Sunshine Healthcare Lanka Limited
Pharmaceutical Importer · Sri Lanka · Cardiovascular Focus · $22.5M Total Trade · DGFT Verified
Sunshine Healthcare Lanka Limited is a pharmaceutical importer based in Sri Lanka with a total trade value of $22.5M across 11 products in 8 therapeutic categories. Based on 572 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Sunshine Healthcare Lanka Limited sources from 23 verified Indian suppliers, with Zydus Lifesciences Limited accounting for 51.8% of imports.
Sunshine Healthcare Lanka Limited — Import Portfolio & Supplier Network
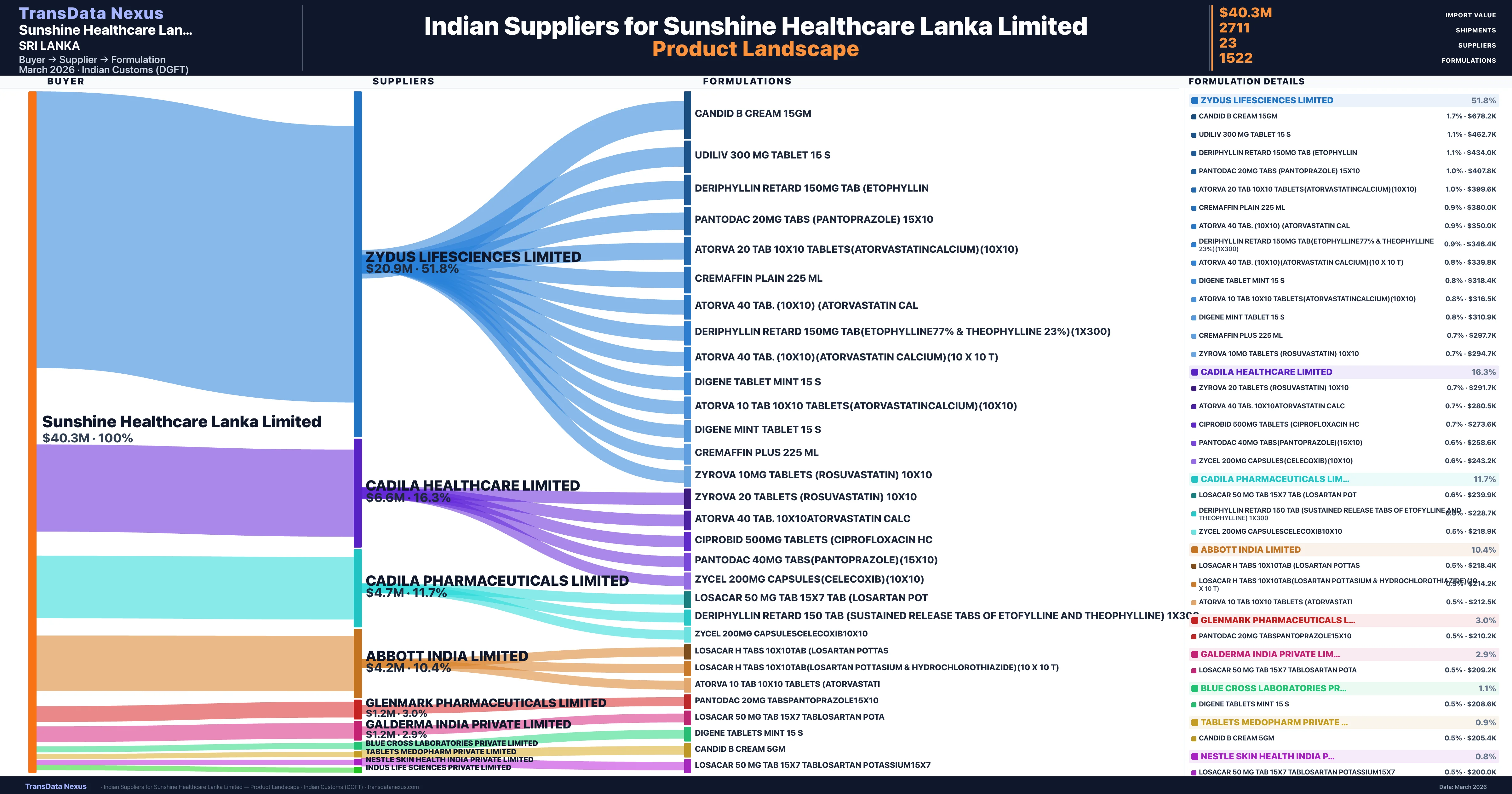
Who Are the Verified Indian Suppliers to Sunshine Healthcare Lanka Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Zydus Lifesciences Limited | $20.9M | 1,363 | 51.8% |
| Cadila Healthcare Limited | $6.6M | 474 | 16.3% |
| Cadila Pharmaceuticals Limited | $4.7M | 440 | 11.7% |
| Abbott India Limited | $4.2M | 162 | 10.4% |
| Glenmark Pharmaceuticals Limited | $1.2M | 30 | 3.0% |
| Galderma India Private Limited | $1.2M | 73 | 2.9% |
| Blue Cross Laboratories Private Limited | $449.0K | 40 | 1.1% |
| Tablets Medopharm Private Limited | $344.0K | 22 | 0.9% |
| Nestle Skin Health India Private Limited | $320.0K | 26 | 0.8% |
| Indus Life Sciences Private Limited | $153.5K | 23 | 0.4% |
| Fourrts I Laboratories Private Limited | $79.5K | 13 | 0.2% |
| Fourrts (india) Laboratories Private Limited | $60.6K | 5 | 0.2% |
| Ferring Pharmaceuticals Private Limited | $56.4K | 11 | 0.1% |
| Macleods Pharmaceuticals Limited | $37.8K | 6 | 0.1% |
| Jomed Health Care | $29.4K | 3 | 0.1% |
| Themis Medicare Limited | $16.7K | 3 | 0.0% |
