Suveda Inc.d/b/a Kerala Ayurveda
Pharmaceutical Importer · United States · Ayurvedic & Herbal Products Focus · $17.4K Total Trade · DGFT Verified
Suveda Inc.d/b/a Kerala Ayurveda is a pharmaceutical importer based in United States with a total trade value of $17.4K across 2 products in 1 therapeutic categories. Based on 11 verified import shipments from Indian Customs (DGFT) records, Suveda Inc.d/b/a Kerala Ayurveda is the #1 buyer in 1 product including Adaptogen. Suveda Inc.d/b/a Kerala Ayurveda sources from 1 verified Indian supplier, with Kerala Ayurveda Limited accounting for 100.0% of imports.
Suveda Inc.d/b/a Kerala Ayurveda — Import Portfolio & Supplier Network
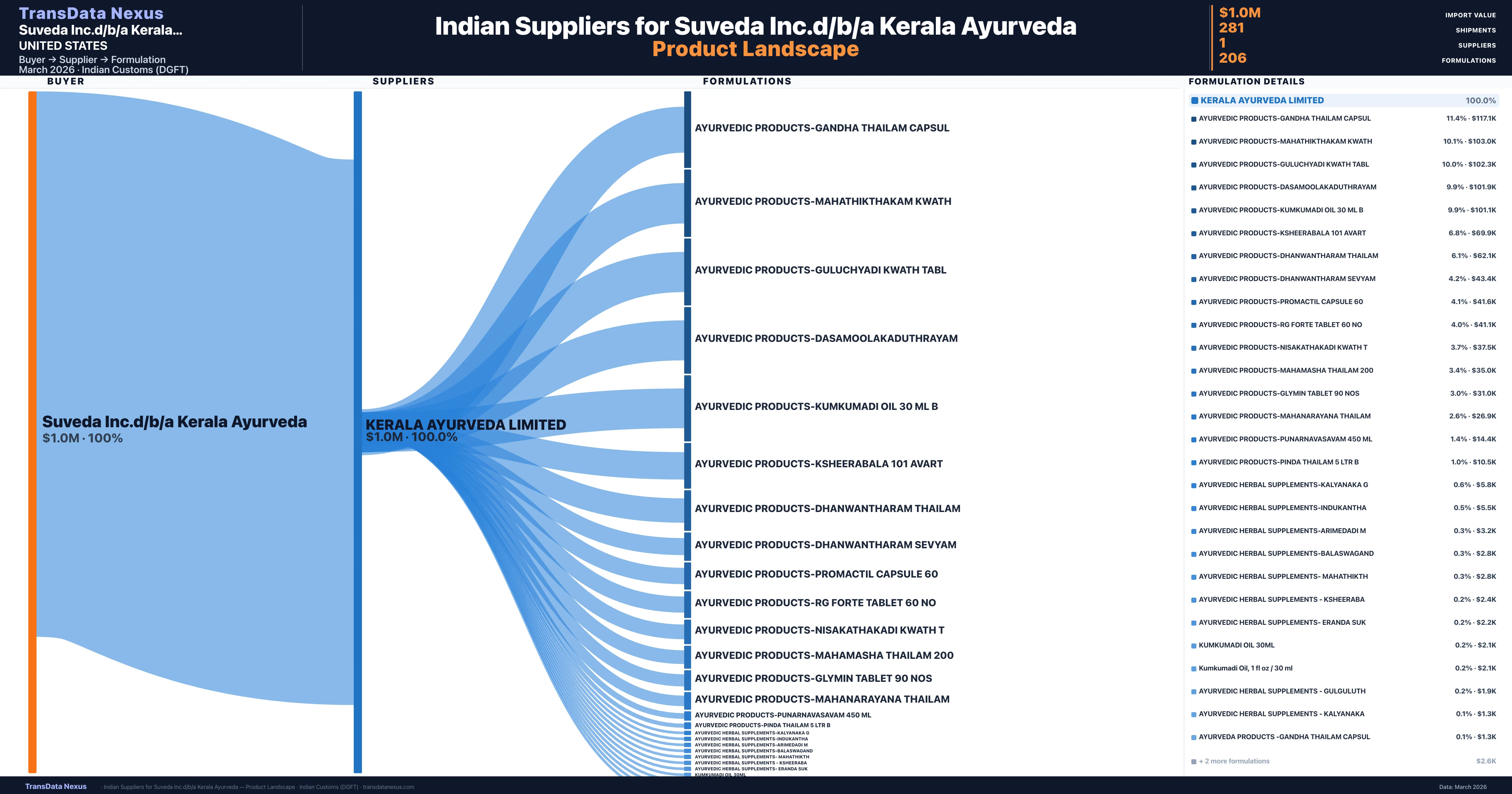
Who Are the Verified Indian Suppliers to Suveda Inc.d/b/a Kerala Ayurveda?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Kerala Ayurveda Limited | $1.0M | 281 | 100.0% |
Suveda Inc.d/b/a Kerala Ayurveda sources from 1 verified Indian supplier across 206 distinct formulations. The sourcing is highly concentrated — Kerala Ayurveda Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Suveda Inc.d/b/a Kerala Ayurveda Import?
