Royal Pharma 2011 Limited
Pharmaceutical Importer · Uganda · Advanced Antibiotics Focus · $8.4M Total Trade · DGFT Verified
Royal Pharma 2011 Limited is a pharmaceutical importer based in Uganda with a total trade value of $8.4M across 2 products in 2 therapeutic categories. Based on 169 verified import shipments from Indian Customs (DGFT) records, Royal Pharma 2011 Limited is the #1 buyer in 1 product including Ceftriaxone. Royal Pharma 2011 Limited sources from 24 verified Indian suppliers, with Innova Captab Limited accounting for 50.8% of imports.
Royal Pharma 2011 Limited — Import Portfolio & Supplier Network
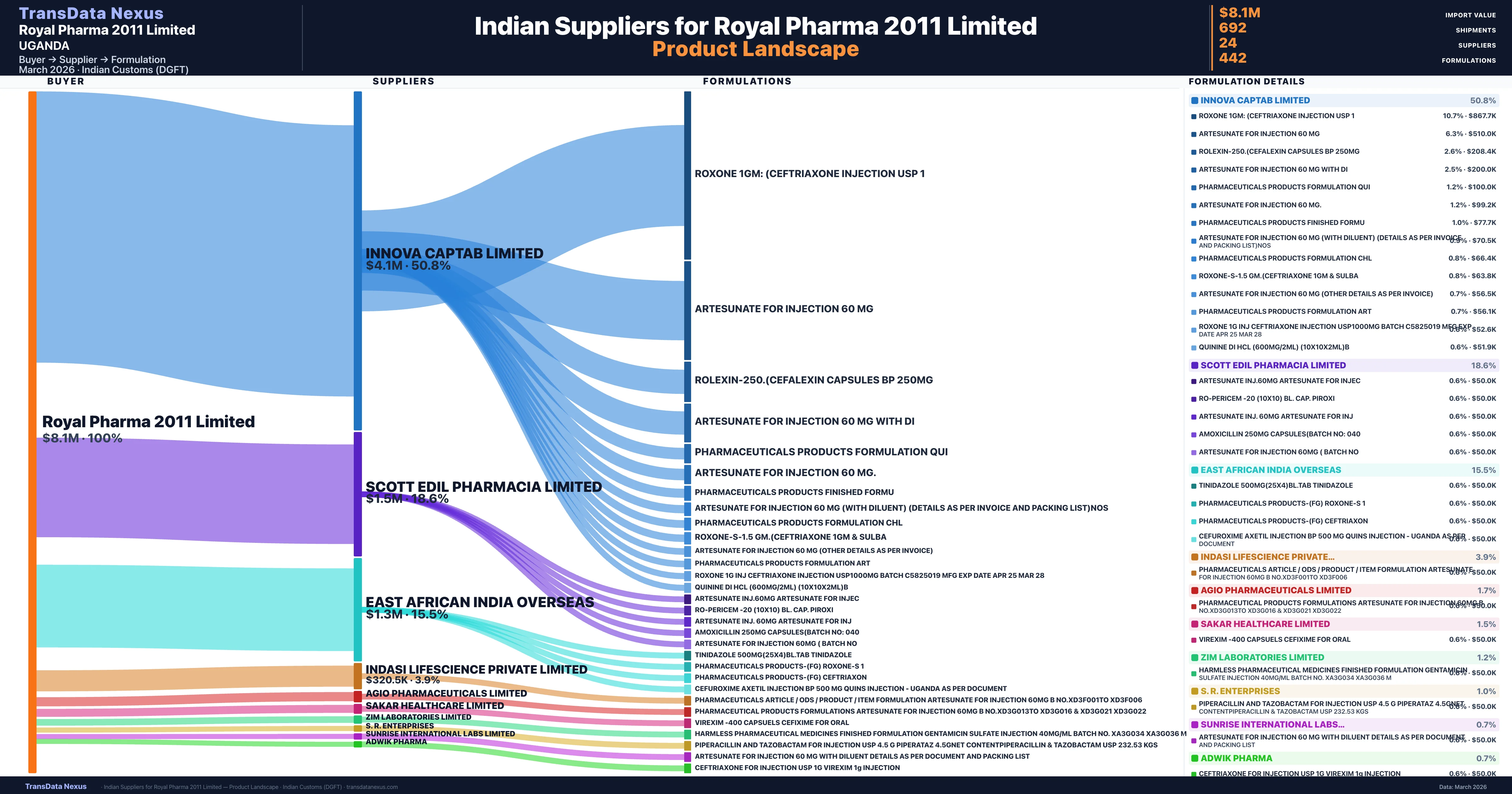
Who Are the Verified Indian Suppliers to Royal Pharma 2011 Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Innova Captab Limited | $4.1M | 456 | 50.8% |
| Scott Edil Pharmacia Limited | $1.5M | 67 | 18.6% |
| East African India Overseas | $1.3M | 63 | 15.5% |
| Indasi Lifescience Private Limited | $320.5K | 8 | 3.9% |
| Agio Pharmaceuticals Limited | $139.0K | 20 | 1.7% |
| Sakar Healthcare Limited | $121.0K | 7 | 1.5% |
| Zim Laboratories Limited | $100.9K | 6 | 1.2% |
| S. R. Enterprises | $78.2K | 8 | 1.0% |
| Sunrise International Labs Limited | $57.2K | 8 | 0.7% |
| Adwik Pharma | $54.4K | 2 | 0.7% |
| Uni Medicolabs | $50.0K | 2 | 0.6% |
| S R Enterprises | $49.7K | 3 | 0.6% |
| Globela Pharma Private Limited | $44.6K | 1 | 0.5% |
| Rainbow Life Sciences Private Limited | $34.4K | 2 | 0.4% |
| Ravian Life Science Private Limited | $33.7K | 12 | 0.4% |
| Oscar Medicare Private Limited | $25.1K | 5 | 0.3% |
| Msn Laboratories Private Limited | $24.0K | 2 | 0.3% |
| Adwik Pharma Private Limited | $22.8K | 3 | 0.3% |
| Alkem Laboratories Limited | $21.0K | 1 | 0.3% |
| Sun Rise International Labs Limited | $20.9K | 1 | 0.3% |
| Higgs Healthcare | $12.1K | 5 | 0.1% |
| Bliss Gvs Pharma Limited | $7.1K | 1 | 0.1% |
| Ipca Laboratories Limited | $5.0K | 8 | 0.1% |
| Inventia Healthcare Limited | $4.3K | 1 | 0.1% |
Royal Pharma 2011 Limited sources from 24 verified Indian suppliers across 442 distinct formulations. The supply base is diversified across 24 suppliers, reducing single-source dependency risk.
What Formulations Does Royal Pharma 2011 Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Roxone 1GM: (ceftriaxone injection USP | $867.7K | 21 |
| Artesunate for injection 60 MG | $510.0K | 15 |
| Rolexin-250.(cefalexin capsules BP 250MG | $208.4K | 18 |
| Artesunate for injection 60 MG with | $200.0K | 4 |
| Pharmaceuticals products formulation | $100.0K | 2 |
| Artesunate for injection 60 MG. | $99.2K | 2 |
| Pharmaceuticals products finished | $77.7K | 5 |
| Artesunate for injection 60 MG (with diluent) (details as per invoice and packing list | $70.5K | 3 |
