Sepros Limited
Pharmaceutical Importer · Kenya · Antimalarial & Antiparasitic Focus · $109.5K Total Trade · DGFT Verified
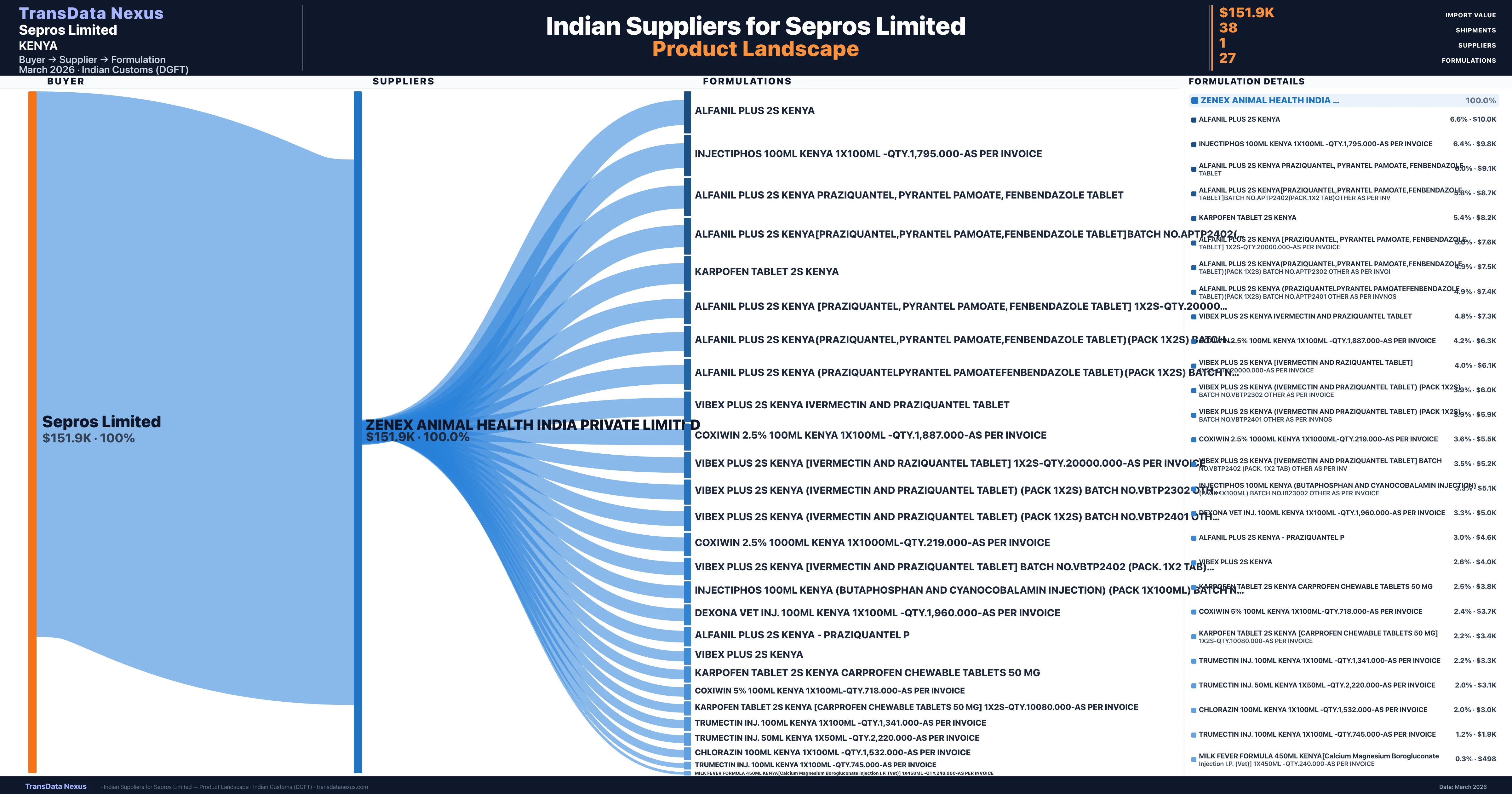
Sepros Limited is a pharmaceutical importer based in Kenya with a total trade value of $109.5K across 2 products in 1 therapeutic categories. Based on 19 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Sepros Limited sources from 1 verified Indian supplier, with Zenex Animal Health India Private Limited accounting for 100.0% of imports.
