Salutas Pharma Gmbh
Pharmaceutical Importer · Germany · Respiratory Focus · $5.3M Total Trade · DGFT Verified
Salutas Pharma Gmbh is a pharmaceutical importer based in Germany with a total trade value of $5.3M across 3 products in 2 therapeutic categories. Based on 129 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Salutas Pharma Gmbh sources from 7 verified Indian suppliers, with Cipla Limited accounting for 50.4% of imports.
Salutas Pharma Gmbh — Import Portfolio & Supplier Network
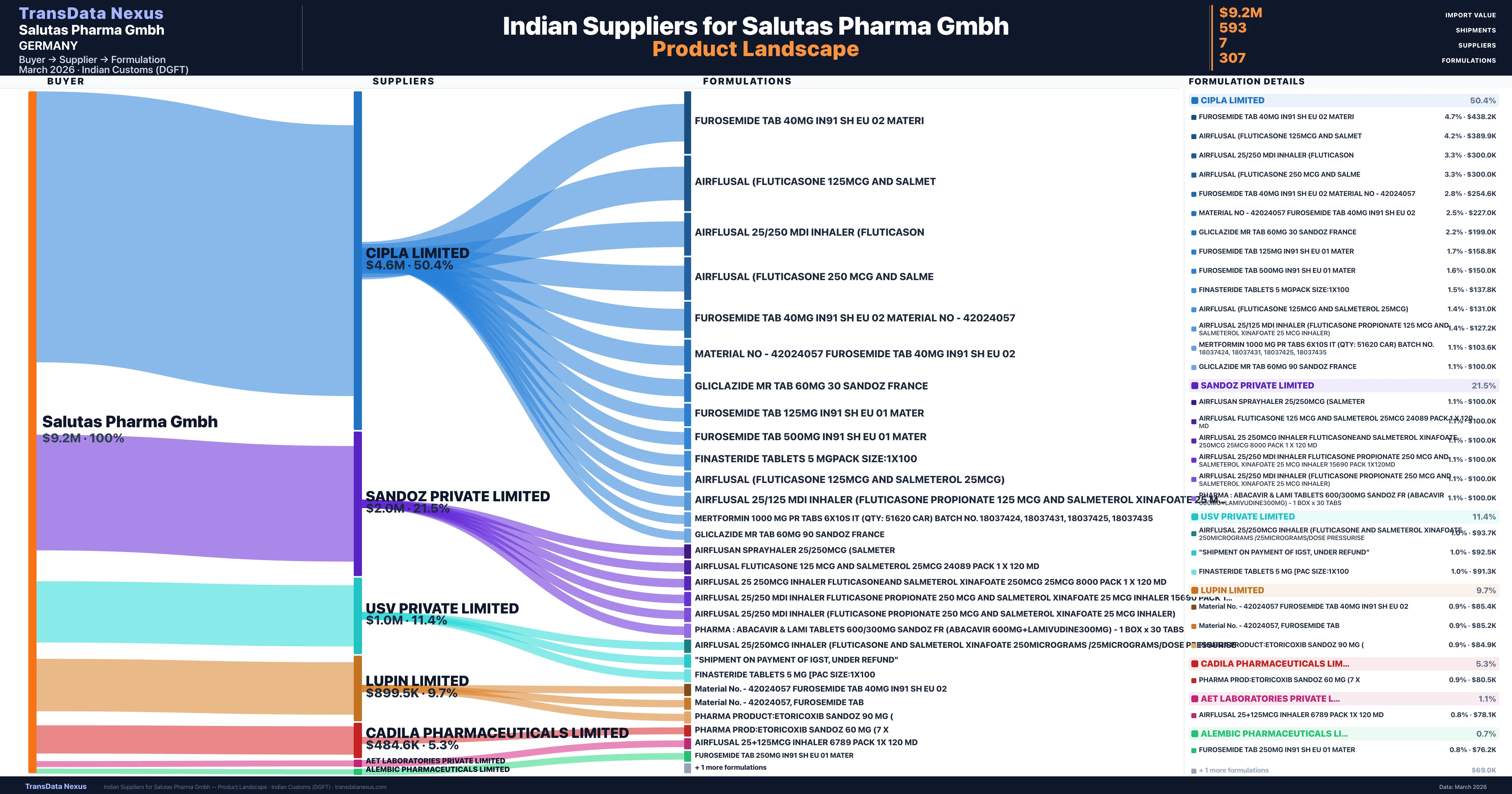
Who Are the Verified Indian Suppliers to Salutas Pharma Gmbh?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $4.6M | 385 | 50.4% |
| Sandoz Private Limited | $2.0M | 64 | 21.5% |
| Usv Private Limited | $1.0M | 60 | 11.4% |
| Lupin Limited | $899.5K | 24 | 9.7% |
| Cadila Pharmaceuticals Limited | $484.6K | 49 | 5.3% |
| Aet Laboratories Private Limited | $102.5K | 8 | 1.1% |
| Alembic Pharmaceuticals Limited | $61.4K | 3 | 0.7% |
Salutas Pharma Gmbh sources from 7 verified Indian suppliers across 307 distinct formulations. The supply base is diversified across 7 suppliers, reducing single-source dependency risk.
