`TO THE Order OF
Pharmaceutical Importer · Brazil · Advanced Oncology Focus · $37.3K Total Trade · DGFT Verified
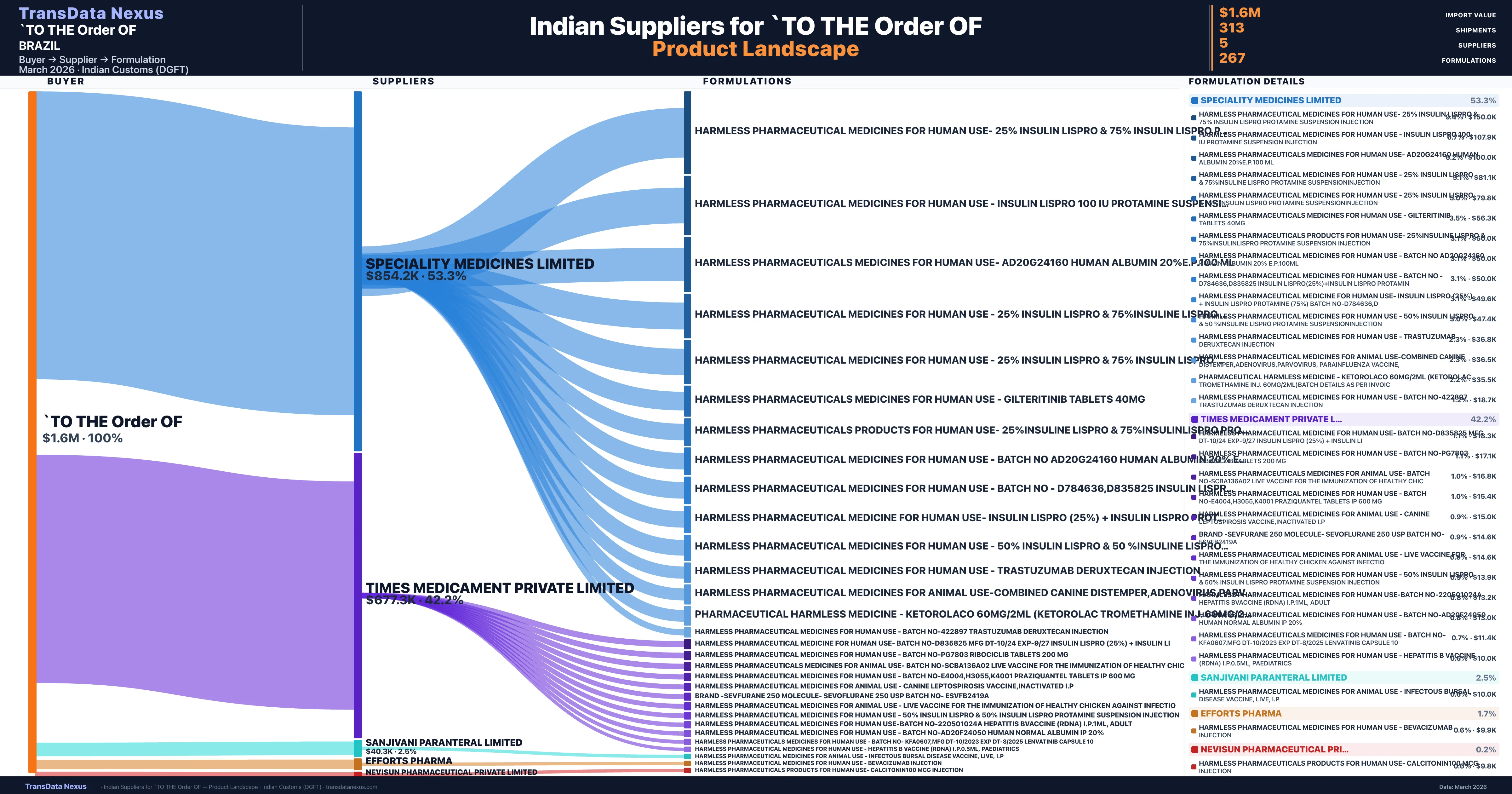
`TO THE Order OF is a pharmaceutical importer based in Brazil with a total trade value of $37.3K across 2 products in 1 therapeutic categories. Based on 9 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. `TO THE Order OF sources from 5 verified Indian suppliers, with Speciality Medicines Limited accounting for 53.3% of imports.