Sepros Limited — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Sepros Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Zenex Animal Health India Private Limited | $151.9K | 38 | 100.0% |
Sepros Limited sources from 1 verified Indian supplier across 27 distinct formulations. The sourcing is highly concentrated — Zenex Animal Health India Private Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Sepros Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Alfanil plus 2s | $10.0K | 1 |
| Injectiphos 100ML kenya 1x100ml -qty.1,795.000-as per | $9.8K | 2 |
| Alfanil plus 2s kenya praziquantel, pyrantel pamoate, fenbendazole | $9.1K | 1 |
| Alfanil plus 2s kenya[praziquantel,pyrantel pamoate,fenbendazole tablet]batch no.aptp2402(Pack.1x2 TAB)other as per | $8.7K | 3 |
| Karpofen tablet 2s | $8.2K | 1 |
| Alfanil plus 2s kenya [praziquantel, pyrantel pamoate, fenbendazole tablet] 1x2s-qty.20000.000-as per | $7.6K | 1 |
| Alfanil plus 2s kenya(praziquantel,pyrantel pamoate,fenbendazole tablet)(Pack 1x2s) batch no.aptp2302 other as per | $7.5K | 1 |
| Alfanil plus 2s kenya (praziquantelpyrantel pamoatefenbendazole tablet)(Pack 1x2s) batch no.aptp2401 other as per | $7.4K | 1 |
| Vibex plus 2s kenya ivermectin and praziquantel | $7.3K | 1 |
| Coxiwin 2.5% 100ML kenya 1x100ml -qty.1,887.000-as per | $6.3K | 2 |
| Vibex plus 2s kenya [ivermectin and raziquantel tablet] 1x2s-qty.20000.000-as per | $6.1K | 1 |
| Vibex plus 2s kenya (ivermectin and praziquantel tablet) (Pack 1x2s) batch no.vbtp2302 other as per | $6.0K | 1 |
| Vibex plus 2s kenya (ivermectin and praziquantel tablet) (Pack 1x2s) batch no.vbtp2401 other as per | $5.9K | 1 |
| Coxiwin 2.5% 1000ML kenya 1x1000ml-qty.219.000-as per | $5.5K | 2 |
| Vibex plus 2s kenya [ivermectin and praziquantel tablet] batch no.vbtp2402 (Pack. 1x2 TAB) other as per | $5.2K | 1 |
Sepros Limited imports 27 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Sepros Limited Import?
Top Products by Import Value
Sepros Limited Therapeutic Categories — 1 Specializations
Sepros Limited imports across 1 therapeutic categories, with Antimalarial & Antiparasitic (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antimalarial & Antiparasitic
2 products · 100.0% · $109.5K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Praziquantel | Antimalarial & Antiparasitic | $69.2K | 12 | 2.4% | 5 |
| 2 | Pyrantel | Antimalarial & Antiparasitic | $40.2K | 7 | 1.2% | 13 |
Sepros Limited imports 2 pharmaceutical products across 1 categories into Kenya totaling $109.5K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Sepros Limited.
