Salehiya Trading Company
Pharmaceutical Importer · Saudi Arabia · Advanced Antibiotics Focus · $3.3M Total Trade · DGFT Verified
Salehiya Trading Company is a pharmaceutical importer based in Saudi Arabia with a total trade value of $3.3M across 3 products in 3 therapeutic categories. Based on 119 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Salehiya Trading Company sources from 3 verified Indian suppliers, with Glenmark Pharmaceuticals Limited accounting for 66.8% of imports.
Salehiya Trading Company — Import Portfolio & Supplier Network
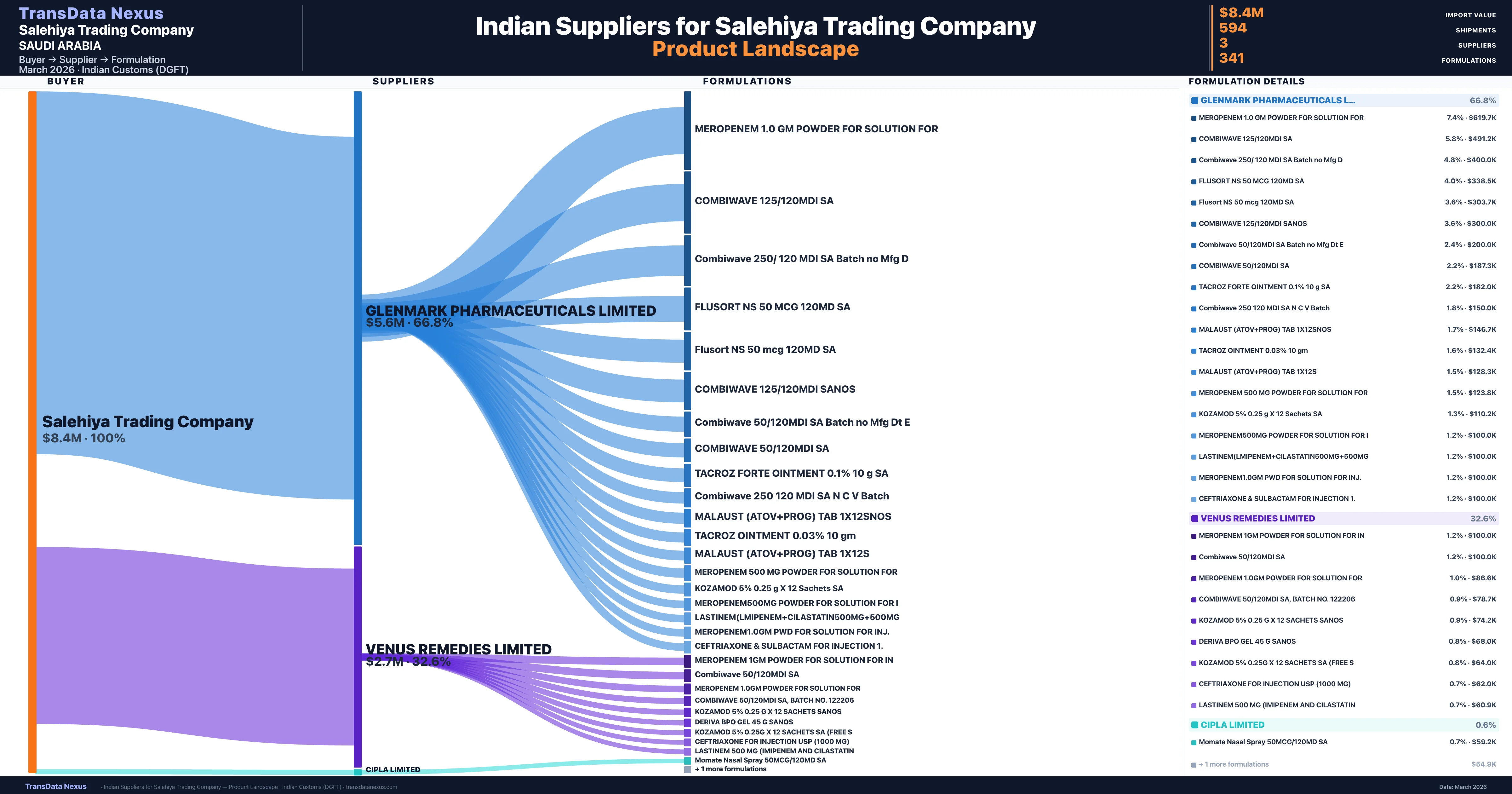
Who Are the Verified Indian Suppliers to Salehiya Trading Company?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Glenmark Pharmaceuticals Limited | $5.6M | 182 | 66.8% |
| Venus Remedies Limited | $2.7M | 399 | 32.6% |
| Cipla Limited | $50.0K | 13 | 0.6% |
Salehiya Trading Company sources from 3 verified Indian suppliers across 341 distinct formulations. Total import value: $8.4M across 594 shipments.
