TO THE Manager-
Pharmaceutical Importer · Brazil · Advanced Diabetes Medications Focus · $94.6K Total Trade · DGFT Verified
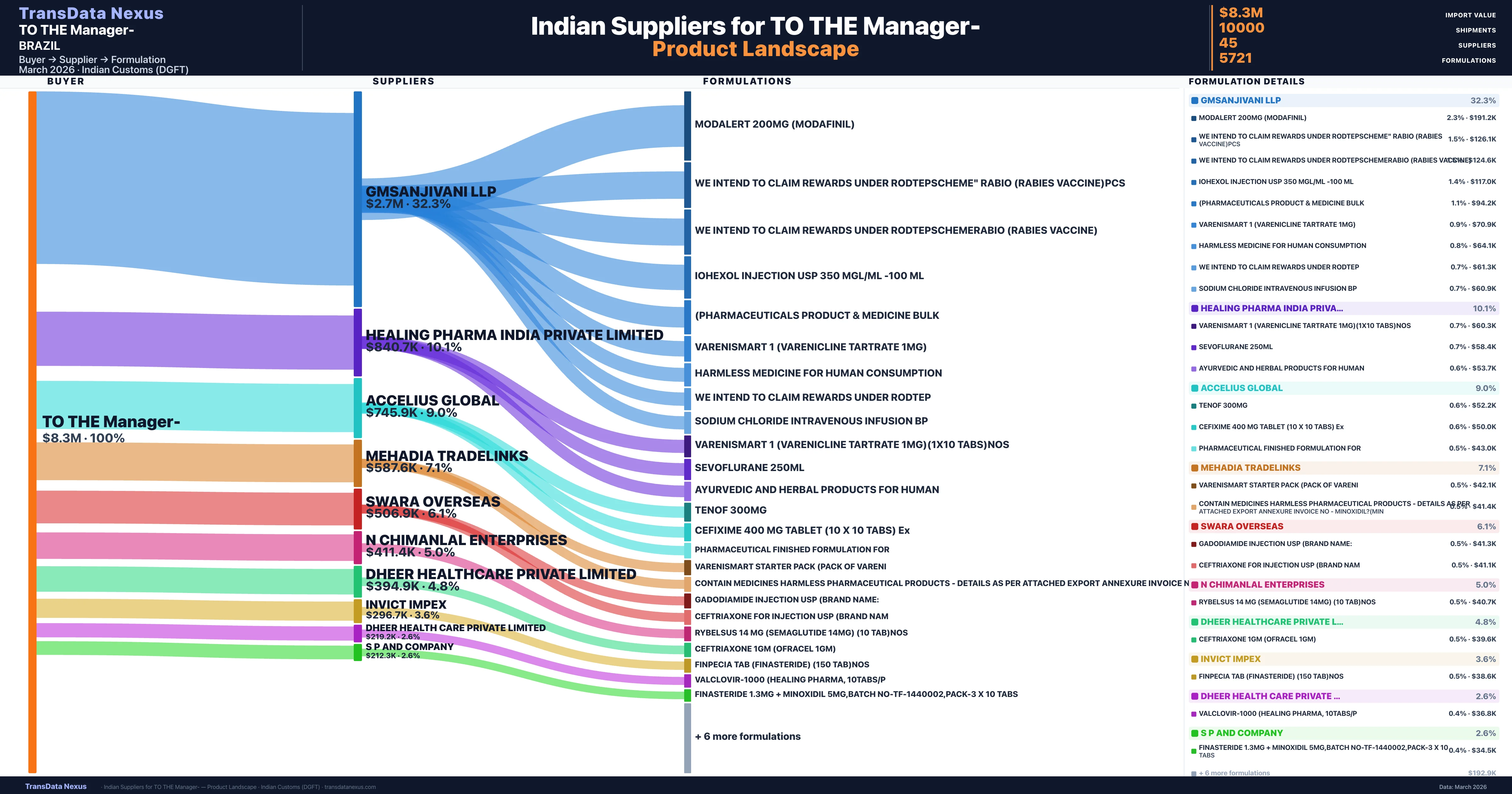
TO THE Manager- is a pharmaceutical importer based in Brazil with a total trade value of $94.6K across 4 products in 2 therapeutic categories. Based on 38 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. TO THE Manager- sources from 45 verified Indian suppliers, with Gmsanjivani Llp accounting for 32.3% of imports.
