Substipharm
Pharmaceutical Importer · France · Analgesics & Antipyretics Focus · $7.3M Total Trade · DGFT Verified
Substipharm is a pharmaceutical importer based in France with a total trade value of $7.3M across 3 products in 3 therapeutic categories. Based on 209 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Substipharm sources from 1 verified Indian supplier, with Medreich Limited accounting for 100.0% of imports.
Substipharm — Import Portfolio & Supplier Network
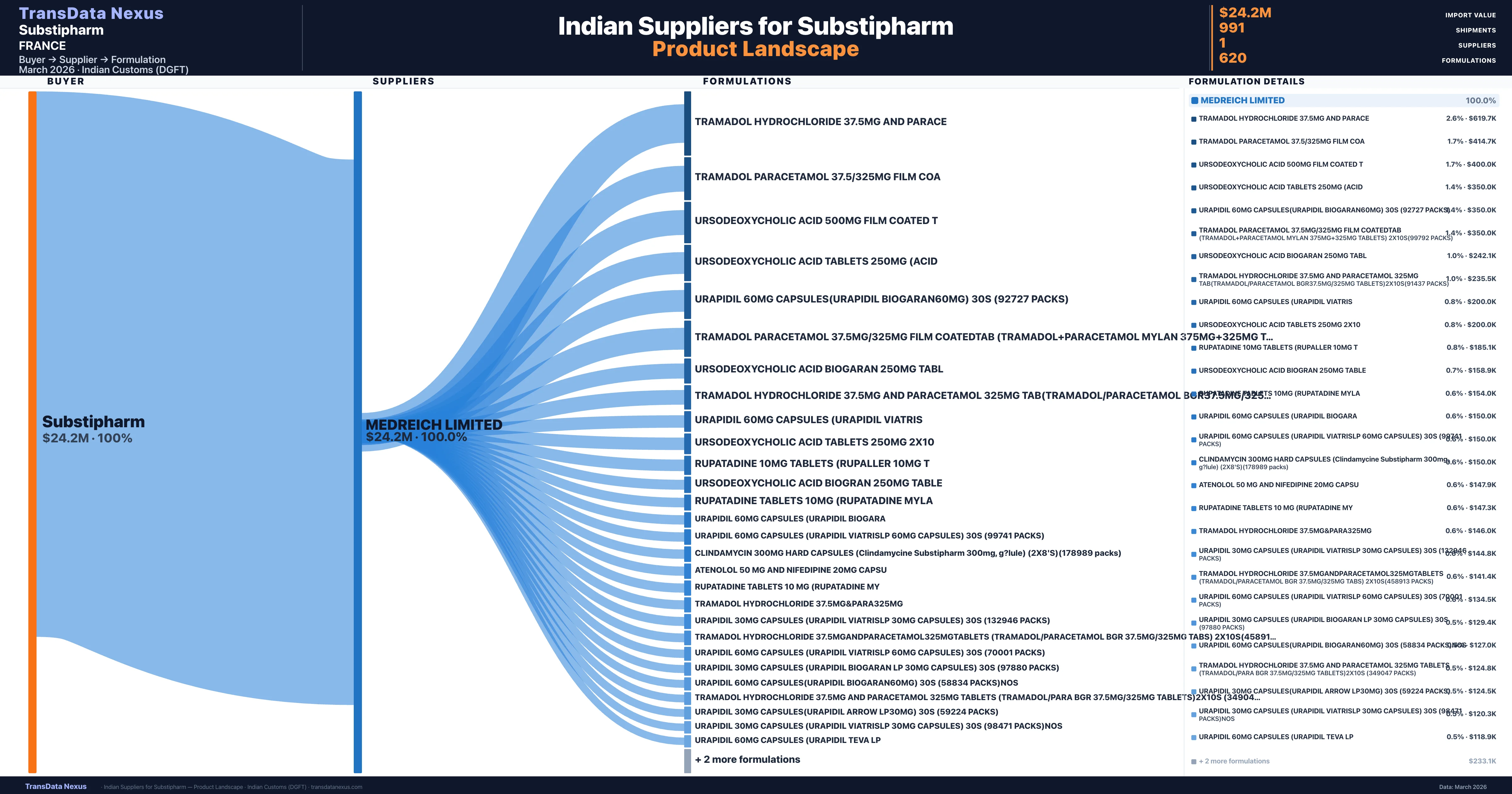
Who Are the Verified Indian Suppliers to Substipharm?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Medreich Limited | $24.2M | 991 | 100.0% |
Substipharm sources from 1 verified Indian supplier across 620 distinct formulations. The sourcing is highly concentrated — Medreich Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Substipharm Import?