What Formulations Does Salehiya Trading Company Import?
| Formulation | Value | Ships |
|---|---|---|
| Meropenem 1.0 GM powder for solution | $619.7K | 32 |
| Combiwave 125/120mdi | $491.2K | 13 |
| Combiwave 250/ 120 mdi sa batch no mfg | $400.0K | 8 |
| Flusort ns 50 MCG 120md | $338.5K | 14 |
| Flusort ns 50 MCG 120md | $303.7K | 10 |
| Combiwave 125/120mdi | $300.0K | 6 |
| Combiwave 50/120mdi sa batch no mfg dt | $200.0K | 4 |
| Combiwave 50/120mdi | $187.3K | 4 |
| Tacroz forte ointment 0.1% 10 g | $182.0K | 4 |
| Combiwave 250 120 mdi sa n c v | $150.0K | 3 |
| Malaust (atov+prog) TAB | $146.7K | 3 |
| Tacroz ointment 0.03% 10 GM | $132.4K | 4 |
| Malaust (atov+prog) TAB 1x12s | $128.3K | 3 |
| Meropenem 500 MG powder for solution | $123.8K | 8 |
| Kozamod 5% 0.25 g x 12 sachets | $110.2K | 3 |
Salehiya Trading Company imports 341 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Salehiya Trading Company Import?
Top Products by Import Value
Salehiya Trading Company Therapeutic Categories — 3 Specializations
Salehiya Trading Company imports across 3 therapeutic categories, with Advanced Antibiotics (96.2%), Oncology (2.2%), Advanced Oncology (1.6%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
1 products · 96.2% · $3.2M
Oncology
1 products · 2.2% · $73.3K
Advanced Oncology
1 products · 1.6% · $52.4K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Meropenem | Advanced Antibiotics | $3.2M | 64 | 1.4% | 8 |
| 2 | Vincristine | Oncology | $73.3K | 36 | 3.6% | 3 |
| 3 | Dacarbazine | Advanced Oncology | $52.4K | 19 | 3.7% | 5 |
Salehiya Trading Company imports 3 pharmaceutical products across 3 categories into Saudi Arabia totaling $3.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Salehiya Trading Company.
Request DemoSalehiya Trading Company — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Salehiya Trading Company, established in 1964, is a prominent Saudi Arabian entity specializing in the distribution of pharmaceuticals, medical equipment, and patient and consumer care products. Headquartered in Riyadh, the company has expanded its operations to include branches in Jeddah, Dammam, Khamis Mushait, Qassim, and Medina Al-Munawarah, ensuring comprehensive coverage across the Kingdom.
As a private joint-stock company, Salehiya Trading Company plays a pivotal role in Saudi Arabia's pharmaceutical supply chain, acting as a primary wholesaler and distributor. The company collaborates with international partners to import and distribute a diverse range of pharmaceutical products, medical devices, and consumer health items, thereby enhancing the healthcare experience within the Kingdom.
2Distribution Network
Salehiya Trading Company operates an extensive distribution network with its headquarters in Riyadh and additional branches in Jeddah, Dammam, Khamis Mushait, Qassim, and Medina Al-Munawarah. This strategic placement enables the company to effectively serve clients across Saudi Arabia. The company has been certified by SGS for compliance with World Health Organization (WHO) Good Distribution Practices (GDP), establishing it as a leader in the warehousing and transportation of pharmaceutical products within the Kingdom.