TO THE Manager- — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to TO THE Manager-?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Gmsanjivani Llp | $2.7M | 2,559 | 32.3% |
| Healing Pharma India Private Limited | $840.7K | 699 | 10.1% |
| Accelius Global | $745.9K | 107 | 9.0% |
| Mehadia Tradelinks | $587.6K | 209 | 7.1% |
| Swara Overseas | $506.9K | 829 | 6.1% |
| N Chimanlal Enterprises | $411.4K | 1,560 | 5.0% |
| Dheer Healthcare Private Limited | $394.9K | 40 | 4.8% |
| Invict Impex | $296.7K | 829 | 3.6% |
| Dheer Health Care Private Limited | $219.2K | 47 | 2.6% |
| S P And Company | $212.3K | 377 | 2.6% |
| Melon Globalcare | $195.6K | 393 | 2.4% |
| Goldfield Healthcare | $158.4K | 181 | 1.9% |
| Saintroy Lifescience Llp | $147.3K | 13 | 1.8% |
| P.b.p Enterprises Private Limited | $117.5K | 141 | 1.4% |
| Mhj Pharmaconcepts Private Limited | $114.8K | 10 | 1.4% |
| Mds Enterprises | $100.6K | 201 | 1.2% |
| Ratna Biopharms | $75.1K | 7 | 0.9% |
| American Remedies Healthcare Private Limited | $73.8K | 108 | 0.9% |
| Prahem Laboratories Llp | $71.1K | 10 | 0.9% |
| Medigen Pharma | $63.5K | 141 | 0.8% |
| San Marino Impex | $38.3K | 740 | 0.5% |
| Gospel Pharmaceuticals | $32.1K | 8 | 0.4% |
| Govadalu Bilateral | $30.1K | 101 | 0.4% |
| Unicure Remedies Private Limited | $27.7K | 2 | 0.3% |
| Mhj Biocure Private Limited | $26.2K | 2 | 0.3% |
| P.m. Trading Company | $24.7K | 7 | 0.3% |
| P B P Enterprises Private Limited | $23.4K | 15 | 0.3% |
| Ambica Pharma | $21.4K | 4 | 0.3% |
| Ananta Pacific | $10.8K | 340 | 0.1% |
| Itt Engineering India Private Limited | $7.0K | 33 | 0.1% |
TO THE Manager- sources from 45 verified Indian suppliers across 5,721 distinct formulations. The supply base is diversified across 45 suppliers, reducing single-source dependency risk.
What Formulations Does TO THE Manager- Import?
| Formulation | Value | Ships |
|---|---|---|
| Modalert 200MG (modafinil) | $191.2K | 18 |
| We intend to claim rewards under rodtepscheme" rabio (rabies vaccine | $126.1K | 4 |
| We intend to claim rewards under rodtepschemerabio (rabies vaccine) | $124.6K | 5 |
| Iohexol injection USP 350 mgl/ML -100 ML | $117.0K | 3 |
| (pharmaceuticals product & medicine | $94.2K | 162 |
| Varenismart 1 (varenicline tartrate 1MG) | $70.9K | 3 |
| Harmless medicine for human | $64.1K | 79 |
| We intend to claim rewards under | $61.3K | 88 |
| Sodium chloride intravenous infusion | $60.9K | 3 |
| Varenismart 1 (varenicline tartrate 1MG)(1x10 tabs | $60.3K | 2 |
| Sevoflurane 250ML | $58.4K | 2 |
| Ayurvedic and herbal products for | $53.7K | 108 |
| Tenof 300MG | $52.2K | 2 |
| Cefixime 400 MG tablet (10 x 10 tabs) | $50.0K | 1 |
| Pharmaceutical finished formulation | $43.0K | 3 |
TO THE Manager- imports 5,721 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does TO THE Manager- Import?
Top Products by Import Value
TO THE Manager- Therapeutic Categories — 2 Specializations
TO THE Manager- imports across 2 therapeutic categories, with Advanced Diabetes Medications (73.9%), Advanced Oncology (26.1%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Diabetes Medications
1 products · 73.9% · $69.9K
Advanced Oncology
3 products · 26.1% · $24.7K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Semaglutide | Advanced Diabetes Medications | $69.9K | 24 | 1.4% | 14 |
| 2 | Tofacitinib | Advanced Oncology | $13.3K | 7 | 0.3% | 11 |
| 3 | Actinomycin | Advanced Oncology | $8.1K | 2 | 0.3% | 8 |
| 4 | Chlorambucil | Advanced Oncology | $3.2K | 5 | 0.9% | 10 |
TO THE Manager- imports 4 pharmaceutical products across 2 categories into Brazil totaling $94.6K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for TO THE Manager-.
Request DemoTO THE Manager- — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
TO THE Manager- is a pharmaceutical importer and buyer based in Brazil, specializing in the acquisition of finished pharmaceutical formulations from international suppliers, particularly from India. The company plays a pivotal role in Brazil's pharmaceutical distribution network by sourcing a diverse range of medications to meet the country's healthcare needs. With a total import value of $95,000 USD over 38 shipments from India between 2022 and 2026, TO THE Manager- demonstrates a consistent engagement in the international pharmaceutical trade.
