Tillomed Laboratories Limited
Pharmaceutical Importer · United Kingdom · Immunosuppressants Focus · $16.6M Total Trade · DGFT Verified
Tillomed Laboratories Limited is a pharmaceutical importer based in United Kingdom with a total trade value of $16.6M across 13 products in 6 therapeutic categories. Based on 487 verified import shipments from Indian Customs (DGFT) records, Tillomed Laboratories Limited is the #1 buyer in 2 products including Amphotericin, Carmustine. Tillomed Laboratories Limited sources from 11 verified Indian suppliers, with Emcure Pharmaceuticals Limited accounting for 58.9% of imports.
Tillomed Laboratories Limited — Import Portfolio & Supplier Network
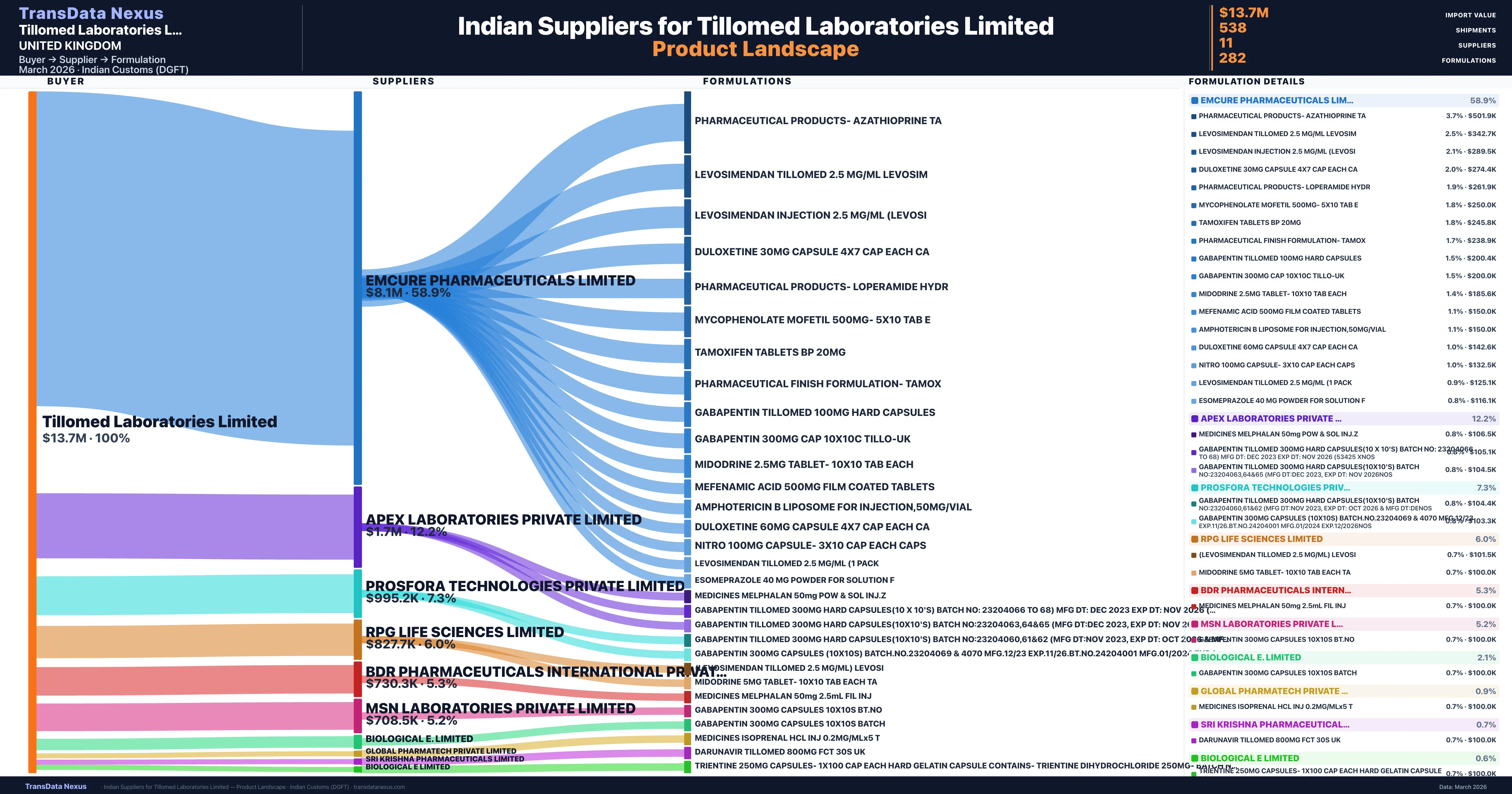
Who Are the Verified Indian Suppliers to Tillomed Laboratories Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Emcure Pharmaceuticals Limited | $8.1M | 368 | 58.9% |
| Apex Laboratories Private Limited | $1.7M | 39 | 12.2% |
| Prosfora Technologies Private Limited | $995.2K | 47 | 7.3% |
| Rpg Life Sciences Limited | $827.7K | 25 | 6.0% |
| Bdr Pharmaceuticals International Private Limited | $730.3K | 25 | 5.3% |
| Msn Laboratories Private Limited | $708.5K | 17 | 5.2% |
| Biological E. Limited | $291.4K | 6 | 2.1% |
| Global Pharmatech Private Limited | $127.7K | 4 | 0.9% |
| Sri Krishna Pharmaceuticals Limited | $102.0K | 3 | 0.7% |
| Biological E Limited | $86.5K | 2 | 0.6% |
| Immacule Lifesciences Private Limited | $83.0K | 2 | 0.6% |
Tillomed Laboratories Limited sources from 11 verified Indian suppliers across 282 distinct formulations. The supply base is diversified across 11 suppliers, reducing single-source dependency risk.