What Formulations Does Salutas Pharma Gmbh Import?
| Formulation | Value | Ships |
|---|---|---|
| Furosemide TAB 40MG in91 sh eu 02 | $438.2K | 10 |
| Airflusal (fluticasone 125mcg and | $389.9K | 8 |
| Airflusal 25/250 mdi inhaler | $300.0K | 6 |
| Airflusal (fluticasone 250 MCG and | $300.0K | 6 |
| Furosemide TAB 40MG in91 sh eu 02 material no - | $254.6K | 7 |
| Material no - 42024057 furosemide TAB 40MG in91 sh eu | $227.0K | 6 |
| Gliclazide mr TAB 60MG 30 sandoz | $199.0K | 4 |
| Furosemide TAB 125MG in91 sh eu 01 | $158.8K | 6 |
| Furosemide TAB 500MG in91 sh eu 01 | $150.0K | 3 |
| Finasteride tablets 5 mgpack | $137.8K | 6 |
| Airflusal (fluticasone 125mcg and salmeterol 25mcg) | $131.0K | 3 |
| Airflusal 25/125 mdi inhaler (fluticasone propionate 125 MCG and salmeterol xinafoate 25 MCG inhaler) | $127.2K | 3 |
| Mertformin 1000 MG pr tabs 6x10s it (qty: 51620 car) batch no. 18037424, 18037431, 18037425 | $103.6K | 3 |
| Gliclazide mr TAB 60MG 90 sandoz | $100.0K | 2 |
| Airflusan sprayhaler 25/250mcg | $100.0K | 2 |
Salutas Pharma Gmbh imports 307 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Salutas Pharma Gmbh Import?
Top Products by Import Value
Salutas Pharma Gmbh Therapeutic Categories — 2 Specializations
Salutas Pharma Gmbh imports across 2 therapeutic categories, with Respiratory (61.2%), Diuretics (38.8%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Respiratory
2 products · 61.2% · $3.3M
Diuretics
1 products · 38.8% · $2.1M
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Fluticasone | Respiratory | $2.1M | 42 | 1.0% | 8 |
| 2 | Furosemide | Diuretics | $2.1M | 64 | 2.9% | 5 |
| 3 | Salmeterol | Respiratory | $1.1M | 23 | 1.6% | 11 |
Salutas Pharma Gmbh imports 3 pharmaceutical products across 2 categories into Germany totaling $5.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Salutas Pharma Gmbh.
Request DemoSalutas Pharma Gmbh — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Salutas Pharma GmbH is a German pharmaceutical company specializing in the development, manufacturing, and distribution of pharmaceutical products. Established in 1993, the company operates as a subsidiary of Sandoz, which is part of the Novartis Group. Headquartered at Otto-von-Guericke-Allee 1, 39179 Barleben, Germany, Salutas Pharma GmbH employs approximately 1,500 individuals across its facilities in Barleben, Osterweddingen, and Magdeburg. (vev.icu) The company offers a diverse range of products, including ointments, creams, gels, and solid oral medications, with a notable focus on highly effective drugs for cancer therapy.