3Industry Role
In Saudi Arabia's pharmaceutical supply chain, Salehiya Trading Company serves as a primary wholesaler and distributor. By importing and distributing a diverse range of pharmaceutical products, medical devices, and consumer health items, the company plays a crucial role in enhancing the healthcare experience within the Kingdom.
Supplier Relationship Intelligence — Salehiya Trading Company
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Salehiya Trading Company's sourcing strategy exhibits a high degree of concentration, with a total import value of $3.3 million USD from India across 119 shipments. The top five imported products—Meropenem, Vincristine, and Dacarbazine—account for 100% of the company's imports from India. This indicates a strategic focus on specific therapeutic areas, particularly advanced antibiotics and oncology treatments.
The company's reliance on a limited number of suppliers—GLENMARK PHARMACEUTICALS LIMITED, VENUS REMEDIES LIMITED, and CIPLA LIMITED—suggests a stable and possibly long-term relationship with these partners. However, this concentration also presents potential risks, such as supply chain disruptions or pricing volatility. Diversifying the supplier base could mitigate these risks and enhance supply chain resilience.
2Supply Chain Resilience
Salehiya Trading Company's supply chain resilience is closely tied to its partnerships with Indian suppliers. The company's focus on a limited number of suppliers for its top imported products indicates a stable relationship, which is beneficial for consistent product availability. However, this concentration also exposes the company to potential risks, such as supply chain disruptions or pricing volatility. Diversifying the supplier base could mitigate these risks and enhance supply chain resilience.
3Strategic Implications
Salehiya Trading Company's concentrated sourcing strategy positions it as a key player in the Saudi Arabian market for advanced antibiotics and oncology treatments. This focus allows the company to leverage specialized expertise and establish strong relationships with its suppliers. For Indian exporters, this presents an opportunity to strengthen existing partnerships and explore avenues for diversification within Salehiya's product portfolio. However, potential suppliers should be aware of the risks associated with high supplier concentration and consider strategies to mitigate these risks.
Importing Pharmaceuticals into Saudi Arabia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Saudi Arabia
1Regulatory Authority & Framework
The Saudi Food and Drug Authority (SFDA) is the primary regulatory body overseeing the pharmaceutical sector in Saudi Arabia. The SFDA is responsible for regulating the import, registration, and distribution of pharmaceutical products, ensuring they meet the Kingdom's health and safety standards. The Law of Pharmaceutical Establishments and Products (2023), issued under Royal Decree No. M/108, provides the legal framework governing medicinal products, herbal preparations, and pharmaceutical operations in the Kingdom.
2Import Licensing & GMP
Importing pharmaceutical products into Saudi Arabia requires compliance with specific licensing and certification requirements. A Certificate of Free Sale from the exporting country's Department of Health and Human Services is mandatory for all pharmaceutical shipments. Additionally, the Saudi Food and Drug Authority (SFDA) mandates that imported pharmaceutical products be registered and approved before entering the Saudi market. The SFDA also recognizes Good Manufacturing Practice (GMP) certifications from reputable international bodies, such as the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S), ensuring that imported products meet high-quality standards.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing and meet stability requirements as stipulated by the Saudi Food and Drug Authority (SFDA). Labeling must be in Arabic, including essential information such as product name, dosage form, strength, and usage instructions. Serialization mandates are enforced to enhance traceability and prevent counterfeit products from entering the market. These regulations ensure that pharmaceutical products in Saudi Arabia are safe, effective, and of high quality. (beta.sfda.gov.sa)
4Recent Regulatory Changes
Between 2024 and 2026, Saudi Arabia implemented several regulatory changes affecting pharmaceutical imports. The Law of Pharmaceutical Establishments and Products (2023), issued under Royal Decree No. M/108, provides a comprehensive legal framework for the regulation of medicinal products, herbal preparations, and pharmaceutical operations in the Kingdom. This law, enforced by the Saudi Food and Drug Authority (SFDA), establishes the SFDA's full regulatory authority across the lifecycle of medicinal products, covering manufacturing, importing, exporting, product registration, marketing authorization, distribution, retail operations, and advertising oversight.