`TO THE Order OF — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to `TO THE Order OF?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Speciality Medicines Limited | $854.2K | 264 | 53.3% |
| Times Medicament Private Limited | $677.3K | 31 | 42.2% |
| Sanjivani Paranteral Limited | $40.3K | 2 | 2.5% |
| Efforts Pharma | $27.7K | 6 | 1.7% |
| Nevisun Pharmaceutical Private Limited | $3.8K | 10 | 0.2% |
`TO THE Order OF sources from 5 verified Indian suppliers across 267 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does `TO THE Order OF Import?
| Formulation | Value | Ships |
|---|---|---|
| Harmless pharmaceutical medicines for human use- 25% insulin lispro & 75% insulin lispro protamine suspension | $150.0K | 3 |
| Harmless pharmaceutical medicines for human use - insulin lispro 100 IU protamine suspension | $107.9K | 4 |
| Harmless pharmaceuticals medicines for human use- ad20g24160 human albumin 20%e.p.100 ML | $100.0K | 2 |
| Harmless pharmaceutical medicines for human use - 25% insulin lispro & 75%insuline lispro protamine | $81.1K | 2 |
| Harmless pharmaceutical medicines for human use - 25% insulin lispro & 75% insulin lispro protamine | $79.8K | 2 |
| Harmless pharmaceuticals medicines for human use - gilteritinib tablets 40MG | $56.3K | 6 |
| Harmless pharmaceuticals products for human use- 25%insuline lispro & 75%insulinlispro protamine suspension | $50.0K | 1 |
| Harmless pharmaceutical medicines for human use - batch no ad20g24160 human albumin 20% e.p.100ML | $50.0K | 1 |
| Harmless pharmaceutical medicines for human use - batch no - d784636,d835825 insulin lispro(25%)+insulin lispro | $50.0K | 1 |
| Harmless pharmaceutical medicine for human use- insulin lispro (25%) + insulin lispro protamine (75%) batch no-d784636 | $49.6K | 1 |
| Harmless pharmaceutical medicines for human use - 50% insulin lispro & 50 %insuline lispro protamine | $47.4K | 2 |
| Harmless pharmaceutical medicines for human use - trastuzumab deruxtecan | $36.8K | 4 |
| Harmless pharmaceutical medicines for animal use-combined canine distemper,adenovirus,parvovirus, parainfluenza vaccine | $36.5K | 1 |
| Pharmaceutical harmless medicine - ketorolaco 60MG/2ML (ketorolac tromethamine INJ. 60MG/2ML)batch details as per | $35.5K | 1 |
| Harmless pharmaceutical medicines for human use - batch no-422897 trastuzumab deruxtecan | $18.7K | 2 |
`TO THE Order OF imports 267 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does `TO THE Order OF Import?
Top Products by Import Value
`TO THE Order OF Therapeutic Categories — 1 Specializations
`TO THE Order OF imports across 1 therapeutic categories, with Advanced Oncology (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
2 products · 100.0% · $37.3K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Lenvatinib | Advanced Oncology | $22.6K | 6 | 1.1% | 10 |
| 2 | Olaparib | Advanced Oncology | $14.8K | 3 | 1.6% | 8 |
`TO THE Order OF imports 2 pharmaceutical products across 1 categories into Brazil totaling $37.3K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for `TO THE Order OF.
Request Demo`TO THE Order OF — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
`TO THE Order OF` is a pharmaceutical importer based in Brazil, specializing in the acquisition of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company has engaged in nine shipments from India, totaling an import value of $37,000 USD. The primary products imported are Lenvatinib and Olaparib, both utilized in advanced oncology treatments. The company's operations are integral to Brazil's pharmaceutical distribution network, focusing on sourcing specific oncology medications to meet domestic demand.
