Saam Group Ltd
Pharmaceutical Importer · Afghanistan · Advanced Antibiotics Focus · $6.4M Total Trade · DGFT Verified
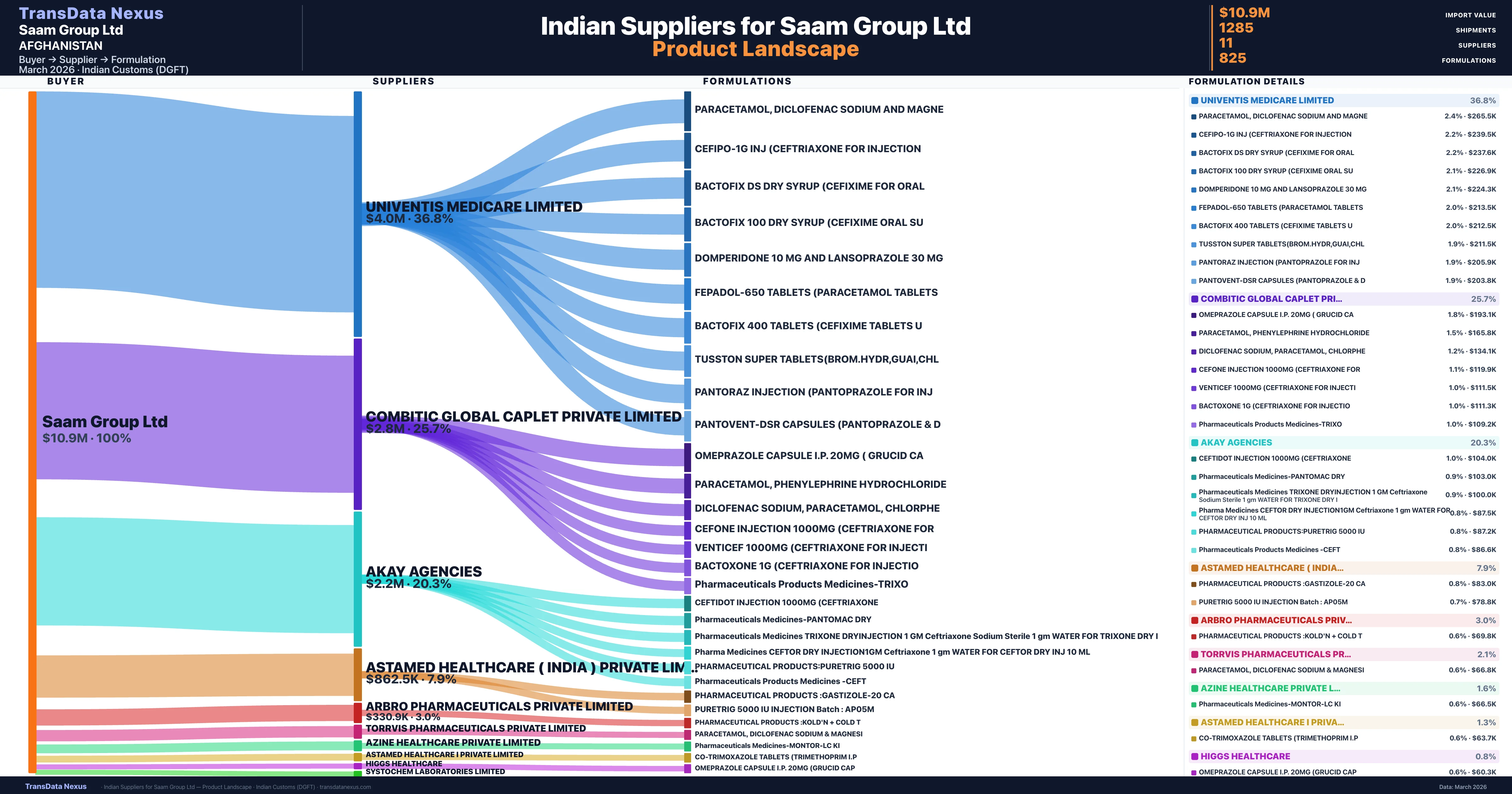
Saam Group Ltd is a pharmaceutical importer based in Afghanistan with a total trade value of $6.4M across 4 products in 3 therapeutic categories. Based on 145 verified import shipments from Indian Customs (DGFT) records, Saam Group Ltd is the #1 buyer in 1 product including Cefixime. Saam Group Ltd sources from 11 verified Indian suppliers, with Univentis Medicare Limited accounting for 36.8% of imports.
