinoin) | $9.7K | 26 |
| Lonitab 10MG (minoxidil) size-10 | $9.5K | 11 |
| Rpigat 20MG (rivaroxaban) size-30 | $9.5K | 20 |
| A-ret gel 0.1% (tretinoin gel) size-20 | $9.1K | 24 |
| Mirago 50MG (mirabegron) size-100 | $8.5K | 19 |
| Hcqs 200MG (hydroxychloroquine) | $8.3K | 25 |
Trans Sped AG - SMI imports 1,059 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Trans Sped AG - SMI Import?
Top Products by Import Value
Trans Sped AG - SMI Therapeutic Categories — 3 Specializations
Trans Sped AG - SMI imports across 3 therapeutic categories, with Antimalarial & Antiparasitic (56.7%), Advanced Diabetes Medications (23.3%), Immunosuppressants (20.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antimalarial & Antiparasitic
1 products · 56.7% · $149.3K
Advanced Diabetes Medications
1 products · 23.3% · $61.5K
Immunosuppressants
1 products · 20.0% · $52.7K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ivermectin | Antimalarial & Antiparasitic | $149.3K | 1,525 | 1.7% | 7 |
| 2 | Canagliflozin | Advanced Diabetes Medications | $61.5K | 241 | 7.0% | 4 |
| 3 | Sirolimus | Immunosuppressants | $52.7K | 404 | 0.4% | 13 |
Trans Sped AG - SMI imports 3 pharmaceutical products across 3 categories into Switzerland totaling $263.5K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Trans Sped AG - SMI.
Request DemoTrans Sped AG - SMI — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Trans Sped AG is a Swiss company specializing in international transport and logistics services, including customs clearance and warehousing. Established in 1989, the company has been serving clients with a focus on efficient and reliable logistics solutions. Headquartered in Embrach, Switzerland, Trans Sped AG operates additional facilities at Zurich Airport, indicating a strategic presence in key logistics hubs. (transsped.ch)
While Trans Sped AG's primary business revolves around logistics and transport services, it also engages in the importation of pharmaceutical products. The company's role in the Swiss pharmaceutical distribution network includes sourcing and importing finished pharmaceutical formulations from India, which are then distributed within Switzerland. This dual focus on logistics and pharmaceutical importation positions Trans Sped AG as a significant player in the Swiss pharmaceutical supply chain.
2Distribution Network
Trans Sped AG's distribution network is strategically designed to ensure efficient logistics operations across Switzerland and beyond. The company operates warehouses in Embrach and at Zurich Airport, facilitating streamlined storage and distribution processes. These locations are well-connected to major transportation routes, enabling swift movement of goods. Additionally, Trans Sped AG's customs clearance services are integrated into their logistics operations, ensuring compliance with Swiss regulations and expediting the import and export processes. (transsped.ch)
The company's logistics capabilities encompass land transport, airfreight, sea freight, and customs clearance, providing a comprehensive suite of services to meet diverse client needs. This extensive service offering allows Trans Sped AG to cater to a wide range of industries, including pharmaceuticals, by ensuring timely and secure delivery of goods. (transsped.ch)
3Industry Role
In the Swiss pharmaceutical supply chain, Trans Sped AG functions as a pharmaceutical importer and logistics intermediary. The company imports finished pharmaceutical formulations from India, handling the customs clearance and warehousing processes, and subsequently distributes these products within Switzerland. This role is crucial in ensuring the availability of imported pharmaceutical products in the Swiss market, bridging the gap between international manufacturers and local healthcare providers.
By managing both the importation and distribution aspects, Trans Sped AG plays a pivotal role in maintaining the efficiency and reliability of the pharmaceutical supply chain in Switzerland. Their expertise in logistics and customs procedures ensures that imported pharmaceutical products comply with Swiss regulations and reach their intended destinations without delay.
Supplier Relationship Intelligence — Trans Sped AG - SMI
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Trans Sped AG's sourcing strategy exhibits a high degree of concentration, with a significant portion of their pharmaceutical imports originating from two primary suppliers: DERRIC WOOD and LAWRENCE WALTER. DERRIC WOOD accounts for 61.3% of the total import value, while LAWRENCE WALTER contributes 38.7%. This concentration indicates a reliance on a limited number of suppliers for pharmaceutical products.
The stability of this supplier relationship is evidenced by the consistent volume of shipments over the years. However, such dependency on a small number of suppliers can pose risks, including potential supply disruptions or price fluctuations. To mitigate these risks, Trans Sped AG may consider diversifying its supplier base and establishing contingency plans to ensure a continuous and reliable supply of pharmaceutical products.
2Supply Chain Resilience
Trans Sped AG's supply chain resilience is influenced by its sourcing strategy, which is heavily concentrated on two suppliers from India. This concentration can lead to vulnerabilities, such as exposure to supply disruptions or regulatory changes affecting these suppliers. The company's reliance on a limited number of suppliers may also impact its ability to negotiate favorable terms or adapt to market changes swiftly.
To enhance supply chain resilience, Trans Sped AG could explore partnerships with additional suppliers, both within India and globally, to diversify its sourcing options. Implementing robust risk management strategies, including regular assessments of supplier performance and market conditions, would further strengthen the company's ability to maintain a steady supply of pharmaceutical products.
3Strategic Implications
Trans Sped AG's concentrated sourcing pattern from two primary suppliers positions the company as a significant partner for these suppliers, potentially securing favorable terms and priority access to products. However, this dependency also exposes Trans Sped AG to risks associated with supply chain disruptions or changes in supplier dynamics.
