TO THE Director OF
Pharmaceutical Importer · Sudan · Advanced Antibiotics Focus · $4.1M Total Trade · DGFT Verified
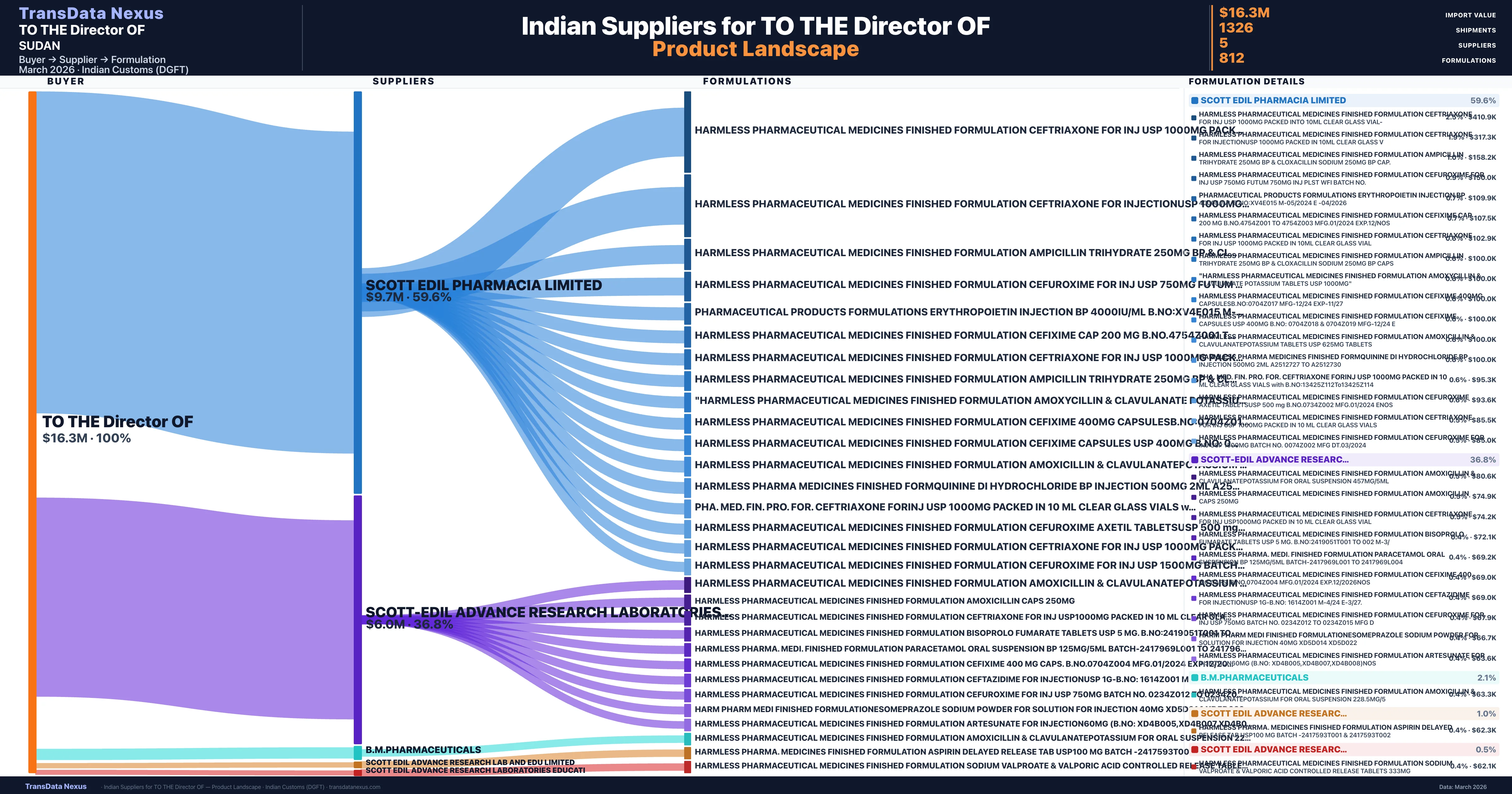
TO THE Director OF is a pharmaceutical importer based in Sudan with a total trade value of $4.1M across 3 products in 1 therapeutic categories. Based on 175 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. TO THE Director OF sources from 5 verified Indian suppliers, with Scott Edil Pharmacia Limited accounting for 59.6% of imports.