| Formulation | Value | Ships |
|---|---|---|
| Ayurvedic products-gandha thailam | $117.1K | 6 |
| Ayurvedic products-mahathikthakam | $103.0K | 4 |
| Ayurvedic products-guluchyadi kwath | $102.3K | 4 |
| Ayurvedic products-dasamoolakaduthrayam | $101.9K | 4 |
| Ayurvedic products-kumkumadi oil 30 ML | $101.1K | 3 |
| Ayurvedic products-ksheerabala 101 | $69.9K | 3 |
| Ayurvedic products-dhanwantharam | $62.1K | 8 |
| Ayurvedic products-dhanwantharam | $43.4K | 4 |
| Ayurvedic products-promactil capsule | $41.6K | 4 |
| Ayurvedic products-rg forte tablet 60 | $41.1K | 3 |
| Ayurvedic products-nisakathakadi kwath | $37.5K | 4 |
| Ayurvedic products-mahamasha thailam | $35.0K | 4 |
| Ayurvedic products-glymin tablet 90 | $31.0K | 4 |
| Ayurvedic products-mahanarayana | $26.9K | 4 |
| Ayurvedic products-punarnavasavam 450 ML | $14.4K | 3 |
Suveda Inc.d/b/a Kerala Ayurveda imports 206 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Suveda Inc.d/b/a Kerala Ayurveda Import?
Top Products by Import Value
Suveda Inc.d/b/a Kerala Ayurveda Therapeutic Categories — 1 Specializations
Suveda Inc.d/b/a Kerala Ayurveda imports across 1 therapeutic categories, with Ayurvedic & Herbal Products (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Ayurvedic & Herbal Products
2 products · 100.0% · $17.4K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Asava | Ayurvedic & Herbal Products | $14.5K | 4 | 3.2% | 5 |
| 2 | Adaptogen | Ayurvedic & Herbal Products | $2.9K | 7 | 46.6% | 1 |
Suveda Inc.d/b/a Kerala Ayurveda imports 2 pharmaceutical products across 1 categories into United States totaling $17.4K. The company is the #1 buyer for 1 product: Adaptogen.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Suveda Inc.d/b/a Kerala Ayurveda.
Request DemoSuveda Inc.d/b/a Kerala Ayurveda — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Suveda Inc., operating as Kerala Ayurveda, is a U.S.-based pharmaceutical importer specializing in Ayurvedic and herbal products. Headquartered at 691 S Milpitas Blvd, Milpitas, California, the company imports finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, from India. Suveda Inc. is a wholly owned subsidiary of Kerala Ayurveda Limited, an India-based company engaged in the manufacture of Ayurvedic products, research, academies, clinics, hospitals, wellness resorts, and the cultivation of medicinal herbs.
In the United States pharmaceutical distribution landscape, Suveda Inc. serves as a niche importer, focusing on Ayurvedic and herbal formulations. By leveraging its parent company's extensive network and expertise in Ayurvedic medicine, Suveda Inc. plays a pivotal role in introducing traditional Indian remedies to the U.S. market, catering to consumers seeking alternative and complementary health solutions.
2Distribution Network
Suveda Inc. operates its distribution network primarily from its headquarters in Milpitas, California. While specific details about additional warehouse locations and logistics capabilities are not publicly disclosed, the company's strategic position in California facilitates efficient distribution across the United States. California's robust infrastructure, including major ports and transportation networks, supports Suveda Inc.'s ability to manage imports and deliveries effectively. The company's focus on Ayurvedic and herbal products suggests a targeted distribution approach, likely emphasizing regions with higher demand for alternative health products.