The company's portfolio is concentrated in two therapeutic categories: Advanced Diabetes Medications and Advanced Oncology, reflecting a strategic focus on high-demand areas within Brazil's healthcare sector. Notably, the top five imported products—Semaglutide, Tofacitinib, Actinomycin, and Chlorambucil—account for 100% of the company's imports, indicating a highly specialized sourcing strategy. This concentration suggests that TO THE Manager- may serve as a primary wholesaler or distributor, facilitating the availability of these critical medications across Brazil.
2Distribution Network
While specific details about TO THE Manager-'s warehouse locations and logistics capabilities are not publicly available, the company's import activities indicate a well-established distribution network within Brazil. The consistent importation of pharmaceutical products from India over a four-year period suggests efficient logistics and warehousing operations capable of handling international shipments. Given the therapeutic focus on diabetes and oncology medications, it is likely that TO THE Manager- collaborates with hospitals, clinics, and pharmacies nationwide to ensure the timely delivery of these essential drugs.
3Industry Role
TO THE Manager- occupies a significant position in Brazil's pharmaceutical supply chain, likely functioning as a primary wholesaler or distributor. The company's specialized focus on high-demand therapeutic areas, such as Advanced Diabetes Medications and Advanced Oncology, positions it as a key player in ensuring the availability of critical medications. By importing finished pharmaceutical formulations from India, TO THE Manager- contributes to the diversification and stability of Brazil's pharmaceutical market, catering to the specific needs of healthcare providers and patients.
Supplier Relationship Intelligence — TO THE Manager-
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The sourcing strategy of TO THE Manager- exhibits a high degree of concentration, with the top five imported products accounting for 100% of the company's imports. This indicates a deliberate focus on a narrow range of medications, which could be a strategic choice to specialize in high-demand therapeutic areas. However, such concentration also presents potential risks, including supply chain disruptions or regulatory changes affecting these specific products. The company's reliance on a single source for these medications may expose it to vulnerabilities if any issues arise with the supplier or the products themselves.
2Supply Chain Resilience
TO THE Manager-'s supply chain resilience appears to be robust, given the consistent importation of pharmaceutical products from India over a four-year period. The company's engagement with multiple Indian suppliers, including GMSANJIVANI LLP, HEALING PHARMA INDIA PRIVATE LIMITED, and ACCELIUS GLOBAL, suggests a diversified sourcing strategy that can mitigate risks associated with single-source dependency. Additionally, the importation of 5,721 unique pharmaceutical formulations indicates a broad product base, which can enhance supply chain flexibility. However, the company's focus on specific therapeutic areas may limit its ability to adapt to sudden shifts in market demand or regulatory changes affecting these categories.
3Strategic Implications
The concentrated sourcing pattern of TO THE Manager- positions it as a specialized distributor in Brazil's pharmaceutical market, catering to specific therapeutic needs. This specialization can enhance the company's competitive edge by establishing expertise and strong relationships within these niches. For Indian exporters, understanding TO THE Manager-'s focus on Advanced Diabetes Medications and Advanced Oncology presents an opportunity to align their product offerings with the company's needs, potentially leading to a fruitful partnership. However, Indian exporters should be aware of the risks associated with the company's concentrated sourcing strategy and consider diversifying their product portfolios to appeal to a broader range of importers.