2Distribution Network
While specific details about `TO THE Order OF`'s warehouse locations and logistics capabilities are not publicly available, the company's import activities suggest a distribution network capable of handling specialized pharmaceutical products. The focus on advanced oncology medications indicates a targeted approach to distribution, likely involving partnerships with healthcare providers and institutions across Brazil. The company's logistics operations are presumably designed to ensure the timely and compliant delivery of sensitive pharmaceutical products.
3Industry Role
`TO THE Order OF` functions as a pharmaceutical importer, sourcing finished formulations from Indian manufacturers to supply the Brazilian market. By focusing on advanced oncology treatments, the company plays a crucial role in addressing the specific therapeutic needs within Brazil's healthcare system. Its operations contribute to the diversification of the pharmaceutical supply chain, ensuring access to specialized medications that may not be readily available from domestic producers.
Supplier Relationship Intelligence — `TO THE Order OF
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The company's sourcing strategy is highly concentrated, with 100% of its imports comprising two products: Lenvatinib and Olaparib. This dual-product focus indicates a strategic decision to specialize in advanced oncology treatments, aligning with specific market demands. The consistent importation of these products suggests a stable and ongoing relationship with Indian suppliers, reflecting reliability and mutual trust. However, this concentration also exposes the company to risks associated with supply chain disruptions or regulatory changes affecting these specific products.
2Supply Chain Resilience
`TO THE Order OF` demonstrates resilience in its supply chain by maintaining relationships with multiple Indian suppliers, including SPECIALITY MEDICINES LIMITED, TIMES MEDICAMENT PRIVATE LIMITED, and others. This diversification mitigates risks associated with dependency on a single supplier. The importation of 267 unique formulations indicates a broad sourcing strategy within the oncology therapeutic category, enhancing the company's ability to adapt to changing market demands. While the focus on advanced oncology treatments suggests a specialized supply chain, the company's multiple supplier relationships and product diversity contribute to its overall resilience.
3Strategic Implications
The concentrated sourcing pattern of `TO THE Order OF` positions the company as a specialized importer of advanced oncology medications, catering to a niche segment of the Brazilian pharmaceutical market. This specialization allows the company to build expertise and establish strong relationships with both suppliers and healthcare providers. For Indian exporters, this focus presents an opportunity to become key suppliers of high-demand oncology products, potentially leading to long-term partnerships. However, the limited product range may also restrict the company's ability to diversify its offerings and respond to broader market shifts.
Importing Pharmaceuticals into Brazil — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Brazil
1Regulatory Authority & Framework
In Brazil, the National Health Surveillance Agency (ANVISA) is the primary regulatory body overseeing the pharmaceutical sector. ANVISA is responsible for the registration, inspection, and monitoring of pharmaceutical products, ensuring they meet safety, efficacy, and quality standards. The regulatory framework governing pharmaceutical imports includes Law No. 6,360/1976, which establishes procedures for the importation of medicines, and various resolutions such as RDC 753/2022, which outlines requirements for new synthetic and semi-synthetic drugs. These regulations ensure that imported pharmaceutical products comply with Brazilian health standards. (gov.br)
2Import Licensing & GMP
Importers of pharmaceutical products into Brazil must obtain an Import Authorization (AI) from ANVISA before the shipment reaches the country. This authorization ensures that the imported products are registered and comply with Brazilian health regulations. Additionally, ANVISA requires that foreign manufacturers provide proof of Good Manufacturing Practices (GMP) certification, aligning with international standards such as those set by the Pharmaceutical Inspection Co-operation Scheme (PIC/S). This certification is essential for maintaining the quality and safety of pharmaceutical products imported into Brazil. (gov.br)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality and compliance with Brazilian standards. Stability studies are required to ensure that products maintain their efficacy and safety throughout their shelf life. Labeling must be in Portuguese and include detailed information such as dosage instructions, indications, contraindications, and storage conditions. Serialization mandates are in place to facilitate traceability and prevent counterfeit products from entering the market. These requirements are part of ANVISA's efforts to protect public health and ensure the integrity of the pharmaceutical supply chain. (softgroup.eu)
4Recent Regulatory Changes
Between 2024 and 2026, ANVISA implemented several regulatory changes affecting pharmaceutical imports into Brazil. Notably, RDC 430/2020, which came into force on March 16, 2021, established new Good Distribution Practices for the distribution, storage, and transport of medicines. This regulation introduced mandatory temperature and humidity monitoring for pharmaceutical products, effective from March 17, 2022. These changes aim to enhance the quality and safety of pharmaceutical products throughout the supply chain.