| Formulation | Value | Ships |
|---|---|---|
| Tramadol hydrochloride 37.5MG and | $619.7K | 20 |
| Tramadol paracetamol 37.5/325MG film | $414.7K | 9 |
| Ursodeoxycholic acid 500MG film coated | $400.0K | 8 |
| Ursodeoxycholic acid tablets 250MG | $350.0K | 7 |
| Urapidil 60MG capsules(urapidil biogaran60mg) 30s (92727 packs) | $350.0K | 7 |
| Tramadol paracetamol 37.5MG/325MG film coatedtab (tramadol+paracetamol mylan 375MG+325MG tablets) 2x10s(99792 packs) | $350.0K | 7 |
| Ursodeoxycholic acid biogaran 250MG | $242.1K | 5 |
| Tramadol hydrochloride 37.5MG and paracetamol 325MG TAB(tramadol/paracetamol bgr37.5MG/325MG tablets)2x10s(91437 packs) | $235.5K | 7 |
| Urapidil 60MG capsules (urapidil | $200.0K | 4 |
| Ursodeoxycholic acid tablets 250MG | $200.0K | 4 |
| Rupatadine 10MG tablets (rupaller 10MG | $185.1K | 5 |
| Ursodeoxycholic acid biogran 250MG | $158.9K | 4 |
| Rupatadine tablets 10MG (rupatadine | $154.0K | 6 |
| Urapidil 60MG capsules (urapidil | $150.0K | 3 |
| Urapidil 60MG capsules (urapidil viatrislp 60MG capsules) 30s (99741 packs) | $150.0K | 3 |
Substipharm imports 620 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Substipharm Import?
Top Products by Import Value
Substipharm Therapeutic Categories — 3 Specializations
Substipharm imports across 3 therapeutic categories, with Analgesics & Antipyretics (92.8%), Cardiovascular (5.2%), Gastrointestinal (2.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Analgesics & Antipyretics
1 products · 92.8% · $6.8M
Cardiovascular
1 products · 5.2% · $376.0K
Gastrointestinal
1 products · 2.0% · $148.5K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Tramadol | Analgesics & Antipyretics | $6.8M | 190 | 1.8% | 4 |
| 2 | Nifedipine | Cardiovascular | $376.0K | 14 | 1.2% | 14 |
| 3 | Bisacodyl | Gastrointestinal | $148.5K | 5 | 1.7% | 8 |
Substipharm imports 3 pharmaceutical products across 3 categories into France totaling $7.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Substipharm.
Request DemoSubstipharm — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Substipharm is a privately held French pharmaceutical group headquartered in Paris, France. Established in 1995, the company specializes in the development, registration, and commercialization of generic and specialty drugs, as well as vaccines. With a portfolio exceeding 90 different molecules, Substipharm operates in over 90 countries, including direct presence in Europe and Southeast Asia.
The company's operations are divided into two main divisions: Pharmaceuticals and Biologics. The Pharmaceuticals division focuses on branded specialties and generics, while the Biologics division is dedicated to vaccines, particularly the Japanese Encephalitis vaccine, IMOJEV®. Substipharm's business model combines in-house development of pharmaceutical dossiers with direct commercialization and strategic partnerships.