What Formulations Does Tillomed Laboratories Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Pharmaceutical products- azathioprine | $501.9K | 12 |
| Levosimendan tillomed 2.5 MG/ML | $342.7K | 10 |
| Levosimendan injection 2.5 MG/ML | $289.5K | 6 |
| Duloxetine 30MG capsule 4x7 cap Each | $274.4K | 10 |
| Pharmaceutical products- loperamide | $261.9K | 6 |
| Mycophenolate mofetil 500MG- 5x10 TAB | $250.0K | 5 |
| Tamoxifen tablets BP 20MG | $245.8K | 7 |
| Pharmaceutical finish formulation- | $238.9K | 7 |
| Gabapentin tillomed 100MG hard | $200.4K | 6 |
| Gabapentin 300MG cap 10x10c | $200.0K | 4 |
| Midodrine 2.5MG tablet- 10x10 TAB | $185.6K | 5 |
| Mefenamic acid 500MG film coated | $150.0K | 3 |
| Amphotericin b liposome for injection | $150.0K | 5 |
| Duloxetine 60MG capsule 4x7 cap Each | $142.6K | 6 |
| Nitro 100MG capsule- 3x10 cap Each CAPS | $132.5K | 4 |
Tillomed Laboratories Limited imports 282 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Tillomed Laboratories Limited Import?
Tillomed Laboratories Limited Therapeutic Categories — 6 Specializations
Tillomed Laboratories Limited imports across 6 therapeutic categories, with Immunosuppressants (42.3%), Advanced Oncology (31.0%), Antifungals (14.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 77% of total imports.
Immunosuppressants
3 products · 42.3% · $7.0M
Advanced Oncology
5 products · 31.0% · $5.1M
Antifungals
1 products · 14.8% · $2.5M
Advanced Antibiotics
2 products · 7.2% · $1.2M
Gastrointestinal
1 products · 3.0% · $489.8K
Antimalarial & Antiparasitic
1 products · 1.8% · $291.0K
Import Portfolio — Top 13 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Mycophenolate | Immunosuppressants | $4.6M | 109 | 0.7% | 10 |
| 2 | Melphalan | Advanced Oncology | $2.7M | 78 | 12.2% | 2 |
| 3 | Amphotericin | Antifungals | $2.5M | 49 | 0.7% | 1 |
| 4 | Azathioprine | Immunosuppressants | $2.0M | 43 | 2.1% | 8 |
| 5 | Busulfan | Advanced Oncology | $1.0M | 47 | 12.6% | 2 |
| 6 | Carmustine | Advanced Oncology | $900.0K | 18 | 3.6% | 1 |
| 7 | Nitrofurantoin | Advanced Antibiotics | $646.0K | 25 | 1.9% | 10 |
| 8 | Amikacin | Advanced Antibiotics | $550.0K | 11 | 2.6% | 4 |
| 9 | Loperamide | Gastrointestinal | $489.8K | 28 | 0.6% | 14 |
| 10 | Leflunomide | Immunosuppressants | $466.4K | 48 | 1.7% | 13 |
| 11 | Tamoxifen | Advanced Oncology | $404.1K | 14 | 2.7% | 7 |
| 12 | Dapsone | Antimalarial & Antiparasitic | $291.0K | 15 | 0.9% | 11 |
| 13 | Cabazitaxel | Advanced Oncology | $100.0K | 2 | 1.1% | 12 |
Tillomed Laboratories Limited imports 13 pharmaceutical products across 6 categories into United Kingdom totaling $16.6M. The company is the #1 buyer for 2 products: Amphotericin, Carmustine.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Tillomed Laboratories Limited.