Saam Group Ltd — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Saam Group Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Univentis Medicare Limited | $4.0M | 329 | 36.8% |
| Combitic Global Caplet Private Limited | $2.8M | 303 | 25.7% |
| Akay Agencies | $2.2M | 277 | 20.3% |
| Astamed Healthcare ( India ) Private Limited | $862.5K | 239 | 7.9% |
| Arbro Pharmaceuticals Private Limited | $330.9K | 15 | 3.0% |
| Torrvis Pharmaceuticals Private Limited | $230.3K | 20 | 2.1% |
| Azine Healthcare Private Limited | $179.4K | 35 | 1.6% |
| Astamed Healthcare I Private Limited | $138.7K | 51 | 1.3% |
| Higgs Healthcare | $85.3K | 9 | 0.8% |
| Systochem Laboratories Limited | $38.4K | 3 | 0.4% |
| Amster Labs | $17.3K | 4 | 0.2% |
Saam Group Ltd sources from 11 verified Indian suppliers across 825 distinct formulations. The supply base is diversified across 11 suppliers, reducing single-source dependency risk.
What Formulations Does Saam Group Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Paracetamol, diclofenac sodium and | $265.5K | 16 |
| Cefipo-1g INJ (ceftriaxone for | $239.5K | 6 |
| Bactofix ds dry syrup (cefixime for | $237.6K | 8 |
| Bactofix 100 dry syrup (cefixime oral | $226.9K | 10 |
| Domperidone 10 MG and lansoprazole 30 MG | $224.3K | 9 |
| Fepadol-650 tablets (paracetamol | $213.5K | 6 |
| Bactofix 400 tablets (cefixime tablets | $212.5K | 6 |
| Tusston super tablets(brom.hydr,guai | $211.5K | 6 |
| Pantoraz injection (pantoprazole for INJ | $205.9K | 6 |
| Pantovent-dsr capsules (pantoprazole & | $203.8K | 6 |
| Omeprazole capsule i.p. 20MG ( grucid | $193.1K | 9 |
| Paracetamol, phenylephrine | $165.8K | 8 |
| Diclofenac sodium, paracetamol | $134.1K | 7 |
| Cefone injection 1000MG (ceftriaxone | $119.9K | 3 |
| Venticef 1000MG (ceftriaxone for | $111.5K | 3 |
Saam Group Ltd imports 825 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Saam Group Ltd Import?
Top Products by Import Value
Saam Group Ltd Therapeutic Categories — 3 Specializations
Saam Group Ltd imports across 3 therapeutic categories, with Advanced Antibiotics (94.3%), Gastrointestinal (4.7%), Combination Drugs (1.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
2 products · 94.3% · $6.0M
Gastrointestinal
1 products · 4.7% · $300.7K
Combination Drugs
1 products · 1.0% · $63.5K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ceftriaxone | Advanced Antibiotics | $3.6M | 73 | 1.3% | 2 |
| 2 | Cefixime | Advanced Antibiotics | $2.4M | 48 | 1.4% | 1 |
| 3 | Domperidone | Gastrointestinal | $300.7K | 18 | 1.0% | 13 |
| 4 | Paracetamol Caffeine | Combination Drugs | $63.5K | 6 | 4.6% | 2 |
Saam Group Ltd imports 4 pharmaceutical products across 3 categories into Afghanistan totaling $6.4M. The company is the #1 buyer for 1 product: Cefixime.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Saam Group Ltd.
Request DemoSaam Group Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Saam Group Ltd is a privately owned Afghan company established in 2008 and headquartered in Kabul. The company specializes in the importation and distribution of pharmaceutical products, medical equipment, and general supplies, serving both international and local organizations operating within Afghanistan. Over the years, Saam Group has expanded its operations from Kandahar to various provinces, including central bases such as KAF, BAF, Leatherneck, and Bastion, as well as minor bases (FOBs). Their clientele includes major contractors of the Afghan government and non-governmental organizations (NGOs).