2Distribution Network
Substipharm's distribution network is extensive, with operations spanning over 90 countries worldwide. The company has established commercial affiliates in key markets, including the United Kingdom, Italy, and Switzerland. In the UK, Substipharm operates through its affiliate, Vygoris, which commenced sales of its product portfolio in 2016. The Italian affiliate, Substipharm Srl, was established in 2020 to further expand its European presence. Additionally, Substipharm has a subsidiary in Geneva, Switzerland, and a biologics division in Bangkok, Thailand, focusing on the distribution of IMOJEV® in Southeast Asia.
3Industry Role
In France's pharmaceutical supply chain, Substipharm functions as a pharmaceutical importer and distributor. The company imports finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, from various countries, including India. Substipharm's role encompasses both direct commercialization of its own product portfolio and the supply of products through licensing and distribution partnerships. This dual approach allows the company to serve a broad spectrum of the pharmaceutical market, from generics to specialty drugs and vaccines.
Supplier Relationship Intelligence — Substipharm
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Substipharm's sourcing strategy exhibits a high degree of concentration, with a significant reliance on a single supplier, MEDREICH LIMITED, accounting for 100% of its pharmaceutical imports from India. This exclusive partnership has resulted in 991 shipments, indicating a stable and long-term relationship. While such concentration can lead to favorable terms and streamlined logistics, it also poses risks related to supply chain disruptions, regulatory changes, or operational issues within the supplier. Diversifying the supplier base could mitigate these risks and enhance supply chain resilience.
2Supply Chain Resilience
The resilience of Substipharm's Indian supply chain is closely tied to its exclusive relationship with MEDREICH LIMITED. The absence of backup suppliers for Indian imports suggests potential vulnerabilities in the event of disruptions. The company's import portfolio comprises 620 unique formulations, indicating a diverse range of products. However, the heavy reliance on a single supplier for these formulations may limit flexibility and responsiveness to market changes. Ensuring that MEDREICH LIMITED adheres to international quality standards and maintains robust operational capabilities is crucial for sustaining supply chain stability.
3Strategic Implications
Substipharm's concentrated sourcing from MEDREICH LIMITED positions the company to negotiate favorable terms and maintain a streamlined supply chain. However, this strategy also exposes Substipharm to risks associated with supplier dependency. For Indian exporters, the existing relationship between Substipharm and MEDREICH LIMITED may present challenges in entering the French market. To establish a foothold, Indian suppliers would need to demonstrate superior product quality, competitive pricing, and the ability to meet regulatory requirements. Building direct relationships with Substipharm could offer opportunities to diversify their customer base and reduce reliance on a single distributor.
Importing Pharmaceuticals into France — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for France
1Regulatory Authority & Framework
In France, the primary regulatory authority overseeing pharmaceutical imports is the French National Agency for Medicines and Health Products Safety (ANSM). The agency is responsible for ensuring that all medicinal products, including those imported from countries like India, meet stringent safety, efficacy, and quality standards. Key legislation governing pharmaceutical imports includes the French Public Health Code, which aligns with European Union directives and regulations. The marketing authorization pathway for Indian generics involves obtaining a Marketing Authorization (MA) from ANSM, which requires comprehensive documentation demonstrating bioequivalence to the reference product, compliance with Good Manufacturing Practice (GMP), and adherence to labeling and packaging requirements.
2Import Licensing & GMP
Import licensing in France necessitates that foreign manufacturers hold a valid GMP certificate recognized by ANSM. Certificates from the World Health Organization (WHO) or the Pharmaceutical Inspection Co-operation Scheme (PIC/S) are typically accepted. Indian exporters must ensure that their manufacturing facilities comply with these standards to facilitate market entry. Additionally, wholesalers like Substipharm must obtain authorization from ANSM to distribute pharmaceutical products within France. This authorization process includes demonstrating compliance with storage, handling, and distribution regulations to ensure product integrity and patient safety.
3Quality & Labeling
Pharmaceutical products imported into France must undergo batch testing to confirm quality and safety. Stability studies are required to establish shelf-life and storage conditions. Labeling must be in French and include essential information such as dosage instructions, side effects, and storage conditions. Serialization mandates are in place to prevent counterfeit products, requiring unique identifiers on packaging to trace and verify products throughout the supply chain. Compliance with these requirements is essential for market authorization and consumer trust.