2Distribution Network
Salutas Pharma GmbH operates a robust distribution network within Germany and internationally. Its primary manufacturing and logistics centers are located in Barleben, Osterweddingen, and Magdeburg, which are among the most modern and efficient pharmaceutical production and logistics centers in Europe. (vev.icu) The company employs approximately 1,500 individuals across these facilities, indicating a substantial operational scale. While specific details about warehouse locations and logistics capabilities beyond Germany are not publicly disclosed, the company's extensive workforce and advanced facilities suggest a well-established distribution network capable of serving both domestic and international markets.
3Industry Role
Salutas Pharma GmbH plays a significant role in Germany's pharmaceutical supply chain as a manufacturer and distributor of pharmaceutical products. As a subsidiary of Sandoz, it contributes to the production and distribution of a wide range of pharmaceutical formulations, including generic drugs and specialized treatments. The company's focus on high-quality manufacturing and efficient distribution aligns with its role in ensuring the availability of essential medications within Germany and potentially in other European markets.
Supplier Relationship Intelligence — Salutas Pharma Gmbh
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Salutas Pharma GmbH's sourcing strategy exhibits a high degree of concentration, with a total import value from India amounting to $5.3 million USD across 129 shipments. The top five products imported—Fluticasone, Furosemide, and Salmeterol—account for 100% of the company's imports from India, indicating a complete reliance on these specific formulations. This sourcing pattern suggests a strategic focus on these products, likely due to their therapeutic importance and market demand. However, the lack of diversification in sourcing could pose risks related to supply chain disruptions, price fluctuations, or regulatory changes affecting these specific products.
2Supply Chain Resilience
Salutas Pharma GmbH's supply chain resilience is closely tied to its sourcing strategy from India. The company's complete reliance on a limited number of products from a single country introduces potential vulnerabilities, such as exposure to geopolitical risks, regulatory changes, or disruptions in manufacturing. The absence of backup suppliers for these specific products further amplifies this risk. Additionally, the company's focus on a narrow range of formulations may limit its ability to adapt to changing market demands or therapeutic advancements. To enhance supply chain resilience, diversifying the supplier base and expanding the product portfolio could be considered.
3Strategic Implications
Salutas Pharma GmbH's concentrated sourcing pattern positions it as a specialized player in the German pharmaceutical market, particularly in the respiratory and diuretics therapeutic categories. This focus allows the company to leverage expertise and potentially achieve economies of scale in these areas. For Indian exporters, this presents an opportunity to establish partnerships by offering high-quality formulations in these therapeutic categories. However, the limited diversification in Salutas Pharma GmbH's sourcing strategy may also indicate a preference for stability and predictability in supply chains, which could influence the company's openness to new suppliers.
Importing Pharmaceuticals into Germany — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Germany
1Regulatory Authority & Framework
In Germany, the primary regulatory authority overseeing pharmaceutical imports and marketing is the Federal Institute for Drugs and Medical Devices (BfArM). The key legislation governing pharmaceutical imports includes the German Medicines Act (AMG), which outlines the requirements for the import, distribution, and marketing of medicinal products. The marketing authorization pathway for Indian generics involves obtaining approval from BfArM, which assesses the quality, safety, and efficacy of the products. This process ensures that imported generics meet the stringent standards set for the German market.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products in Germany necessitate that manufacturers and importers hold a valid wholesale distribution authorization issued by the competent authority. Additionally, imported products must be accompanied by a Good Manufacturing Practice (GMP) certificate, which can be recognized from countries with equivalent standards, such as the European Union (EU) GMP, World Health Organization (WHO) GMP, or Pharmaceutical Inspection Co-operation Scheme (PIC/S). This ensures that imported pharmaceuticals adhere to the high-quality manufacturing standards required in Germany.
3Quality & Labeling
Pharmaceutical products imported into Germany must undergo batch testing to verify their quality and compliance with regulatory standards. Stability requirements are also enforced to ensure that products maintain their efficacy and safety throughout their shelf life. Labeling must be in German and include all necessary information, such as dosage instructions, side effects, and storage conditions. Serialization mandates are in place to facilitate traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Germany has implemented several regulatory changes affecting pharmaceutical imports. These include stricter requirements for GMP certification, enhanced scrutiny of import licenses, and updated labeling standards to improve patient safety and product traceability. Additionally, there has been an increased emphasis on sustainability and environmental considerations in pharmaceutical manufacturing and distribution. These changes aim to ensure that imported pharmaceuticals meet the highest standards of quality, safety, and environmental responsibility.