Request DemoTillomed Laboratories Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Tillomed Laboratories Limited is a UK-based pharmaceutical company specializing in the licensing, marketing, and supply of generic and branded pharmaceutical products. Established on 28 September 1990, the company is headquartered at 220 Butterfield, Great Marlings, Luton, Bedfordshire, LU2 8DL, England. Tillomed operates as a private limited company and is a subsidiary of Emcure Pharmaceuticals, a leading Indian pharmaceutical firm with a global presence.
The company focuses on providing cost-effective and timely access to a comprehensive range of medicines across various therapeutic areas. Tillomed's operations encompass the entire supply chain, from sourcing and importing pharmaceutical products to distribution within the United Kingdom. This integrated approach enables the company to meet the needs of hospitals, wholesalers, and pharmacists nationwide.
2Distribution Network
Tillomed Laboratories Limited's distribution network is strategically designed to ensure efficient delivery of pharmaceutical products across the United Kingdom. While specific warehouse locations are not publicly disclosed, the company's headquarters in Luton serves as a central hub for its operations. The logistics capabilities are tailored to handle a diverse portfolio of pharmaceutical products, ensuring compliance with regulatory standards and timely distribution. The geographic coverage primarily focuses on the UK market, with potential expansion into other European regions, leveraging the company's subsidiaries in France, Spain, Germany, Italy, and the Czech Republic.
3Industry Role
In the United Kingdom's pharmaceutical supply chain, Tillomed Laboratories Limited functions as a primary wholesaler and distributor. The company imports a wide array of pharmaceutical products, including both generic and branded medicines, and supplies them to hospitals, wholesalers, and pharmacists. This role positions Tillomed as a critical intermediary between manufacturers and end-users, ensuring the availability of essential medicines throughout the UK. The company's operations are characterized by a focus on cost-effectiveness and timely delivery, aligning with the needs of the healthcare sector.
Supplier Relationship Intelligence — Tillomed Laboratories Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Tillomed Laboratories Limited's sourcing strategy exhibits a significant concentration on a select group of suppliers, particularly from India. The company's total import value from India amounts to $16.6 million USD, distributed across 487 shipments and 13 products spanning six therapeutic categories. The top five imported products—Mycophenolate, Melphalan, Amphotericin, Azathioprine, and Busulfan—collectively account for 76.8% of the total import value. This concentration indicates a strategic focus on specific high-demand products, potentially leveraging favorable pricing and supply chain efficiencies.
The reliance on a limited number of suppliers, such as Emcure Pharmaceuticals Limited, which accounts for 58.9% of the total import value, suggests a strong partnership and possibly favorable contractual terms. However, this dependency also introduces risks related to supply chain disruptions, regulatory changes, or geopolitical factors affecting the supplier's operations. Diversifying the supplier base could mitigate these risks and enhance supply chain resilience.
2Supply Chain Resilience
Tillomed Laboratories Limited's supply chain resilience is influenced by its sourcing practices, particularly the heavy reliance on a few key suppliers from India. While this strategy may offer cost advantages and streamlined logistics, it also exposes the company to potential disruptions. The absence of publicly available information regarding backup suppliers and the diversity of formulations imported indicates a need for strategic planning to enhance supply chain robustness. Engaging with multiple suppliers and diversifying the product portfolio could strengthen the company's ability to withstand supply chain challenges.
3Strategic Implications
The current sourcing pattern of Tillomed Laboratories Limited, characterized by a concentrated supplier base and a focus on specific therapeutic areas, positions the company to leverage cost efficiencies and strong supplier relationships. However, this strategy may limit flexibility and responsiveness to market changes. For Indian exporters, understanding Tillomed's product preferences and therapeutic focus areas presents an opportunity to align offerings with the company's needs. Proposing alternative suppliers or products within these categories could enhance competitiveness and appeal to Tillomed's procurement strategy.