TO THE Director OF — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to TO THE Director OF?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Scott Edil Pharmacia Limited | $9.7M | 1,003 | 59.6% |
| Scott-edil Advance Research Laboratories & Educati | $6.0M | 266 | 36.8% |
| B.m.pharmaceuticals | $341.6K | 49 | 2.1% |
| Scott Edil Advance Research Lab And Edu Limited | $161.6K | 4 | 1.0% |
| Scott Edil Advance Research Laboratories Educati | $82.0K | 4 | 0.5% |
TO THE Director OF sources from 5 verified Indian suppliers across 812 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does TO THE Director OF Import?
| Formulation | Value | Ships |
|---|---|---|
| Harmless pharmaceutical medicines finished formulation ceftriaxone for INJ USP 1000MG packed into 10ML clear glass | $410.9K | 16 |
| Harmless pharmaceutical medicines finished formulation ceftriaxone for injectionusp 1000MG packed in 10ML clear glass | $317.3K | 18 |
| Harmless pharmaceutical medicines finished formulation ampicillin trihydrate 250MG BP & cloxacillin sodium 250MG BP cap. | $158.2K | 5 |
| Harmless pharmaceutical medicines finished formulation cefuroxime for INJ USP 750MG futum 750MG INJ plst wfi batch no. | $150.0K | 3 |
| Pharmaceutical products formulations erythropoietin injection BP 4000iu/ML b.no:xv4e015 m-05/2024 e | $109.9K | 4 |
| Harmless pharmaceutical medicines finished formulation cefixime cap 200 MG b.no.4754z001 to 4754z003 mfg.01/2024 | $107.5K | 3 |
| Harmless pharmaceutical medicines finished formulation ceftriaxone for INJ USP 1000MG packed in 10ML clear glass | $102.9K | 3 |
| Harmless pharmaceutical medicines finished formulation ampicillin trihydrate 250MG BP & cloxacillin sodium 250MG BP CAPS | $100.0K | 2 |
| "harmless pharmaceutical medicines finished formulation amoxycillin & clavulanate potassium tablets USP | $100.0K | 2 |
| Harmless pharmaceutical medicines finished formulation cefixime 400MG capsulesb.no:0704z017 mfg-12/24 | $100.0K | 2 |
| Harmless pharmaceutical medicines finished formulation cefixime capsules USP 400MG b.no: 0704z018 & 0704z019 mfg-12/24 | $100.0K | 2 |
| Harmless pharmaceutical medicines finished formulation amoxicillin & clavulanatepotassium tablets USP 625MG | $100.0K | 2 |
| Harmless pharma medicines finished formquinine di hydrochloride BP injection 500MG 2ML a2512727 to | $100.0K | 2 |
| Pha. med. fin. pro. for. ceftriaxone forinj USP 1000MG packed in 10 ML clear glass vials with | $95.3K | 2 |
| Harmless pharmaceutical medicines finished formulation cefuroxime axetil tabletsusp 500 MG b.no.0734z002 mfg.01/2024 | $93.6K | 3 |
TO THE Director OF imports 812 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does TO THE Director OF Import?
Top Products by Import Value
TO THE Director OF Therapeutic Categories — 1 Specializations
TO THE Director OF imports across 1 therapeutic categories, with Advanced Antibiotics (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
3 products · 100.0% · $4.1M
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ceftriaxone | Advanced Antibiotics | $2.3M | 97 | 0.8% | 9 |
| 2 | Cefixime | Advanced Antibiotics | $1.7M | 75 | 1.0% | 12 |
| 3 | Ceftazidime | Advanced Antibiotics | $100.5K | 3 | 0.9% | 12 |
TO THE Director OF imports 3 pharmaceutical products across 1 categories into Sudan totaling $4.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for TO THE Director OF.
Request DemoTO THE Director OF — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
TO THE Director OF is a pharmaceutical importer and buyer based in Sudan, specializing in the procurement of finished pharmaceutical formulations from India. The company has established itself as a significant player in Sudan's pharmaceutical supply chain, focusing on advanced antibiotics. With a total import value of $4.1 million USD over 175 shipments, TO THE Director OF demonstrates a robust engagement in the importation of critical medical supplies. The company's portfolio is highly concentrated, with its top three products—Ceftriaxone, Cefixime, and Ceftazidime—accounting for 100% of its imports. This strategic focus underscores TO THE Director OF's commitment to providing essential antibiotics to meet the healthcare needs of the Sudanese population.
