State Pharmaceuticals Corporation O
Pharmaceutical Importer · Sri Lanka · Advanced Diabetes Medications Focus · $1.1M Total Trade · DGFT Verified
State Pharmaceuticals Corporation O is a pharmaceutical importer based in Sri Lanka with a total trade value of $1.1M across 4 products in 4 therapeutic categories. Based on 23 verified import shipments from Indian Customs (DGFT) records, State Pharmaceuticals Corporation O is the #1 buyer in 1 product including Diagnostic. State Pharmaceuticals Corporation O sources from 53 verified Indian suppliers, with Wallace Pharmaceuticals Private Limited accounting for 7.4% of imports.
State Pharmaceuticals Corporation O — Import Portfolio & Supplier Network
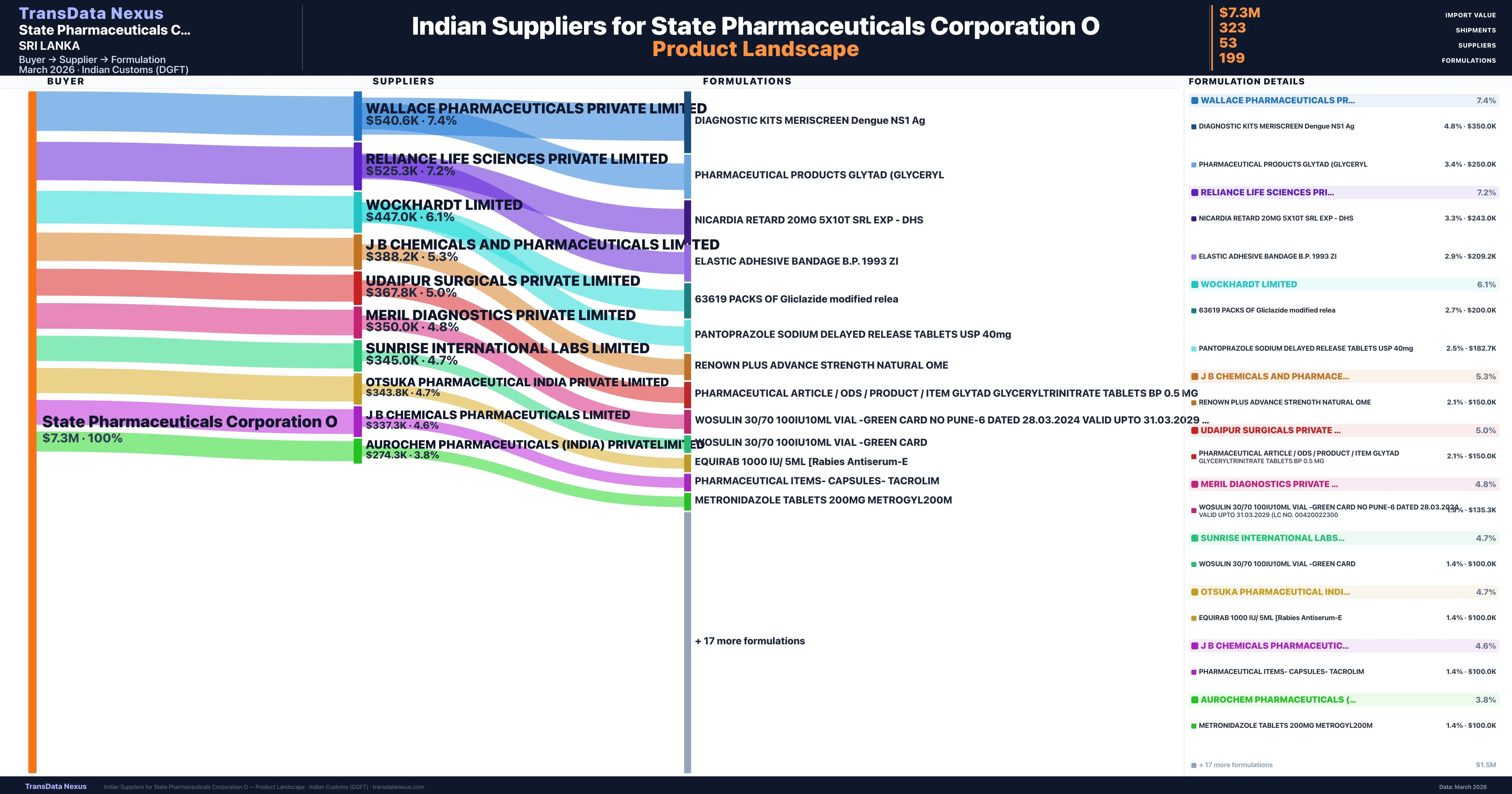
Who Are the Verified Indian Suppliers to State Pharmaceuticals Corporation O?