In 2015, Saam Group extended its services internationally, engaging in import and export activities with countries like Turkey, China, Pakistan, and India. This expansion reflects the company's commitment to meeting the diverse needs of its clients by sourcing quality products from various international markets.
2Distribution Network
Saam Group Ltd operates a comprehensive distribution network across Afghanistan, with major logistical operations in Kabul and extended services to central and minor bases throughout the country. The company has established a robust supply chain to ensure timely delivery of pharmaceutical products and medical supplies to its diverse clientele. While specific warehouse locations are not publicly disclosed, Saam Group's extensive experience in logistics and transportation enables efficient distribution within Afghanistan.
3Industry Role
Saam Group Ltd functions primarily as a pharmaceutical importer and distributor within Afghanistan's supply chain. By sourcing a wide range of pharmaceutical products and medical equipment from international markets, including India, China, and Turkey, the company plays a crucial role in supplying essential medicines and medical supplies to both public and private sectors. Their clientele includes major contractors of the Afghan government and various NGOs, highlighting their significant presence in the Afghan pharmaceutical distribution landscape.
Supplier Relationship Intelligence — Saam Group Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Saam Group Ltd's sourcing strategy exhibits a notable concentration, with a significant portion of its pharmaceutical imports originating from India. This concentrated sourcing approach can be viewed as both a strategic choice and a potential risk. On one hand, it allows for streamlined procurement processes and potentially favorable pricing due to bulk purchasing. On the other hand, over-reliance on a single source country exposes the company to risks such as supply chain disruptions, geopolitical tensions, and regulatory changes in the supplier country. The shipment data indicates a stable relationship with Indian suppliers, as evidenced by the consistent volume of imports over the years. However, the lack of diversification in sourcing could pose challenges in the event of unforeseen disruptions in the Indian pharmaceutical market.
2Supply Chain Resilience
Saam Group Ltd's supply chain resilience is closely tied to its sourcing strategy. The company's heavy reliance on Indian suppliers for pharmaceutical imports means that any disruptions in India's pharmaceutical industry—such as regulatory changes, production issues, or logistical challenges—could significantly impact Saam Group's ability to meet the demands of its Afghan clientele. The absence of publicly available information regarding backup suppliers or alternative sourcing strategies further underscores the potential vulnerability of the supply chain. To enhance resilience, Saam Group may consider diversifying its supplier base and establishing contingency plans to mitigate risks associated with over-reliance on a single source country.
3Strategic Implications
Saam Group Ltd's concentrated sourcing pattern from India positions the company to benefit from established trade relationships and potentially favorable pricing. However, this strategy also exposes the company to risks associated with supply chain disruptions and geopolitical uncertainties. For Indian pharmaceutical exporters, Saam Group's significant import volume presents an opportunity to strengthen partnerships and explore long-term collaborations. Conversely, the lack of diversification in Saam Group's sourcing strategy may limit its flexibility in adapting to changes in the global pharmaceutical market.
Importing Pharmaceuticals into Afghanistan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Afghanistan
1Regulatory Authority & Framework
The Afghanistan Food and Drug Authority (AFDA) is the primary regulatory body overseeing the importation and commercialization of pharmaceutical products in Afghanistan. Established to address concerns related to the quality and safety of medicines entering the Afghan market, the AFDA is responsible for issuing licenses to pharmaceutical importers, ensuring compliance with national standards, and regulating the distribution of medicines. (afda.gov.af)
The legal framework governing pharmaceutical imports includes the National Licensed Medicines List, which specifies the medicines authorized for importation and commercialization in Afghanistan. This list is maintained by the General Directorate of Pharmacy Affairs (GDPA) under the Ministry of Public Health (MoPH). Importers must ensure that their products are included in this list to be eligible for importation. (platform.who.int)
2Import Licensing & GMP
To import pharmaceutical products into Afghanistan, companies must obtain an activity license from the GDPA and receive an import permit for each shipment. These licenses are granted based on compliance with laws and guidelines related to good manufacturing practices (GMP), distribution, and storage. Importers are required to provide documentation demonstrating that their products meet the quality standards set by the AFDA.