2Distribution Network
While specific details about TO THE Director OF's warehouse locations and logistics capabilities are not publicly available, the company's substantial import volume suggests a well-established distribution network within Sudan. The efficient handling of 175 shipments indicates a capacity to manage complex logistics operations, ensuring timely delivery of pharmaceutical products across the country. Given the critical nature of the imported antibiotics, it is likely that TO THE Director OF collaborates with local distributors and healthcare facilities to ensure widespread availability of its products.
3Industry Role
TO THE Director OF plays a pivotal role in Sudan's pharmaceutical supply chain as a primary importer of essential antibiotics. By focusing exclusively on advanced antibiotics, the company addresses a critical segment of the market, ensuring the availability of these life-saving medications. Its significant import volume and product concentration position TO THE Director OF as a key supplier to hospitals, clinics, and pharmacies throughout Sudan, contributing to the overall healthcare infrastructure.
Supplier Relationship Intelligence — TO THE Director OF
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The sourcing strategy of TO THE Director OF reveals a high degree of supplier concentration, with five verified Indian suppliers accounting for the entirety of its imports. This single-source dependency could pose risks related to supply chain disruptions, such as production delays or geopolitical issues affecting trade relations. However, the consistent import volumes from these suppliers over multiple years suggest a stable and reliable relationship, mitigating potential risks associated with such concentration. The company's focus on a limited number of suppliers may also allow for more favorable terms and streamlined logistics.
2Supply Chain Resilience
TO THE Director OF's supply chain resilience appears robust, given the consistent importation of a diverse range of 812 unique pharmaceutical formulations from its five Indian suppliers. This diversity in formulations indicates a strategic approach to sourcing, reducing dependency on any single product line and enhancing the company's ability to adapt to changing market demands. While the concentration of suppliers could be a vulnerability, the established relationships and diversified product range suggest a well-prepared supply chain capable of maintaining stability in the face of potential disruptions.
3Strategic Implications
The concentrated sourcing pattern of TO THE Director OF positions the company as a significant partner for its Indian suppliers, potentially leading to preferential treatment and better pricing. For Indian exporters, this presents an opportunity to strengthen partnerships with a key importer in the Sudanese market. Expanding the product range offered to TO THE Director OF could lead to increased market share in Sudan, capitalizing on the company's established distribution network and reputation.
Importing Pharmaceuticals into Sudan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Sudan
1Regulatory Authority & Framework
In Sudan, the National Medicines and Poisons Board (NMPB) serves as the primary regulatory authority overseeing the importation, manufacturing, and distribution of pharmaceutical products. Established under the Drugs and Toxins Act of 2009, the NMPB is responsible for ensuring the safety, efficacy, and quality of medicines available in the country. The NMPB's mandate includes the registration of pharmaceutical products, inspection of manufacturing facilities, and enforcement of regulations to prevent the circulation of counterfeit or substandard medicines. The NMPB collaborates with international organizations, such as the World Health Organization (WHO), to align Sudan's pharmaceutical regulations with global standards. (emro.who.int)
2Import Licensing & GMP
Importers in Sudan must obtain a license from the NMPB to legally import pharmaceutical products. This process involves submitting an application that includes detailed information about the products, their manufacturers, and compliance with Good Manufacturing Practice (GMP) standards. The NMPB requires that foreign manufacturers provide a Certificate of Pharmaceutical Product (CPP) issued by the health authority in the exporting country, authenticated by the Sudanese Ministry of Foreign Affairs. This certificate must detail the product's composition and confirm that it is registered and freely sold in the country of origin. Additionally, the NMPB conducts inspections of manufacturing facilities to ensure adherence to GMP guidelines, with overseas manufacturers typically inspected every five years. (nmpb.gov.sd)
3Quality & Labeling
Pharmaceutical products imported into Sudan must undergo testing and inspection by the Sudanese Standardization and Metrology Organization (SSMO) before entry into the country. This process ensures that all imported medicines meet the required safety and quality standards. The NMPB mandates that all pharmaceutical products be registered before marketing, and that labeling includes information in English, detailing the product's composition, dosage, and usage instructions. Additionally, the NMPB requires that the manufacturing plants be registered within the board, and that the pharmaceutical product with the same specification be registered and freely sold in the country of origin. (nmpb.gov.sd)
4Recent Regulatory Changes
In August 2018, the Federal Ministry of Health, the Federal Humanitarian Aid Commission, and the WHO collaborated to update Sudan's medicines purchase and import regulations. This initiative aimed to streamline procedures for the clearance and transfer of medicines through the National Medical Supplies Fund, as well as to establish certified common purchasing windows. The updated regulations were designed to enhance the efficiency and transparency of the pharmaceutical supply chain, ensuring that both national and international organizations operating in Sudan adhere to standardized procedures for importing and distributing medical supplies. (emro.who.int)
TO THE Director OF — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
TO THE Director OF's exclusive focus on advanced antibiotics—Ceftriaxone, Cefixime, and Ceftazidime—aligns with the critical healthcare needs in Sudan, where antibiotic resistance and infectious diseases are prevalent concerns. The substantial import values of these products indicate a strong market demand and a strategic decision to concentrate on high-impact therapeutic areas. By specializing in these antibiotics, TO THE Director OF positions itself as a key supplier of essential medications, contributing significantly to the treatment of bacterial infections in the country.