Salehiya Trading Company — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Salehiya Trading Company's product strategy focuses on advanced antibiotics and oncology treatments, with Meropenem, Vincristine, and Dacarbazine being the top imported products. This focus aligns with the growing demand for specialized healthcare solutions in Saudi Arabia, driven by an aging population and increasing prevalence of chronic diseases. By concentrating on these therapeutic areas, Salehiya aims to address critical healthcare needs and strengthen its position in the market.
2Sourcing Profile
Salehiya Trading Company's sourcing strategy emphasizes importing finished pharmaceutical formulations from India, particularly in the categories of advanced antibiotics and oncology treatments. This approach leverages India's established pharmaceutical manufacturing capabilities and cost advantages. The company's focus on a limited number of suppliers suggests a strategic choice to maintain quality control and supplier reliability. However, this concentration also exposes the company to potential risks, such as supply chain disruptions or pricing volatility.
3Market Positioning
Salehiya Trading Company's focus on advanced antibiotics and oncology treatments positions it to serve the specialized segments of the Saudi Arabian pharmaceutical market. By importing and distributing these high-demand products, the company caters to hospitals, specialized clinics, and healthcare providers requiring advanced therapeutic solutions. This strategic positioning allows Salehiya to address critical healthcare needs and strengthen its presence in the market.
Seller's Guide — How to Become a Supplier to Salehiya Trading Company
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
For new Indian suppliers, there is a realistic opportunity to partner with Salehiya Trading Company, given the company's focus on advanced antibiotics and oncology treatments. Suppliers can explore avenues to diversify Salehiya's product portfolio within these therapeutic areas. However, potential suppliers should be aware of the risks associated with high supplier concentration and consider strategies to mitigate these risks.
2Requirements & Qualifications
Indian exporters seeking to supply Salehiya Trading Company and the Saudi Arabian market must ensure compliance with the Saudi Food and Drug Authority's (SFDA) regulations. This includes obtaining a Certificate of Free Sale from the exporting country's Department of Health and Human Services and ensuring that products meet SFDA's quality standards, including Good Manufacturing Practice (GMP) certifications from recognized international bodies. Additionally, products must be registered with the SFDA before importation.
3How to Approach
Indian exporters interested in supplying Salehiya Trading Company should initiate contact through the company's official channels, providing detailed product information and demonstrating compliance with SFDA regulations. Participating in tenders and engaging in direct communication can facilitate the establishment of a partnership. Understanding the regulatory requirements and ensuring timely product registration with the SFDA are crucial steps in the process. Establishing a relationship with Salehiya may involve navigating the competitive landscape, but with a focus on quality and compliance, Indian exporters can position themselves as valuable partners.
Frequently Asked Questions — Salehiya Trading Company
What products does Salehiya Trading Company import from India?
Salehiya Trading Company imports 3 pharmaceutical products across 3 categories. Top imports: Meropenem ($3.2M), Vincristine ($73.3K), Dacarbazine ($52.4K).
Who supplies pharmaceuticals to Salehiya Trading Company from India?
Salehiya Trading Company sources from 3 verified Indian suppliers. The primary supplier is Glenmark Pharmaceuticals Limited (66.8% of imports, $5.6M).
What is Salehiya Trading Company's total pharmaceutical import value?
Salehiya Trading Company's total pharmaceutical import value from India is $3.3M, based on 119 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Salehiya Trading Company focus on?
Salehiya Trading Company imports across 3 categories. The largest: Advanced Antibiotics (96.2%), Oncology (2.2%), Advanced Oncology (1.6%).
Get Full Salehiya Trading Company Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Salehiya Trading Company identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Salehiya Trading Company's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 119 individual customs records matching Salehiya Trading Company.
- 5.Supplier Verification: Salehiya Trading Company sources from 3 verified Indian suppliers across 341 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.