Customs-verified supplier relationships from Indian DGFT records
State Pharmaceuticals Corporation O sources from 53 verified Indian suppliers across 199 distinct formulations. The supply base is diversified across 53 suppliers, reducing single-source dependency risk.
What Formulations Does State Pharmaceuticals Corporation O Import?
| Formulation | Value | Ships |
|---|---|---|
| Diagnostic kits meriscreen dengue ns1 | $350.0K | 7 |
| Pharmaceutical products glytad | $250.0K | 5 |
| Nicardia retard 20MG 5x10t srl exp - | $243.0K | 5 |
| Elastic adhesive bandage b.p. 1993 | $209.2K | 6 |
| 63619 packs of gliclazide modified | $200.0K | 4 |
| Pantoprazole sodium delayed release tablets USP 40MG | $182.7K | 7 |
| Renown plus advance strength natural | $150.0K | 3 |
| Pharmaceutical article / ods / product / item glytad glyceryltrinitrate tablets BP 0.5 MG | $150.0K | 3 |
| Wosulin 30/70 100iu10ml vial -green card no pune-6 dated 28.03.2024 valid upto 31.03.2029 (lc no. | $135.3K | 3 |
| Wosulin 30/70 100iu10ml vial -green | $100.0K | 2 |
| Equirab 1000 IU/ 5ML [rabies | $100.0K | 2 |
| Pharmaceutical items- capsules- | $100.0K | 2 |
| Metronidazole tablets 200MG | $100.0K | 2 |
| Wosulin 30 70 100iu10ml vial green cardno 20956 dated 28 03 2019 valid upto 31 03 | $100.0K | 2 |
| Fexofenadine hydrochloride tablets USP 120MG fegra 120 10x6 tablets qty | $100.0K | 2 |
State Pharmaceuticals Corporation O imports 199 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does State Pharmaceuticals Corporation O Import?
Top Products by Import Value
State Pharmaceuticals Corporation O Therapeutic Categories — 4 Specializations
State Pharmaceuticals Corporation O imports across 4 therapeutic categories, with Advanced Diabetes Medications (39.1%), Medical Devices & Diagnostics (30.4%), Other (17.4%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Diabetes Medications
1 products · 39.1% · $450.0K
Medical Devices & Diagnostics
1 products · 30.4% · $350.0K
Other
1 products · 17.4% · $200.0K
Advanced Oncology
1 products · 13.0% · $150.0K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Gliclazide | Advanced Diabetes Medications | $450.0K | 9 | 0.1% | 14 |
| 2 | Diagnostic | Medical Devices & Diagnostics | $350.0K | 7 | 1.9% | 1 |
| 3 | Immunoglobulin | Other | $200.0K | 4 | 0.1% | 3 |
| 4 | Rituximab | Advanced Oncology | $150.0K | 3 | 0.8% | 4 |
State Pharmaceuticals Corporation O imports 4 pharmaceutical products across 4 categories into Sri Lanka totaling $1.1M. The company is the #1 buyer for 1 product: Diagnostic.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for State Pharmaceuticals Corporation O.
Request DemoState Pharmaceuticals Corporation O — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
State Pharmaceuticals Corporation O is a state-owned enterprise headquartered in Colombo, Sri Lanka. Established in 1971 under the State Industrial Corporations Act, it serves as the largest supplier of pharmaceuticals, surgical consumables, and laboratory chemicals in the country. The corporation's mission is to provide quality-assured healthcare items that are safe, effective, and affordable, while promoting the rational use of drugs among the public.
As a government procurement entity, State Pharmaceuticals Corporation O plays a pivotal role in Sri Lanka's pharmaceutical distribution network. It is the sole supplier of pharmaceuticals and related products to all institutions administered by the Ministry of Health. Additionally, it maintains a significant presence in the private sector, holding a 30% market share. The corporation operates a network of retail outlets known as Rajya Osu Salas (state drug shops), with 18 locations, and 36 franchise outlets, 48 distributors, and 20 authorized retailers across the country.