| Corona Remedies Private Limited | $10.9K | 4 | 0.0% |
| Fdc Limited | $9.8K | 3 | 0.0% |
| S R S Enterprises | $8.8K | 2 | 0.0% |
| S.r.s.enterprises | $5.4K | 1 | 0.0% |
| Pharm Products Private Limited | $3.4K | 3 | 0.0% |
| Johnson Johnson Private Limited | $1.4K | 2 | 0.0% |
| Sterimed Surgicals (india) Private Limited | $1.0K | 2 | 0.0% |
Sunshine Healthcare Lanka Limited sources from 23 verified Indian suppliers across 1,522 distinct formulations. The supply base is diversified across 23 suppliers, reducing single-source dependency risk.
What Formulations Does Sunshine Healthcare Lanka Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Candid b cream 15GM | $678.2K | 15 |
| Udiliv 300 MG tablet 15 | $462.7K | 18 |
| Deriphyllin retard 150MG TAB | $434.0K | 13 |
| Pantodac 20MG tabs (pantoprazole) | $407.8K | 9 |
| Atorva 20 TAB 10x10 tablets(atorvastatincalcium)(10x10) | $399.6K | 8 |
| Cremaffin plain 225 ML | $380.0K | 9 |
| Atorva 40 TAB. (10x10) (atorvastatin | $350.0K | 7 |
| Deriphyllin retard 150MG TAB(etophylline77% & theophylline 23%)(1x300) | $346.4K | 14 |
| Atorva 40 TAB. (10x10)(atorvastatin calcium)(10 x 10 t) | $339.8K | 8 |
| Digene tablet mint 15 | $318.4K | 14 |
| Atorva 10 TAB 10x10 tablets(atorvastatincalcium)(10x10) | $316.5K | 9 |
| Digene mint tablet 15 | $310.9K | 11 |
| Cremaffin plus 225 ML | $297.7K | 11 |
| Zyrova 10MG tablets (rosuvastatin) | $294.7K | 7 |
| Zyrova 20 tablets (rosuvastatin) | $291.7K | 8 |
Sunshine Healthcare Lanka Limited imports 1,522 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Sunshine Healthcare Lanka Limited Import?