Request DemoSepros Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Sepros Limited is a Kenyan-based company specializing in the importation and distribution of pharmaceutical products, particularly focusing on animal health and crop science solutions. Established in August 2000 by two veterinarians, the company began with a modest shop in Kiambu Town, aiming to serve the diverse needs of the local farming community. Over the years, Sepros Limited has expanded its operations, offering a broad portfolio of products achieved through partnerships with both local and overseas suppliers. (sepros.co.ke)
The company's mission is to provide distinctive and responsible service, contributing to the country's food security and enhancing the value placed on companion animals and plants. Sepros Limited delivers quality animal health and crop production solutions to resellers, practitioners, producers, and households. Their vision is to become the preferred supplier of these solutions through enhanced customer experience. (sepros.co.ke)
2Distribution Network
Sepros Limited operates from its main office located at Peflo Centre, Biashara Street, Kiambu Town, Kenya. The company also maintains a retail shop at Kenyatu House, Biashara Street, opposite Equity Bank, Kiambu Town. (sepros.co.ke) While specific details about their warehouse locations and logistics capabilities are not publicly disclosed, the company's extensive partnerships with local and international suppliers suggest a well-established distribution network. This network enables Sepros Limited to effectively serve the Kenyan market, ensuring timely availability and accessibility of their products across the country. (sepros.co.ke)
3Industry Role
Sepros Limited plays a significant role in Kenya's pharmaceutical supply chain, primarily acting as a wholesaler and distributor of pharmaceutical products. The company's focus on animal health and crop science solutions positions it as a key player in the agricultural sector, catering to the needs of farmers, veterinary practitioners, and agribusinesses. By sourcing products from both local and international suppliers, Sepros Limited ensures a diverse and reliable supply of pharmaceutical products, contributing to the overall health and productivity of the agricultural community in Kenya. (sepros.co.ke)
Supplier Relationship Intelligence — Sepros Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Sepros Limited's sourcing strategy exhibits a notable concentration, with a significant portion of its pharmaceutical imports originating from India. This concentration indicates a strategic choice to leverage India's established pharmaceutical manufacturing capabilities, particularly in the production of animal health and crop science products. The company's reliance on a single supplier, Zenex Animal Health India Private Limited, for a substantial number of shipments suggests a stable and long-term partnership. However, this dependency also presents potential risks, such as supply chain disruptions or regulatory changes in India that could impact product availability. To mitigate these risks, Sepros Limited may consider diversifying its supplier base and exploring alternative sourcing options to ensure a resilient supply chain.
2Supply Chain Resilience
The resilience of Sepros Limited's supply chain is closely tied to its sourcing practices. The company's reliance on a single supplier from India for a significant portion of its imports could pose challenges in the event of supply chain disruptions or regulatory changes. To enhance supply chain resilience, Sepros Limited could consider diversifying its supplier base by engaging with additional manufacturers from different countries. This approach would reduce dependency on a single source and provide alternative options in case of unforeseen disruptions. Additionally, ensuring that all suppliers adhere to recognized Good Manufacturing Practices (GMP) and maintaining compliance with Kenyan regulatory standards would further strengthen the supply chain's reliability and product quality.
3Strategic Implications
Sepros Limited's current sourcing pattern, characterized by a high dependency on a single supplier from India, has strategic implications for its competitive position in the Kenyan market. While this approach may offer cost advantages and streamlined logistics, it also exposes the company to risks associated with supply chain disruptions and regulatory changes in the supplier's country. For Indian exporters seeking to become alternative suppliers to Sepros Limited, understanding the company's reliance on a single source presents an opportunity to offer competitive pricing, reliable delivery schedules, and compliance with Kenyan regulatory standards. By addressing these factors, Indian exporters can position themselves as viable alternatives, potentially capturing a share of Sepros Limited's import requirements.
Importing Pharmaceuticals into Kenya — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Kenya
1Regulatory Authority & Framework
In Kenya, the regulation of pharmaceutical products is overseen by the Pharmacy and Poisons Board (PPB), which operates under the Ministry of Health. The PPB is responsible for ensuring that all pharmaceutical products imported, manufactured, and sold in Kenya meet the required standards of quality, safety, and efficacy. Key legislation governing pharmaceutical imports includes the Pharmacy and Poisons Act (Cap. 244) and the Pharmacy and Poisons (Registration of Health Products and Technologies) Rules. These regulations stipulate that no person shall import, manufacture for sale, or sell any drug in Kenya unless that drug has been registered and listed in accordance with the provisions of these Rules.