2Distribution Network
State Pharmaceuticals Corporation O's distribution network is extensive, encompassing both public and private healthcare sectors. Its retail outlets, Rajya Osu Salas, are strategically located to ensure nationwide coverage, with the original outlet in Colombo operating 24/7. The corporation's logistics capabilities are robust, enabling efficient supply chain management to meet the demands of various healthcare institutions and the general public. This extensive network underscores its critical role in ensuring the availability of essential medicines and healthcare products throughout Sri Lanka.
3Industry Role
In Sri Lanka's pharmaceutical supply chain, State Pharmaceuticals Corporation O functions primarily as a wholesaler and government procurement entity. It is the sole supplier of pharmaceuticals and related products to all institutions administered by the Ministry of Health, ensuring a centralized and standardized supply of essential medicines. In the private sector, it holds a 30% market share, supplying a wide range of pharmaceutical products to various private healthcare providers. This dual role enables the corporation to maintain a comprehensive presence across the healthcare spectrum, from public hospitals to private clinics and pharmacies.
Supplier Relationship Intelligence — State Pharmaceuticals Corporation O
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
State Pharmaceuticals Corporation O's sourcing strategy reveals a high degree of concentration, with its top five imported products accounting for 100% of its imports from India. This indicates a strategic focus on a select range of products, potentially to leverage bulk purchasing advantages and ensure consistent quality. However, such concentration also poses risks, including supply chain disruptions if any of these key products face shortages or regulatory issues. The corporation's reliance on a limited number of suppliers for these products suggests a need for robust contingency planning and supplier relationship management to mitigate potential risks.
2Supply Chain Resilience
The resilience of State Pharmaceuticals Corporation O's supply chain is closely tied to its sourcing strategy. The corporation's focus on a limited number of key products and suppliers may enhance efficiency but also increases vulnerability to supply chain disruptions. The absence of backup suppliers for these critical products suggests a potential area of concern. To bolster resilience, the corporation could consider diversifying its supplier base and establishing contingency plans to address potential disruptions. Additionally, ensuring that all suppliers adhere to stringent regulatory standards and maintaining a diverse portfolio of formulations can further strengthen the supply chain's robustness.
3Strategic Implications
State Pharmaceuticals Corporation O's concentrated sourcing pattern positions it to negotiate favorable terms with its suppliers, potentially securing better pricing and consistent product quality. For Indian exporters, this presents an opportunity to become preferred suppliers by meeting the corporation's stringent quality standards and demonstrating reliability. However, the concentration also means that any issues with these key products or suppliers could have significant repercussions. Therefore, Indian exporters seeking to supply State Pharmaceuticals Corporation O should focus on building strong, reliable partnerships and ensuring compliance with the corporation's quality and regulatory requirements.
Importing Pharmaceuticals into Sri Lanka — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Sri Lanka
1Regulatory Authority & Framework
The National Medicines Regulatory Authority (NMRA) is the primary regulatory body overseeing the pharmaceutical sector in Sri Lanka. Established in 2015 through an Act of Parliament, the NMRA is responsible for ensuring the safety, efficacy, and quality of medicines, medical devices, and related products in the country. It regulates the manufacturing, importation, distribution, and sale of these products, as well as clinical trials conducted within Sri Lanka. The NMRA operates under the Ministry of Health and is governed by a board that provides strategic leadership and oversight. (nmra.gov.lk)
2Import Licensing & GMP
Import licensing is a critical component of Sri Lanka's pharmaceutical regulatory framework. The NMRA requires all pharmaceutical imports to be accompanied by valid import licenses, ensuring that only approved products enter the market. Additionally, the NMRA recognizes Good Manufacturing Practice (GMP) certifications from reputable international bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). This recognition facilitates the importation of high-quality pharmaceutical products that meet international standards. Wholesale distribution authorization is also mandated, ensuring that entities involved in the distribution of pharmaceuticals adhere to regulatory requirements and maintain the integrity of the supply chain. (nmra.gov.lk)
3Quality & Labeling
The NMRA enforces stringent quality control measures for pharmaceutical products entering Sri Lanka. Imported medicines must undergo batch testing to verify their quality, safety, and efficacy. Stability requirements are also enforced to ensure that products maintain their intended quality throughout their shelf life. Labeling regulations stipulate that all pharmaceutical products display accurate and comprehensive information, including dosage instructions, side effects, and storage conditions. Labeling must be in the official languages of Sri Lanka, primarily Sinhala and Tamil, to ensure clear communication with consumers. Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the market. (nmra.gov.lk)
4Recent Regulatory Changes
Between 2024 and 2026, the NMRA implemented several policy changes affecting pharmaceutical imports into Sri Lanka. Notably, in January 2026, the NMRA announced that import licenses expired from February 1, 2026, necessitating renewal for continued importation. This policy aims to ensure that all imported pharmaceuticals meet current regulatory standards and are subject to periodic review. Additionally, the NMRA has been actively updating its guidelines and procedures to align with international best practices, enhancing the overall regulatory framework governing pharmaceutical imports. (nmra.gov.lk)
State Pharmaceuticals Corporation O — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
State Pharmaceuticals Corporation O's product strategy focuses on a select range of therapeutic areas, including advanced diabetes medications, medical devices and diagnostics, and other categories. The corporation's top five imported products—Gliclazide, Diagnostic, Immunoglobulin, Rituximab, and a category labeled 'Other'—account for the entirety of its imports from India. This concentrated approach suggests a strategic emphasis on high-demand, critical healthcare products. The market demand for these products is driven by the prevalence of conditions such as diabetes and the need for advanced diagnostic and therapeutic solutions in the Sri Lankan healthcare system.