2Sourcing Profile
TO THE Director OF's sourcing strategy is centered on importing finished pharmaceutical formulations from India, a country known for its robust pharmaceutical manufacturing sector. The company's focus on advanced antibiotics suggests a preference for high-quality, effective treatments that meet international standards. India's adherence to WHO-GMP guidelines and its established reputation in the global pharmaceutical market make it a reliable source for these critical medications. This sourcing approach ensures that TO THE Director OF can provide Sudanese healthcare providers with essential antibiotics that are both effective and accessible.
3Market Positioning
By concentrating on advanced antibiotics, TO THE Director OF serves a vital segment of the Sudanese pharmaceutical market, primarily supplying hospitals, clinics, and pharmacies with essential medications for treating bacterial infections. The company's significant import volumes and product specialization position it as a key player in the healthcare sector, addressing critical therapeutic needs and contributing to the overall health infrastructure of Sudan.
Seller's Guide — How to Become a Supplier to TO THE Director OF
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter the Sudanese market by partnering with TO THE Director OF. The company's established distribution network and focus on advanced antibiotics present a platform for introducing additional products that meet the therapeutic needs of the Sudanese population. Potential gaps in TO THE Director OF's current sourcing, such as the inclusion of other essential antibiotics or related therapeutic categories, could be addressed by new suppliers, thereby expanding the product portfolio and enhancing market presence.
2Requirements & Qualifications
Indian exporters seeking to supply TO THE Director OF and the Sudanese market must ensure that their products are registered with the NMPB and comply with Sudanese regulations. This includes providing a Certificate of Pharmaceutical Product (CPP) issued by the health authority in the exporting country, authenticated by the Sudanese Ministry of Foreign Affairs. Additionally, manufacturers must adhere to Good Manufacturing Practice (GMP) standards, with inspections conducted by the NMPB to verify compliance. Labeling must be in English, detailing the product's composition, dosage, and usage instructions. (nmpb.gov.sd)
3How to Approach
To establish a partnership with TO THE Director OF, Indian exporters should initiate contact by providing detailed product information, including compliance with NMPB regulations and GMP standards. Engaging in discussions about the specific needs of the Sudanese market and how their products can meet these requirements will be crucial. Participating in relevant tenders and aligning with the regulatory filing strategy of TO THE Director OF will facilitate the integration of new products into the company's portfolio. Given the regulatory processes involved, a timeline of several months may be expected for product registration and approval.
Frequently Asked Questions — TO THE Director OF
What products does TO THE Director OF import from India?
TO THE Director OF imports 3 pharmaceutical products across 1 categories. Top imports: Ceftriaxone ($2.3M), Cefixime ($1.7M), Ceftazidime ($100.5K).
Who supplies pharmaceuticals to TO THE Director OF from India?
TO THE Director OF sources from 5 verified Indian suppliers. The primary supplier is Scott Edil Pharmacia Limited (59.6% of imports, $9.7M).
What is TO THE Director OF's total pharmaceutical import value?
TO THE Director OF's total pharmaceutical import value from India is $4.1M, based on 175 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does TO THE Director OF focus on?
TO THE Director OF imports across 1 categories. The largest: Advanced Antibiotics (100.0%).
Get Full TO THE Director OF Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: TO THE Director OF identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as TO THE Director OF's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 175 individual customs records matching TO THE Director OF.
- 5.Supplier Verification: TO THE Director OF sources from 5 verified Indian suppliers across 812 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.