Importing Pharmaceuticals into United Kingdom — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Kingdom
1Regulatory Authority & Framework
The Medicines and Healthcare products Regulatory Agency (MHRA) is the primary regulatory authority overseeing pharmaceutical imports into the United Kingdom. MHRA is responsible for ensuring that medicines meet appropriate standards of safety, quality, and efficacy. Key legislation governing pharmaceutical imports includes the Medicines for Human Use (Clinical Trials) Regulations 2004 and the Medicines Act 1968. These regulations outline requirements for marketing authorizations, manufacturing licenses, and importation procedures. (cms.mhra.gov.uk)
The marketing authorization pathway for Indian generics involves obtaining a Marketing Authorization (MA) license from MHRA. This process includes submitting comprehensive data on the product's quality, safety, and efficacy. Additionally, a Qualified Person (QP) must certify that the product has been manufactured and tested according to the MA and Good Manufacturing Practice (GMP) standards. MHRA conducts inspections of manufacturing sites to ensure compliance with GMP requirements. (gov.uk)
2Import Licensing & GMP
To import human medicines into the UK, companies must obtain a Wholesale Distribution License (WDA(H)) from MHRA. This license authorizes the holder to import, store, and distribute medicines within the UK. For importing unlicensed medicines, a Manufacturer's "Specials" License (MSL) is required. The manufacturing site must hold a valid GMP certificate, which can be from EU GMP, WHO GMP, or PIC/S standards. MHRA recognizes GMP certificates from countries with equivalent standards, facilitating the importation of medicines from approved countries. (gov.uk)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure compliance with UK standards. Stability requirements are set to guarantee that medicines maintain their quality throughout their shelf life. Labeling must be in English and include essential information such as the product name, dosage form, strength, batch number, and expiry date. Serialization mandates are in place to prevent counterfeit medicines, requiring unique identifiers on packaging to track and trace products throughout the supply chain. (gov.uk)
4Recent Regulatory Changes
In January 2025, the MHRA implemented new UK-wide licensing arrangements for human medicines as part of the Windsor Framework. These changes affect labeling, packaging, and importing requirements, including the use of 'UK Only' labels and the disapplication of the EU Falsified Medicines Directive (FMD). Specific instructions are provided for Qualified Persons (QPs) and Responsible Persons for Import (RPis).
Tillomed Laboratories Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Tillomed Laboratories Limited's product strategy focuses on specific therapeutic areas, including immunosuppressants, advanced oncology, and antifungals. The company's imports from India reflect this focus, with the top five products—Mycophenolate, Melphalan, Amphotericin, Azathioprine, and Busulfan—collectively accounting for 76.8% of the total import value. This strategy aligns with market demand for specialized treatments in these areas, catering to the needs of hospitals and healthcare providers in the UK.
2Sourcing Profile
Tillomed Laboratories Limited's sourcing strategy emphasizes generic drug procurement, particularly from India, which is known for its robust pharmaceutical manufacturing capabilities. The company's focus on finished pharmaceutical formulations, such as tablets, capsules, syrups, and injections, indicates a preference for ready-to-use products that meet regulatory standards. India's compliance with international GMP standards and its cost-effective manufacturing processes make it a strategic partner for Tillomed's procurement needs.
3Market Positioning
Based on its product mix, Tillomed Laboratories Limited serves the hospital and wholesale distribution segments of the UK market. The company's focus on specialized therapeutic areas and its role as a primary wholesaler position it to meet the needs of healthcare providers requiring cost-effective and timely access to essential medicines. This market positioning aligns with the company's mission to deliver effective, high-quality, and sustainable healthcare solutions.
Seller's Guide — How to Become a Supplier to Tillomed Laboratories Limited
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Tillomed Laboratories Limited, particularly in the therapeutic areas of immunosuppressants, advanced oncology, and antifungals. The company's concentrated sourcing pattern suggests potential gaps in its supplier base that could be addressed by offering alternative products within these categories. Establishing partnerships with Tillomed could enhance supply chain diversity and resilience, benefiting both parties.
2Requirements & Qualifications
Indian exporters seeking to supply Tillomed Laboratories Limited and the UK market must ensure compliance with MHRA's regulatory requirements. This includes obtaining appropriate marketing authorizations, ensuring manufacturing sites hold valid GMP certificates recognized by MHRA, and adhering to labeling and packaging standards. Additionally, products must meet quality standards, including batch testing and stability requirements, to ensure safety and efficacy
Frequently Asked Questions — Tillomed Laboratories Limited
What products does Tillomed Laboratories Limited import from India?
Tillomed Laboratories Limited imports 13 pharmaceutical products across 6 categories. Top imports: Mycophenolate ($4.6M), Melphalan ($2.7M), Amphotericin ($2.5M), Azathioprine ($2.0M), Busulfan ($1.0M).
Who supplies pharmaceuticals to Tillomed Laboratories Limited from India?
Tillomed Laboratories Limited sources from 11 verified Indian suppliers. The primary supplier is Emcure Pharmaceuticals Limited (58.9% of imports, $8.1M).
What is Tillomed Laboratories Limited's total pharmaceutical import value?
Tillomed Laboratories Limited's total pharmaceutical import value from India is $16.6M, based on 487 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Tillomed Laboratories Limited focus on?
Tillomed Laboratories Limited imports across 6 categories. The largest: Immunosuppressants (42.3%), Advanced Oncology (31.0%), Antifungals (14.8%).
Get Full Tillomed Laboratories Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Tillomed Laboratories Limited identified across shipments using consignee name normalization, aggregating 6 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Tillomed Laboratories Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 487 individual customs records matching Tillomed Laboratories Limited.
- 5.Supplier Verification: Tillomed Laboratories Limited sources from 11 verified Indian suppliers across 282 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
13 Products Tracked
6 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 6 company name variants from customs records. For current shipment-level data, contact TransData Nexus.