3Industry Role
Suveda Inc., operating as Kerala Ayurveda, functions as a specialized pharmaceutical importer within the U.S. market. By focusing exclusively on Ayurvedic and herbal formulations imported from India, the company serves a niche segment of consumers interested in traditional and alternative health remedies. This strategic positioning allows Suveda Inc. to cater to a growing demand for natural and holistic health solutions, differentiating itself from mainstream pharmaceutical distributors.
Supplier Relationship Intelligence — Suveda Inc.d/b/a Kerala Ayurveda
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Suveda Inc. demonstrates a high degree of supplier concentration, sourcing all its imported products from a single supplier, Kerala Ayurveda Limited. This single-source dependency indicates a strategic choice to maintain consistency and quality control over the imported formulations. The substantial volume of imports—totaling $17,000 across 11 shipments—reflects a stable and ongoing relationship between Suveda Inc. and Kerala Ayurveda Limited. However, this concentration also exposes Suveda Inc. to potential risks associated with supply chain disruptions, such as production delays or regulatory changes affecting the sole supplier.
2Supply Chain Resilience
The resilience of Suveda Inc.'s supply chain is closely tied to its exclusive relationship with Kerala Ayurveda Limited. While this partnership ensures product consistency and quality, it also means that any disruptions within Kerala Ayurveda Limited—such as production issues, regulatory challenges, or logistical problems—could directly impact Suveda Inc.'s ability to meet U.S. market demands. The lack of publicly available information regarding backup suppliers or alternative sourcing strategies suggests a potential vulnerability in the supply chain. To enhance resilience, Suveda Inc. may consider diversifying its supplier base or establishing contingency plans to mitigate risks associated with sole-source dependency.
3Strategic Implications
Suveda Inc.'s sourcing pattern, characterized by exclusive reliance on Kerala Ayurveda Limited, positions the company as a dedicated importer of Ayurvedic and herbal products from India. This focus allows Suveda Inc. to offer a curated selection of traditional health remedies, catering to a specific consumer segment seeking alternative health solutions. For Indian exporters, this presents an opportunity to establish a direct partnership with Suveda Inc., potentially gaining access to the U.S. market through a dedicated importer. However, the exclusivity of the relationship may limit opportunities for other exporters unless they can offer unique products or formulations that complement Suveda Inc.'s existing portfolio.
Importing Pharmaceuticals into United States — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United States
1Regulatory Authority & Framework
In the United States, the Food and Drug Administration (FDA) serves as the primary regulatory authority overseeing the importation and marketing of pharmaceutical products, including Ayurvedic and herbal formulations. The FDA enforces regulations to ensure that imported drugs meet safety, efficacy, and quality standards. Key legislation governing pharmaceutical imports includes the Federal Food, Drug, and Cosmetic Act, which outlines requirements for drug approval, labeling, and manufacturing practices. For Indian generics, the FDA's Abbreviated New Drug Application (ANDA) pathway allows for the approval of generic drugs based on bioequivalence to a reference listed drug, facilitating market entry for generic formulations.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products in the U.S. are stringent. Importers must ensure that their products comply with FDA regulations, which may include obtaining an import license and providing evidence of Good Manufacturing Practice (GMP) compliance. The FDA recognizes GMP certifications from reputable international bodies, such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S), provided they meet FDA standards. Importers must also obtain wholesale distribution authorization, which involves registration with state authorities and adherence to specific state regulations governing the distribution of pharmaceutical products.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet FDA standards for safety, efficacy, and quality. Stability requirements are set to ensure that products maintain their intended potency and safety throughout their shelf life. Labeling must comply with FDA regulations, including requirements for labeling language, which must be in English, and serialization mandates to facilitate traceability and prevent counterfeit products. These regulations are designed to protect consumers and ensure the integrity of the pharmaceutical supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, the FDA has implemented several policy changes affecting the importation of pharmaceutical products, including those from India. These changes include stricter enforcement of GMP compliance, enhanced scrutiny of labeling practices, and increased requirements for import documentation to improve transparency and traceability. Additionally, the FDA has updated its guidelines for the approval of generic drugs, impacting the market entry of Indian generics. Importers like Suveda Inc. must stay informed about these regulatory changes to ensure continued compliance and uninterrupted market access.