Sunshine Healthcare Lanka Limited Therapeutic Categories — 8 Specializations
Sunshine Healthcare Lanka Limited imports across 8 therapeutic categories, with Cardiovascular (43.5%), Vitamins & Supplements (20.0%), Gastrointestinal (13.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 86% of total imports.
Cardiovascular
2 products · 43.5% · $9.8M
Vitamins & Supplements
1 products · 20.0% · $4.5M
Gastrointestinal
2 products · 13.8% · $3.1M
Respiratory
2 products · 12.1% · $2.7M
Analgesics & Antipyretics
1 products · 6.1% · $1.4M
Respiratory & OTC
1 products · 1.8% · $413.4K
Brand Names & OTC Products
1 products · 1.5% · $336.3K
Antimalarial & Antiparasitic
1 products · 1.1% · $247.5K
Import Portfolio — Top 11 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Atorvastatin | Cardiovascular | $5.5M | 110 | 0.5% | 16 |
| 2 | Calcium | Vitamins & Supplements | $4.5M | 90 | 0.5% | 12 |
| 3 | Losartan | Cardiovascular | $4.3M | 88 | 1.4% | 8 |
| 4 | Pantoprazole | Gastrointestinal | $3.0M | 59 | 0.2% | 13 |
| 5 | Theophylline | Respiratory | $2.1M | 71 | 13.7% | 2 |
| 6 | Celecoxib | Analgesics & Antipyretics | $1.4M | 40 | 1.0% | 15 |
| 7 | Formoterol | Respiratory | $582.0K | 45 | 1.0% | 16 |
| 8 | Dextromethorphan | Respiratory & OTC | $413.4K | 9 | 2.8% | 5 |
| 9 | Brufen | Brand Names & OTC Products | $336.3K | 22 | 7.5% | 5 |
| 10 | Mebendazole | Antimalarial & Antiparasitic | $247.5K | 14 | 1.8% | 9 |
| 11 | Ranitidine | Gastrointestinal | $154.4K | 24 | 1.2% | 14 |
Sunshine Healthcare Lanka Limited imports 11 pharmaceutical products across 8 categories into Sri Lanka totaling $22.5M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Sunshine Healthcare Lanka Limited.
Request DemoSunshine Healthcare Lanka Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Sunshine Healthcare Lanka Limited, established in 1967, is Sri Lanka's first fully integrated healthcare company, encompassing pharmaceutical manufacturing, importation, distribution, and retailing. Headquartered in Colombo, the company operates under the umbrella of Sunshine Holdings PLC, a diversified conglomerate with interests in healthcare, consumer goods, and agribusiness. Sunshine Healthcare serves as a pivotal player in Sri Lanka's pharmaceutical distribution network, representing over 30 international companies, including Abbott Laboratories, Zydus Cadila, and Galderma. (sunshineholdings.lk)
The company's comprehensive operations span the entire pharmaceutical value chain, from research and development to last-mile distribution across the country. This integrated approach enables Sunshine Healthcare to maintain a significant presence in the Sri Lankan market, holding a 12% share of the private pharmaceutical sector. (sunshineholdings.lk) Their diverse product portfolio includes prescription and over-the-counter drugs, catering to various therapeutic areas such as dermatology, diabetes, cardiovascular health, gastrointestinal disorders, and pain management. (sunshineholdings.lk)
2Distribution Network
Sunshine Healthcare Lanka Limited boasts an extensive distribution network that ensures nationwide coverage of pharmaceutical products. The company operates multiple warehouses strategically located to facilitate efficient logistics and timely delivery. Their distribution infrastructure is designed to meet the stringent requirements of regulatory authorities and international principals, incorporating quality management systems and periodic internal audits to maintain high standards. (sunshineholdings.lk)
The company's logistics capabilities are robust, supporting a wide range of customer channels, including retail pharmacies, modern trade, hospitals, and government tenders. This comprehensive distribution system underscores Sunshine Healthcare's commitment to making quality medicines accessible to all Sri Lankans, thereby enhancing the overall healthcare landscape of the country. (sunshineholdings.lk)
3Industry Role