4Recent Regulatory Changes
Between 2024 and 2026, France implemented several regulatory changes affecting pharmaceutical imports. These include stricter bioequivalence testing requirements for generics, enhanced serialization protocols to combat counterfeit drugs, and updated GMP compliance standards. Indian pharmaceutical exporters must stay informed about these changes to ensure continued access to the French market. Engaging with ANSM and consulting with regulatory experts can aid in navigating these evolving requirements.
Substipharm — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Substipharm's focus on importing analgesics and antipyretics, particularly tramadol, aligns with the high demand for pain management medications in France. The inclusion of cardiovascular and gastrointestinal products indicates a strategic approach to address prevalent health conditions. The company's portfolio of over 90 molecules, including mature brands and generics, reflects a commitment to providing comprehensive therapeutic solutions. This diverse product strategy enables Substipharm to cater to a wide patient demographic and maintain a competitive edge in the market.
2Sourcing Profile
Substipharm's sourcing strategy emphasizes the importation of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, from India. This approach allows the company to leverage India's manufacturing capabilities and cost advantages while focusing on the commercialization and distribution aspects within France. The reliance on a single supplier, MEDREICH LIMITED, suggests a strategic choice to streamline operations and maintain consistency in product quality. However, exploring additional suppliers could enhance supply chain resilience and mitigate potential risks associated with supplier dependency.
3Market Positioning
Substipharm serves multiple segments of the French pharmaceutical market, including retail pharmacies, hospitals, and government tenders. The company's diverse product portfolio, encompassing generics and specialty drugs, enables it to meet the needs of various healthcare providers and patients. By importing and distributing a wide range of pharmaceutical products, Substipharm positions itself as a versatile and reliable partner in the French healthcare system.
Seller's Guide — How to Become a Supplier to Substipharm
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter the French market by partnering with Substipharm. To capitalize on this opportunity, Indian exporters should focus on demonstrating product quality, compliance with regulatory standards, and the ability to meet market demands. Diversifying Substipharm's supplier base could lead to more competitive pricing and a broader product offering, benefiting both the company and its customers.
2Requirements & Qualifications
Indian exporters aiming to supply Substipharm and the French market must ensure compliance with ANSM's GMP standards, which are recognized by WHO and PIC/S. Obtaining a valid GMP certificate from these organizations is essential for import authorization. Additionally, products must meet French labeling requirements, including French language labeling and serialization mandates. Engaging with regulatory consultants familiar with French pharmaceutical regulations can facilitate the compliance process.
3How to Approach
Indian exporters should initiate contact with Substipharm by providing detailed product information, including specifications, certifications, and compliance with regulatory standards. Participating in pharmaceutical trade fairs and industry events can enhance visibility and foster relationships. Understanding the regulatory filing process and preparing necessary documentation for ANSM approval is crucial. Establishing a clear timeline for product registration and market entry, in collaboration with Substipharm, will aid in efficient market penetration.
Frequently Asked Questions — Substipharm
What products does Substipharm import from India?
Substipharm imports 3 pharmaceutical products across 3 categories. Top imports: Tramadol ($6.8M), Nifedipine ($376.0K), Bisacodyl ($148.5K).
Who supplies pharmaceuticals to Substipharm from India?
Substipharm sources from 1 verified Indian suppliers. The primary supplier is Medreich Limited (100.0% of imports, $24.2M).
What is Substipharm's total pharmaceutical import value?
Substipharm's total pharmaceutical import value from India is $7.3M, based on 209 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Substipharm focus on?
Substipharm imports across 3 categories. The largest: Analgesics & Antipyretics (92.8%), Cardiovascular (5.2%), Gastrointestinal (2.0%).
Get Full Substipharm Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Substipharm identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Substipharm's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 209 individual customs records matching Substipharm.
- 5.Supplier Verification: Substipharm sources from 1 verified Indian suppliers across 620 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.