Salutas Pharma Gmbh — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Salutas Pharma GmbH's product strategy focuses on the respiratory and diuretics therapeutic categories, with imports primarily consisting of Fluticasone, Furosemide, and Salmeterol. This focus aligns with the company's expertise in manufacturing and distributing these specific formulations. The market demand for these products is driven by the prevalence of respiratory and cardiovascular conditions in Germany, necessitating effective treatments. By concentrating on these therapeutic areas, Salutas Pharma GmbH aims to address significant healthcare needs and maintain a competitive edge in the market.
2Sourcing Profile
Salutas Pharma GmbH's sourcing strategy is centered on importing generic formulations from India, particularly in the respiratory and diuretics categories. This approach leverages India's established pharmaceutical manufacturing capabilities and cost advantages. The company's preference for specific formulations indicates a strategic choice to maintain product quality and consistency. India's role as a key supplier aligns with Salutas Pharma GmbH's objectives to provide effective and affordable medications to the German market.
3Market Positioning
Based on its product mix, Salutas Pharma GmbH serves the wholesale distribution segment of the German pharmaceutical market. By focusing on specific therapeutic categories and maintaining a specialized product portfolio, the company positions itself as a key supplier to pharmacies, hospitals, and other healthcare providers requiring these medications. This strategic positioning allows Salutas Pharma GmbH to meet the needs of various healthcare institutions and contribute to the overall healthcare infrastructure in Germany.
Seller's Guide — How to Become a Supplier to Salutas Pharma Gmbh
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with Salutas Pharma GmbH, particularly those offering high-quality formulations in the respiratory and diuretics therapeutic categories. The company's concentrated sourcing pattern suggests a preference for stability and reliability in supply chains, which could be advantageous for suppliers who can consistently meet quality and regulatory standards. However, the limited diversification in Salutas Pharma GmbH's sourcing strategy may also indicate a cautious approach to introducing new suppliers, necessitating a strong value proposition and proven track record for potential partners.
2Requirements & Qualifications
Indian exporters seeking to supply Salutas Pharma GmbH and the German market must ensure that their products comply with EU GMP standards and possess valid GMP certificates recognized by German authorities. Additionally, obtaining a wholesale distribution authorization from the competent German authority is essential. Products must meet all quality, safety, and efficacy standards set by German regulations, including appropriate labeling in German and adherence to serialization mandates.
3How to Approach
To establish a partnership with Salutas Pharma GmbH, Indian exporters should focus on demonstrating consistent product quality, regulatory compliance, and the ability to meet the company's specific sourcing needs. Engaging in direct communication to understand Salutas Pharma GmbH's requirements and participating in relevant tenders can facilitate the relationship-building process. Developing a comprehensive regulatory filing strategy that aligns with German standards and preparing for a thorough evaluation process will be crucial. Setting realistic timelines for product approval and market entry, considering the regulatory procedures and quality assessments involved, will enhance the prospects of a successful partnership.
Frequently Asked Questions — Salutas Pharma Gmbh
What products does Salutas Pharma Gmbh import from India?
Salutas Pharma Gmbh imports 3 pharmaceutical products across 2 categories. Top imports: Fluticasone ($2.1M), Furosemide ($2.1M), Salmeterol ($1.1M).
Who supplies pharmaceuticals to Salutas Pharma Gmbh from India?
Salutas Pharma Gmbh sources from 7 verified Indian suppliers. The primary supplier is Cipla Limited (50.4% of imports, $4.6M).
What is Salutas Pharma Gmbh's total pharmaceutical import value?
Salutas Pharma Gmbh's total pharmaceutical import value from India is $5.3M, based on 129 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Salutas Pharma Gmbh focus on?
Salutas Pharma Gmbh imports across 2 categories. The largest: Respiratory (61.2%), Diuretics (38.8%).
Get Full Salutas Pharma Gmbh Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Salutas Pharma Gmbh identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Salutas Pharma Gmbh's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 129 individual customs records matching Salutas Pharma Gmbh.
- 5.Supplier Verification: Salutas Pharma Gmbh sources from 7 verified Indian suppliers across 307 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.