Sunshine Healthcare Lanka Limited plays a multifaceted role in Sri Lanka's pharmaceutical supply chain. As a primary wholesaler, the company imports and distributes a diverse array of pharmaceutical products, ensuring their availability across various healthcare settings. Additionally, Sunshine Healthcare serves as a parallel importer, sourcing products from international markets to meet local demand. Their extensive partnerships with global pharmaceutical companies enable them to supply a wide range of medicines, including those not readily available through other channels. (sunshineholdings.lk)
Furthermore, Sunshine Healthcare acts as a hospital supplier, providing essential medicines and medical devices to both public and private healthcare institutions. Their involvement in government tenders and supply to government-owned pharmacy chains, such as the State Pharmaceutical Corporation (SPC) and Osu Sala, highlights their significant role in the public healthcare sector. (sunshineholdings.lk)
Supplier Relationship Intelligence — Sunshine Healthcare Lanka Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Sunshine Healthcare Lanka Limited's sourcing strategy exhibits a high degree of concentration, with the top five products accounting for 86.2% of their total import value from India. This concentration indicates a strategic focus on key therapeutic areas, particularly in cardiovascular, vitamins and supplements, and gastrointestinal categories. The substantial import values of Atorvastatin ($5.5M), Calcium ($4.5M), Losartan ($4.3M), Pantoprazole ($3.0M), and Theophylline ($2.1M) underscore the company's emphasis on these segments.
The dominance of a few products suggests a deliberate sourcing approach aimed at meeting the most pressing healthcare needs in Sri Lanka. However, this concentration also implies potential risks, such as supply chain disruptions affecting these critical products. The company's reliance on a limited number of suppliers for these items may impact the stability and resilience of their supply chain.
2Supply Chain Resilience
Sunshine Healthcare Lanka Limited's supply chain resilience is closely tied to its sourcing concentration. The company's dependence on a select group of suppliers for its top five imported products necessitates robust risk management strategies to mitigate potential disruptions. While the company has established long-term partnerships with reputable Indian pharmaceutical manufacturers, the limited number of suppliers for key products could pose challenges in maintaining a consistent supply.
To enhance supply chain resilience, Sunshine Healthcare may consider diversifying its supplier base and exploring alternative sourcing options. Additionally, strengthening relationships with existing suppliers and implementing contingency plans can help mitigate risks associated with supply chain disruptions. Ensuring compliance with international quality standards and maintaining a diverse product portfolio can further bolster the company's ability to adapt to changing market dynamics.
3Strategic Implications
The sourcing pattern of Sunshine Healthcare Lanka Limited reflects a strategic alignment with the most prevalent health concerns in Sri Lanka, particularly in cardiovascular diseases, vitamin deficiencies, and gastrointestinal disorders. By focusing on these therapeutic areas, the company positions itself as a key player in addressing critical healthcare needs. This targeted approach allows Sunshine Healthcare to leverage its expertise and resources effectively, enhancing its competitive edge in the market.
For Indian pharmaceutical exporters, understanding Sunshine Healthcare's sourcing preferences presents an opportunity to align their product offerings with the company's focus areas. By ensuring the availability of high-quality products in these therapeutic segments, Indian exporters can strengthen their partnerships with Sunshine Healthcare and expand their presence in the Sri Lankan market. This strategic alignment can lead to mutually beneficial collaborations, fostering long-term business relationships.