`TO THE Order OF — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
`TO THE Order OF` focuses on importing advanced oncology medications, specifically Lenvatinib and Olaparib. This strategic choice aligns with the growing demand for specialized cancer treatments in Brazil. The importation of these products addresses the need for effective therapies in the oncology sector, catering to patients requiring advanced treatment options. The company's product strategy reflects a commitment to providing high-quality, specialized medications to meet critical healthcare needs.
2Sourcing Profile
The company's sourcing strategy is centered on importing generic formulations of Lenvatinib and Olaparib from India. This approach leverages India's established pharmaceutical manufacturing capabilities and cost advantages. By focusing on these specific formulations, `TO THE Order OF` aims to provide cost-effective treatment options for advanced oncology patients in Brazil. India's role as a major supplier of generic drugs positions it as a key partner in the company's procurement strategy.
3Market Positioning
Based on its product mix, `TO THE Order OF` serves a specialized segment of the Brazilian pharmaceutical market, focusing on advanced oncology treatments. The company's imports are likely distributed through hospital pharmacies and specialized clinics, catering to patients requiring advanced cancer therapies. By concentrating on this niche market, `TO THE Order OF` positions itself as a key supplier of critical oncology medications, addressing specific therapeutic needs within Brazil's healthcare system.
Seller's Guide — How to Become a Supplier to `TO THE Order OF
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter the Brazilian market by offering high-quality generic formulations of oncology medications. `TO THE Order OF`'s focus on Lenvatinib and Olaparib indicates a demand for these products, presenting a potential avenue for suppliers to establish partnerships. However, the competitive landscape requires suppliers to meet ANVISA's stringent regulatory requirements, including GMP certification and compliance with import licensing procedures. Suppliers must also navigate the complexities of Brazil's pharmaceutical distribution network to effectively reach the target market.
2Requirements & Qualifications
Indian exporters seeking to supply `TO THE Order OF` and the Brazilian market must obtain GMP certification recognized by ANVISA, aligning with international standards such as those set by PIC/S. They must also comply with ANVISA's import licensing requirements, including obtaining an Import Authorization (AI) before shipment. Additionally, exporters must ensure that their products meet ANVISA's quality and labeling standards, including batch testing, stability studies, and labeling in Portuguese. Adherence to these requirements is essential for successful market entry and distribution in Brazil.
3How to Approach
Indian exporters should initiate the relationship with `TO THE Order OF` by demonstrating compliance with ANVISA's regulatory requirements, including GMP certification and import licensing. Participating in tenders and responding to requests for proposals can facilitate entry into the Brazilian market. A clear regulatory filing strategy, including timely submission of necessary documentation to ANVISA, is crucial. Exporters should also establish a robust logistics plan to navigate Brazil's distribution network effectively. Setting realistic timelines for regulatory approvals and market entry will aid in managing expectations and ensuring a successful partnership.
Frequently Asked Questions — `TO THE Order OF
What products does `TO THE Order OF import from India?
`TO THE Order OF imports 2 pharmaceutical products across 1 categories. Top imports: Lenvatinib ($22.6K), Olaparib ($14.8K).
Who supplies pharmaceuticals to `TO THE Order OF from India?
`TO THE Order OF sources from 5 verified Indian suppliers. The primary supplier is Speciality Medicines Limited (53.3% of imports, $854.2K).
What is `TO THE Order OF's total pharmaceutical import value?
`TO THE Order OF's total pharmaceutical import value from India is $37.3K, based on 9 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does `TO THE Order OF focus on?
`TO THE Order OF imports across 1 categories. The largest: Advanced Oncology (100.0%).
Get Full `TO THE Order OF Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: `TO THE Order OF identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as `TO THE Order OF's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 9 individual customs records matching `TO THE Order OF.
- 5.Supplier Verification: `TO THE Order OF sources from 5 verified Indian suppliers across 267 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.