Importing Pharmaceuticals into Brazil — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Brazil
1Regulatory Authority & Framework
In Brazil, the regulation of pharmaceutical products is overseen by the Agência Nacional de Vigilância Sanitária (ANVISA), the Brazilian Health Regulatory Agency. ANVISA is responsible for ensuring the safety, efficacy, and quality of health-related products, including drugs and biological products. The agency's regulatory framework encompasses the authorization of clinical trials, marketing authorizations for drugs, and post-marketing surveillance. For foreign companies, including Indian exporters, it is mandatory to designate a legally established partner in Brazil to handle administrative procedures for obtaining pre-market approvals with ANVISA. This partner assumes legal responsibility for the products imported and distributed within Brazilian territory. (gov.br)
2Import Licensing & GMP
Importing pharmaceutical products into Brazil requires compliance with ANVISA's regulations, including obtaining the necessary import licenses and ensuring that products meet Good Manufacturing Practices (GMP). ANVISA's Resolution RDC 658/2022 outlines the general GMP guidelines for medicines, aligning with the Pharmaceutical Inspection Co-operation Scheme (PIC/S) standards. Foreign companies must collaborate with a local partner in Brazil to navigate the import licensing process and ensure that their products adhere to ANVISA's GMP requirements. This partnership is essential for the successful importation and commercialization of pharmaceutical products in the Brazilian market. (gov.br)
3Quality & Labeling
Pharmaceutical products imported into Brazil must comply with ANVISA's quality and labeling standards. This includes batch testing, stability requirements, and labeling in Portuguese. ANVISA's regulations stipulate that labeling must provide clear and accurate information about the product, including dosage, administration instructions, and any potential side effects. Additionally, serialization mandates are in place to ensure traceability and prevent counterfeit products from entering the market. Compliance with these requirements is crucial for the legal sale and distribution of pharmaceutical products in Brazil. (softgroup.eu)
4Recent Regulatory Changes
Between 2024 and 2026, Brazil implemented several regulatory changes affecting pharmaceutical imports. Notably, ANVISA's Resolution RDC 888/2024 introduced new guidelines for over-the-counter (OTC) drugs, impacting labeling and marketing practices. Additionally, the National Drug Control System (SNCM) established serialization requirements and timelines for the pharmaceutical supply chain, enhancing product traceability and combating counterfeit drugs. These changes necessitate that importers and distributors, including TO THE Manager-, adapt their operations to remain compliant with Brazil's evolving regulatory landscape. (softgroup.eu)
TO THE Manager- — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
TO THE Manager-'s focus on Advanced Diabetes Medications and Advanced Oncology reflects a strategic alignment with Brazil's healthcare priorities. The high prevalence of diabetes and cancer in the country drives significant demand for effective treatments in these therapeutic areas. By specializing in these categories, TO THE Manager- positions itself to meet critical healthcare needs, ensuring the availability of essential medications for patients and healthcare providers. This targeted approach also allows the company to build expertise and strong relationships within these niches, enhancing its competitive position in the Brazilian pharmaceutical market.
2Sourcing Profile
TO THE Manager- employs a sourcing strategy that focuses on finished pharmaceutical formulations, particularly from India. This approach allows the company to leverage India's robust pharmaceutical manufacturing capabilities and cost advantages. By importing finished products, TO THE Manager- can offer a diverse range of medications without the complexities associated with manufacturing. This strategy aligns with the company's focus on high-demand therapeutic areas, ensuring a steady supply of essential drugs to the Brazilian market.
3Market Positioning
Based on its product mix, TO THE Manager- serves the wholesale distribution segment of the Brazilian pharmaceutical market. The company's specialization in Advanced Diabetes Medications and Advanced Oncology positions it as a key supplier to hospitals, clinics, and pharmacies that require these critical treatments. By focusing on these therapeutic areas, TO THE Manager- addresses specific market needs, contributing to the overall healthcare infrastructure in Brazil.
Seller's Guide — How to Become a Supplier to TO THE Manager-
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with TO THE Manager-. The company's focus on Advanced Diabetes Medications and Advanced Oncology presents a niche market where specialized products are in demand. Indian exporters with expertise in these therapeutic areas can align their offerings with TO THE Manager-'s needs, potentially leading to a fruitful partnership. However, new suppliers should be prepared to meet ANVISA's regulatory requirements, including GMP compliance and product registration, to successfully enter the Brazilian market.
2Requirements & Qualifications
Indian exporters seeking to supply TO THE Manager- and enter the Brazilian market must ensure that their products comply with ANVISA's regulations. This includes obtaining the necessary import licenses, ensuring Good Manufacturing Practices (GMP) compliance, and registering products with ANVISA. Additionally, labeling must be in Portuguese and meet ANVISA's standards. Collaboration with a local partner in Brazil is essential to navigate the regulatory landscape and facilitate the importation process. (gov.br)
3How to Approach
To establish a relationship with TO THE Manager-, Indian exporters should identify and engage with the company's procurement or purchasing department. Presenting a comprehensive portfolio of products that align with
Frequently Asked Questions — TO THE Manager-
What products does TO THE Manager- import from India?
TO THE Manager- imports 4 pharmaceutical products across 2 categories. Top imports: Semaglutide ($69.9K), Tofacitinib ($13.3K), Actinomycin ($8.1K), Chlorambucil ($3.2K).
Who supplies pharmaceuticals to TO THE Manager- from India?
TO THE Manager- sources from 45 verified Indian suppliers. The primary supplier is Gmsanjivani Llp (32.3% of imports, $2.7M).
What is TO THE Manager-'s total pharmaceutical import value?
TO THE Manager-'s total pharmaceutical import value from India is $94.6K, based on 38 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does TO THE Manager- focus on?
TO THE Manager- imports across 2 categories. The largest: Advanced Diabetes Medications (73.9%), Advanced Oncology (26.1%).
Get Full TO THE Manager- Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: TO THE Manager- identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as TO THE Manager-'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 38 individual customs records matching TO THE Manager-.
- 5.Supplier Verification: TO THE Manager- sources from 45 verified Indian suppliers across 5,721 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.