For Indian exporters, the existing relationship between Trans Sped AG and its current suppliers may present challenges in entering the Swiss market through this channel. New entrants would need to demonstrate competitive advantages, such as superior product quality, cost-effectiveness, or unique formulations, to capture Trans Sped AG's attention and establish a partnership.
Importing Pharmaceuticals into Switzerland — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Switzerland
1Regulatory Authority & Framework
In Switzerland, the primary regulatory authority overseeing pharmaceutical products is Swissmedic, the Swiss Agency for Therapeutic Products. Swissmedic is responsible for ensuring the safety, efficacy, and quality of medicinal products available in Switzerland. The regulatory framework governing pharmaceutical imports includes the Therapeutic Products Act (TPA) and the Therapeutic Products Ordinance (TPO), which outline the requirements for marketing authorization, importation, and distribution of pharmaceutical products.
For Indian generics to be marketed in Switzerland, they must undergo a parallel import procedure, which involves obtaining marketing authorization from Swissmedic. This process ensures that imported products meet Swiss standards and are interchangeable with the original products authorized in Switzerland. The parallel import procedure requires compliance with specific labeling, packaging, and documentation standards to facilitate the acceptance of the imported products in the Swiss market. (swissmedic.ch)
2Import Licensing & GMP
Import licensing in Switzerland is governed by Swissmedic, which issues import licenses to companies involved in the importation of pharmaceutical products. To obtain an import license, companies must demonstrate compliance with Good Manufacturing Practice (GMP) standards, ensuring that imported products are manufactured in facilities adhering to recognized quality standards.
Swissmedic recognizes GMP certificates from various international bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). This recognition facilitates the importation of pharmaceutical products from countries with established GMP standards, such as India. Additionally, companies must obtain wholesale distribution authorization to distribute pharmaceutical products within Switzerland, ensuring compliance with local regulations and standards.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. Swissmedic conducts these tests to ensure that products meet the required standards before they are released into the Swiss market.
Stability requirements for pharmaceutical products are stringent, ensuring that products maintain their quality throughout their shelf life. Labeling must be in one of the official Swiss languages (German, French, or Italian) and include comprehensive information for healthcare professionals and patients. Serialization mandates are in place to track and trace pharmaceutical products, enhancing supply chain security and preventing counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Swissmedic has implemented several policy changes affecting the importation of pharmaceutical products, including those from India. These changes encompass stricter requirements for marketing authorization, enhanced documentation standards, and updated labeling and packaging regulations. The aim is to improve the safety and efficacy of imported pharmaceutical products and to align with international standards.
Additionally, there have been updates to the parallel import procedure, including more detailed guidelines for labeling and packaging to ensure that imported products are clearly distinguishable from original products authorized in Switzerland. These regulatory changes necessitate that importers, such as Trans Sped AG, stay informed and compliant to maintain uninterrupted access to the Swiss pharmaceutical market. (swissmedic.ch)
Trans Sped AG - SMI — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Trans Sped AG's focus on importing pharmaceutical products in the therapeutic categories of Antimalarial & Antiparasitic, Advanced Diabetes Medications, and Immunosuppressants indicates a strategic alignment with prevalent health concerns in Switzerland. The importation of Ivermectin, Canagliflozin, and Sirolimus suggests a response to the demand for treatments addressing parasitic infections, diabetes management, and immune system modulation.
The company's product strategy is likely driven by market demand, regulatory considerations, and the availability of these products from Indian suppliers. By focusing on these therapeutic areas, Trans Sped AG aims to meet the needs of healthcare providers and patients in Switzerland, ensuring access to essential medications.
2Sourcing Profile
Trans Sped AG's sourcing strategy for pharmaceutical products involves importing finished formulations from India, a country known for its robust pharmaceutical manufacturing sector. The company's preference for sourcing from India is likely due to the availability of high-quality generics at competitive prices, as well as the established regulatory frameworks that facilitate the importation of Indian pharmaceuticals into Switzerland.
By focusing on finished formulations, Trans Sped AG ensures that the imported products are ready for distribution, reducing the complexities associated with sourcing raw active pharmaceutical ingredients (APIs) and manufacturing processes. This approach streamlines the supply chain and allows the company to focus on logistics and distribution within Switzerland.
3Market Positioning
Based on its product mix, Trans Sped AG serves the wholesale distribution segment of the Swiss pharmaceutical market.
Frequently Asked Questions — Trans Sped AG - SMI
What products does Trans Sped AG - SMI import from India?
Trans Sped AG - SMI imports 3 pharmaceutical products across 3 categories. Top imports: Ivermectin ($149.3K), Canagliflozin ($61.5K), Sirolimus ($52.7K).
Who supplies pharmaceuticals to Trans Sped AG - SMI from India?
Trans Sped AG - SMI sources from 2 verified Indian suppliers. The primary supplier is Derric Wood (61.3% of imports, $478.4K).
What is Trans Sped AG - SMI's total pharmaceutical import value?
Trans Sped AG - SMI's total pharmaceutical import value from India is $263.5K, based on 2,170 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Trans Sped AG - SMI focus on?
Trans Sped AG - SMI imports across 3 categories. The largest: Antimalarial & Antiparasitic (56.7%), Advanced Diabetes Medications (23.3%), Immunosuppressants (20.0%).
Get Full Trans Sped AG - SMI Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Trans Sped AG - SMI identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Trans Sped AG - SMI's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 2,170 individual customs records matching Trans Sped AG - SMI.
- 5.Supplier Verification: Trans Sped AG - SMI sources from 2 verified Indian suppliers across 1,059 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.