Suveda Inc.d/b/a Kerala Ayurveda — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Suveda Inc.'s focus on Ayurvedic and herbal products aligns with a growing consumer interest in natural and alternative health solutions in the United States. The company's import strategy emphasizes products that cater to specific therapeutic areas, such as adaptogens and asavas, which are traditional Ayurvedic formulations used to balance bodily functions and promote overall health. Market demand for these products is driven by consumers seeking holistic approaches to health and wellness, as well as a desire for natural remedies with fewer side effects compared to conventional pharmaceuticals.
2Sourcing Profile
Suveda Inc.'s sourcing strategy is centered on importing finished pharmaceutical formulations from India, specifically from Kerala Ayurveda Limited. This approach ensures product authenticity and quality, as Kerala Ayurveda Limited is a well-established manufacturer with expertise in Ayurvedic medicine. The focus on finished formulations allows Suveda Inc. to offer ready-to-market products, streamlining the supply chain and reducing time to market. India's rich heritage in Ayurvedic medicine and its established manufacturing capabilities make it a strategic sourcing partner for Suveda Inc.
3Market Positioning
Suveda Inc. serves a niche segment of the U.S. market, focusing on consumers interested in Ayurvedic and herbal health products. By importing specialized formulations from India, the company positions itself as a provider of alternative health solutions, catering to those seeking natural remedies. This market positioning differentiates Suveda Inc. from mainstream pharmaceutical distributors and appeals to a growing demographic interested in holistic health approaches.
Seller's Guide — How to Become a Supplier to Suveda Inc.d/b/a Kerala Ayurveda
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with Suveda Inc., especially if they can offer unique Ayurvedic formulations that complement the existing product portfolio. However, the exclusivity of Suveda Inc.'s relationship with Kerala Ayurveda Limited may limit opportunities for other exporters unless they can provide distinctive products or formulations. Suppliers should also be prepared to meet the stringent regulatory requirements of the U.S. market, including FDA compliance and GMP certifications.
2Requirements & Qualifications
Indian exporters seeking to supply Suveda Inc. and enter the U.S. market must obtain FDA approval for their products, which involves demonstrating safety, efficacy, and quality. Products must comply with FDA regulations, including labeling requirements and GMP standards. Additionally, exporters should ensure that their products meet the specific needs and preferences of the U.S. consumer market, which may involve adapting formulations or packaging to align with local expectations.
3How to Approach
To establish a relationship with Suveda Inc., Indian exporters should first ensure that their products meet all FDA regulatory requirements and possess the necessary certifications. Engaging in direct communication with Suveda Inc. to understand their specific product needs and preferences is crucial. Participating in industry trade shows and networking events can also provide opportunities to connect with Suveda Inc. and other potential U.S. partners. Given the competitive nature of the U.S. market, exporters should be prepared to demonstrate the unique value proposition of their products and their ability to meet the demands of the U.S. consumer base.
Frequently Asked Questions — Suveda Inc.d/b/a Kerala Ayurveda
What products does Suveda Inc.d/b/a Kerala Ayurveda import from India?
Suveda Inc.d/b/a Kerala Ayurveda imports 2 pharmaceutical products across 1 categories. Top imports: Asava ($14.5K), Adaptogen ($2.9K).
Who supplies pharmaceuticals to Suveda Inc.d/b/a Kerala Ayurveda from India?
Suveda Inc.d/b/a Kerala Ayurveda sources from 1 verified Indian suppliers. The primary supplier is Kerala Ayurveda Limited (100.0% of imports, $1.0M).
What is Suveda Inc.d/b/a Kerala Ayurveda's total pharmaceutical import value?
Suveda Inc.d/b/a Kerala Ayurveda's total pharmaceutical import value from India is $17.4K, based on 11 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Suveda Inc.d/b/a Kerala Ayurveda focus on?
Suveda Inc.d/b/a Kerala Ayurveda imports across 1 categories. The largest: Ayurvedic & Herbal Products (100.0%).
Get Full Suveda Inc.d/b/a Kerala Ayurveda Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Suveda Inc.d/b/a Kerala Ayurveda identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Suveda Inc.d/b/a Kerala Ayurveda's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 11 individual customs records matching Suveda Inc.d/b/a Kerala Ayurveda.
- 5.Supplier Verification: Suveda Inc.d/b/a Kerala Ayurveda sources from 1 verified Indian suppliers across 206 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.