2Import Licensing & GMP
Importers of pharmaceutical products into Kenya are required to obtain an import license from the Pharmacy and Poisons Board. This license ensures that the importer complies with all regulatory requirements and that the imported products meet the necessary standards. Additionally, imported pharmaceutical products must adhere to recognized Good Manufacturing Practices (GMP). Certificates of GMP compliance, such as those issued by the World Health Organization (WHO) or the Pharmaceutical Inspection Co-operation Scheme (PIC/S), are essential for the registration and importation of pharmaceutical products into Kenya. Sepros Limited, as an importer, must ensure that its suppliers provide valid GMP certificates to facilitate the smooth importation and distribution of their products.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to confirm their quality and compliance with Kenyan standards. This process involves submitting samples to recognized laboratories for analysis and obtaining a certificate of analysis, which is a requirement for product registration. Stability requirements ensure that the products maintain their efficacy and safety throughout their shelf life. Labeling requirements include providing clear and accurate information in English and Kiswahili, the official languages of Kenya. This information should include the product name, active ingredients, dosage instructions, and expiry date. Serialization mandates may also be implemented to enhance traceability and prevent counterfeit products from entering the market. Sepros Limited must ensure that all imported products comply with these quality and labeling standards to meet regulatory requirements and maintain consumer trust.
4Recent Regulatory Changes
In January 2025, the Pharmacy and Poisons Board issued a public notice warning against the importation and sale of unregistered pharmaceutical products, particularly those referencing the Indian Pharmacopoeia. The notice emphasized that any pharmaceutical product not duly registered by the PPB is strictly prohibited for importation, distribution, sale, or handling in Kenya. This regulatory change underscores the importance of compliance with Kenyan standards and the necessity for importers like Sepros Limited to ensure that all products meet the required registration and quality standards. (pulse.co.ke)
Sepros Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Sepros Limited's focus on animal health and crop science products aligns with the significant demand in Kenya's agricultural sector. The country's reliance on agriculture for economic development and food security drives the need for effective veterinary medicines and crop protection solutions. By importing and distributing these products, Sepros Limited addresses critical needs in the market, supporting farmers and agribusinesses in enhancing productivity and maintaining animal health. The company's partnerships with both local and international suppliers enable it to offer a diverse range of products, catering to various customer requirements and preferences. (sepros.co.ke)
2Sourcing Profile
Sepros Limited's sourcing strategy emphasizes the importation of generic pharmaceutical products, particularly those related to animal health and crop science. The company's preference for finished pharmaceutical formulations, such as tablets, capsules, syrups, and injections, indicates a focus on ready-to-use products that meet the immediate needs of the Kenyan market. India's established pharmaceutical manufacturing industry, known for producing high-quality generic medicines, fits well into Sepros Limited's procurement strategy. By sourcing from India, the company leverages cost advantages and a reliable supply chain, ensuring the availability of essential products for its customers. (sepros.co.ke)
3Market Positioning
Based on its product mix, Sepros Limited primarily serves the agricultural sector in Kenya, including retail pharmacies, veterinary practitioners, and agribusinesses. The company's focus on animal health and crop science solutions positions it as a key supplier of veterinary medicines and crop protection products. By offering a diverse range of products and providing technical advice, Sepros Limited supports the health and productivity of animals and crops, contributing to the overall success of its customers in the agricultural industry. (sepros.co.ke)
Seller's Guide — How to Become a Supplier to Sepros Limited
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Sepros Limited, given the company's significant importation of pharmaceutical products from India. To capitalize on this opportunity, Indian exporters should focus on providing high-quality generic products that
Frequently Asked Questions — Sepros Limited
What products does Sepros Limited import from India?
Sepros Limited imports 2 pharmaceutical products across 1 categories. Top imports: Praziquantel ($69.2K), Pyrantel ($40.2K).
Who supplies pharmaceuticals to Sepros Limited from India?
Sepros Limited sources from 1 verified Indian suppliers. The primary supplier is Zenex Animal Health India Private Limited (100.0% of imports, $151.9K).
What is Sepros Limited's total pharmaceutical import value?
Sepros Limited's total pharmaceutical import value from India is $109.5K, based on 19 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Sepros Limited focus on?
Sepros Limited imports across 1 categories. The largest: Antimalarial & Antiparasitic (100.0%).
Get Full Sepros Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Sepros Limited identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Sepros Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 19 individual customs records matching Sepros Limited.
- 5.Supplier Verification: Sepros Limited sources from 1 verified Indian suppliers across 27 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.