3Quality & Labeling
Imported pharmaceutical products must undergo quality control measures, including batch testing and stability assessments, to ensure they meet the standards set by the AFDA. Labeling requirements stipulate that product information be provided in the official languages of Afghanistan, with clear instructions for use, dosage, and storage. Serialization mandates may be implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, the Afghan government has implemented policy changes affecting pharmaceutical imports, including updates to the National Licensed Medicines List and stricter enforcement of GMP compliance. These changes aim to improve the quality and safety of medicines available in Afghanistan. Importers are required to stay informed about these regulatory updates to ensure continued compliance and uninterrupted market access.
Saam Group Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Saam Group Ltd's focus on importing advanced antibiotics, gastrointestinal medications, and combination drugs aligns with the therapeutic needs prevalent in Afghanistan. The substantial import value of antibiotics, particularly cefixime and ceftriaxone, indicates a significant demand for these treatments in the Afghan market. The inclusion of gastrointestinal and combination drugs in their portfolio suggests a strategic approach to addressing a broad spectrum of health conditions.
2Sourcing Profile
Saam Group Ltd's sourcing strategy emphasizes the procurement of generic pharmaceutical formulations, primarily in tablet and syrup forms, from Indian manufacturers. This approach allows the company to offer cost-effective solutions to its Afghan clientele while maintaining a focus on quality. The extensive range of formulations imported indicates a comprehensive approach to meeting diverse therapeutic needs.
3Market Positioning
Based on its product mix, Saam Group Ltd serves a wide segment of the Afghan pharmaceutical market, including retail pharmacies, hospitals, government tenders, and wholesale distribution. The company's ability to supply a diverse range of pharmaceutical products positions it as a key player in the Afghan healthcare supply chain.
Seller's Guide — How to Become a Supplier to Saam Group Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Saam Group Ltd, given the company's substantial import volume and focus on generic formulations. Potential gaps in Saam Group's current sourcing include the need for diversification in supplier relationships and the exploration of alternative sourcing strategies to enhance supply chain resilience.
2Requirements & Qualifications
Indian exporters seeking to supply Saam Group Ltd must ensure that their products are included in the National Licensed Medicines List of Afghanistan. Compliance with GMP standards recognized by the AFDA is essential, and exporters should be prepared to provide documentation demonstrating adherence to these standards. Additionally, products must meet labeling requirements, including information in the official languages of Afghanistan and adherence to serialization mandates.
3How to Approach
To establish a partnership with Saam Group Ltd, Indian exporters should initiate contact through formal channels, providing detailed product information and evidence of compliance with Afghan regulatory standards. Participating in tenders and responding to procurement opportunities issued by Saam Group can facilitate collaboration. Understanding the regulatory filing process and preparing for the necessary documentation and approvals will be crucial for a successful partnership.
Frequently Asked Questions — Saam Group Ltd
What products does Saam Group Ltd import from India?
Saam Group Ltd imports 4 pharmaceutical products across 3 categories. Top imports: Ceftriaxone ($3.6M), Cefixime ($2.4M), Domperidone ($300.7K), Paracetamol Caffeine ($63.5K).
Who supplies pharmaceuticals to Saam Group Ltd from India?
Saam Group Ltd sources from 11 verified Indian suppliers. The primary supplier is Univentis Medicare Limited (36.8% of imports, $4.0M).
What is Saam Group Ltd's total pharmaceutical import value?
Saam Group Ltd's total pharmaceutical import value from India is $6.4M, based on 145 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Saam Group Ltd focus on?
Saam Group Ltd imports across 3 categories. The largest: Advanced Antibiotics (94.3%), Gastrointestinal (4.7%), Combination Drugs (1.0%).
Get Full Saam Group Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Saam Group Ltd identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Saam Group Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 145 individual customs records matching Saam Group Ltd.
- 5.Supplier Verification: Saam Group Ltd sources from 11 verified Indian suppliers across 825 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.