Importing Pharmaceuticals into Sri Lanka — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Sri Lanka
1Regulatory Authority & Framework
In Sri Lanka, the National Medicines Regulatory Authority (NMRA) serves as the primary regulatory body overseeing the registration, importation, and distribution of pharmaceutical products. The NMRA is responsible for ensuring the quality, safety, and efficacy of medicines available in the country. Key legislation governing pharmaceutical imports includes the National Medicines Regulatory Authority Act, which outlines the procedures for drug registration, import control, and market surveillance. (nmra.gov.lk)
The NMRA's Medicines Evaluation Committee (MEC) conducts scientific and technical reviews of medicines submitted for registration, ensuring compliance with established standards. This committee plays a crucial role in maintaining the integrity of the pharmaceutical supply chain by evaluating the quality and safety of medicinal products. (nmra.gov.lk)
2Import Licensing & GMP
To import pharmaceutical products into Sri Lanka, companies must obtain an import license from the NMRA. This process involves submitting detailed product information, including Certificates of Pharmaceutical Product (COPP), Certificates of Analysis (COA), and Batch Release Certificates. Additionally, the NMRA requires that imported medicines comply with Good Manufacturing Practice (GMP) standards recognized by international bodies such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). (nmra.gov.lk)
Sunshine Healthcare Lanka Limited, as a major importer, ensures that its suppliers adhere to these GMP standards, thereby maintaining the quality and safety of the pharmaceutical products it distributes. The company also holds the necessary wholesale distribution authorizations, enabling it to operate effectively within the regulatory framework established by the NMRA.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality and compliance with Sri Lankan standards. The NMRA mandates that labeling includes essential information such as product name, active ingredients, dosage form, strength, batch number, manufacturing date, expiry date, storage conditions, and the manufacturer's details. Labeling must be in English and Sinhala, ensuring accessibility to a broad segment of the population. (nmra.gov.lk)
Serialization mandates are also in place to enhance traceability and prevent counterfeit products from entering the market. These measures are part of a comprehensive strategy to uphold the integrity of the pharmaceutical supply chain and protect public health.
4Recent Regulatory Changes
Between 2024 and 2026, the NMRA has implemented several policy changes affecting pharmaceutical imports into Sri Lanka. These include revisions to the guidelines for the registration of medicines, updates to import control procedures, and the introduction of stricter quality assurance protocols. The NMRA has also emphasized the need for compliance with international standards, particularly concerning GMP and labeling requirements. (nmra.gov.lk)
These regulatory changes aim to enhance the safety and efficacy of pharmaceutical products in the Sri Lankan market. Companies like Sunshine Healthcare Lanka Limited must stay abreast of these developments to ensure continued compliance and maintain their market position.
Sunshine Healthcare Lanka Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Sunshine Healthcare Lanka Limited's product strategy focuses on therapeutic areas with high prevalence and unmet needs in Sri Lanka, such as cardiovascular diseases, vitamin deficiencies, and gastrointestinal disorders. By concentrating on these segments, the company addresses critical health concerns and aligns its offerings with the demands of the local market. The substantial import values of key products like Atorvastatin, Calcium, Losartan, Pantoprazole, and Theophylline reflect this strategic focus.
The company's emphasis on these therapeutic areas is driven by market demand and the opportunity to provide effective solutions to prevalent health issues. By offering a range of products in these categories, Sunshine Healthcare enhances its competitive position and contributes to improving public health outcomes in Sri Lanka.
Frequently Asked Questions — Sunshine Healthcare Lanka Limited
What products does Sunshine Healthcare Lanka Limited import from India?
Sunshine Healthcare Lanka Limited imports 11 pharmaceutical products across 8 categories. Top imports: Atorvastatin ($5.5M), Calcium ($4.5M), Losartan ($4.3M), Pantoprazole ($3.0M), Theophylline ($2.1M).
Who supplies pharmaceuticals to Sunshine Healthcare Lanka Limited from India?
Sunshine Healthcare Lanka Limited sources from 23 verified Indian suppliers. The primary supplier is Zydus Lifesciences Limited (51.8% of imports, $20.9M).
What is Sunshine Healthcare Lanka Limited's total pharmaceutical import value?
Sunshine Healthcare Lanka Limited's total pharmaceutical import value from India is $22.5M, based on 572 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Sunshine Healthcare Lanka Limited focus on?
Sunshine Healthcare Lanka Limited imports across 8 categories. The largest: Cardiovascular (43.5%), Vitamins & Supplements (20.0%), Gastrointestinal (13.8%).
Get Full Sunshine Healthcare Lanka Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Sunshine Healthcare Lanka Limited identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Sunshine Healthcare Lanka Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 572 individual customs records matching Sunshine Healthcare Lanka Limited.
- 5.Supplier Verification: Sunshine Healthcare Lanka Limited sources from 23 verified Indian suppliers across 1,522 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
11 Products Tracked
8 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.