2Sourcing Profile
State Pharmaceuticals Corporation O's sourcing strategy is characterized by a focus on generic drugs, particularly in the therapeutic areas of advanced diabetes medications and medical devices and diagnostics. The corporation's imports from India are exclusively finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, rather than raw active pharmaceutical ingredients (APIs) or bulk drugs. This approach aligns with Sri Lanka's emphasis on affordable healthcare solutions and the promotion of generic drug use. India's established pharmaceutical manufacturing capabilities and adherence to international quality standards make it a preferred sourcing partner for State Pharmaceuticals Corporation O.
3Market Positioning
Based on its product mix, State Pharmaceuticals Corporation O serves a broad segment of the Sri Lankan market, encompassing both public and private healthcare sectors. Its role as the sole supplier to government institutions positions it as a critical provider for public hospitals and clinics. In the private sector, its 30% market share indicates a significant presence, supplying a wide range of pharmaceutical products to private healthcare providers, including hospitals, clinics, and pharmacies. This extensive reach underscores the corporation's pivotal role in ensuring the availability of essential medicines and healthcare products across the country.
Seller's Guide — How to Become a Supplier to State Pharmaceuticals Corporation O
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter the Sri Lankan market by partnering with State Pharmaceuticals Corporation O. The corporation's concentrated sourcing pattern suggests potential gaps in its current supplier base, particularly for products that align with its strategic focus areas. Indian exporters can capitalize on this by offering high-quality, cost-effective pharmaceutical formulations that meet the corporation's stringent regulatory and quality standards. Establishing strong, reliable partnerships and ensuring compliance with Sri Lankan regulatory requirements will be crucial for success in this market.
2Requirements & Qualifications
Indian exporters seeking to supply State Pharmaceuticals Corporation O and the Sri Lankan market must adhere to several key requirements.
Frequently Asked Questions — State Pharmaceuticals Corporation O
What products does State Pharmaceuticals Corporation O import from India?
State Pharmaceuticals Corporation O imports 4 pharmaceutical products across 4 categories. Top imports: Gliclazide ($450.0K), Diagnostic ($350.0K), Immunoglobulin ($200.0K), Rituximab ($150.0K).
Who supplies pharmaceuticals to State Pharmaceuticals Corporation O from India?
State Pharmaceuticals Corporation O sources from 53 verified Indian suppliers. The primary supplier is Wallace Pharmaceuticals Private Limited (7.4% of imports, $540.6K).
What is State Pharmaceuticals Corporation O's total pharmaceutical import value?
State Pharmaceuticals Corporation O's total pharmaceutical import value from India is $1.1M, based on 23 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does State Pharmaceuticals Corporation O focus on?
State Pharmaceuticals Corporation O imports across 4 categories. The largest: Advanced Diabetes Medications (39.1%), Medical Devices & Diagnostics (30.4%), Other (17.4%).
Get Full State Pharmaceuticals Corporation O Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: State Pharmaceuticals Corporation O identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as State Pharmaceuticals Corporation O's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 23 individual customs records matching State Pharmaceuticals Corporation O.
- 5.Supplier Verification: State Pharmaceuticals Corporation O sources from 53 verified Indian suppliers across 199 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.