| Pharmaceuticals products formulation | $66.4K | 3 |
| Roxone-s-1.5 GM.(ceftriaxone 1GM & | $63.8K | 12 |
| Artesunate for injection 60 MG (other details as per invoice) | $56.5K | 3 |
| Pharmaceuticals products formulation | $56.1K | 3 |
| Roxone 1g INJ ceftriaxone injection usp1000mg batch c5825019 mfg exp date apr 25 mar | $52.6K | 2 |
| Quinine di hcl (600MG/2ML) (10x10x2ml | $51.9K | 2 |
| Artesunate INJ.60MG artesunate for | $50.0K | 1 |
Royal Pharma 2011 Limited imports 442 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Royal Pharma 2011 Limited Import?
Top Products by Import Value
Royal Pharma 2011 Limited Therapeutic Categories — 2 Specializations
Royal Pharma 2011 Limited imports across 2 therapeutic categories, with Advanced Antibiotics (79.9%), Antimalarial & Antiparasitic (20.1%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
1 products · 79.9% · $6.8M
Antimalarial & Antiparasitic
1 products · 20.1% · $1.7M
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ceftriaxone | Advanced Antibiotics | $6.8M | 135 | 2.4% | 1 |
| 2 | Artesunate | Antimalarial & Antiparasitic | $1.7M | 34 | 1.0% | 8 |
Royal Pharma 2011 Limited imports 2 pharmaceutical products across 2 categories into Uganda totaling $8.4M. The company is the #1 buyer for 1 product: Ceftriaxone.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Royal Pharma 2011 Limited.
Request DemoRoyal Pharma 2011 Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Royal Pharma 2011 Limited is a prominent pharmaceutical importer and distributor based in Kampala, Uganda. Established on July 11, 2011, the company has expanded its operations to include multiple branches across Uganda, including locations in Lira and various districts in Kampala. The founders, Mr. Karim Nisarali Tharani and Mr. Saleem Shabuddin, have guided the company to become a key player in Uganda's pharmaceutical sector.
The company's headquarters are situated at Plot 10, Jesco Plaza, Wilson Road, P.O. Box 7429, Kampala, Uganda. Royal Pharma 2011 Limited specializes in the wholesale and retail distribution of a wide range of pharmaceutical products, including surgical instruments, medical disposables, and medical drugs that adhere to international standards. Their clientele includes over 2,500 corporate accounts and more than 5,000 other clients, such as retail pharmacies, wholesale pharmacies, drug shops, government hospitals, private hospitals, clinics, NGOs, schools, colleges, and universities.
2Distribution Network
Royal Pharma 2011 Limited operates an extensive distribution network across Uganda, with multiple branches strategically located to serve both urban and rural areas. In Kampala, the company has established several outlets, including those at Wilson Road, William Street, Nateete, and Ben Kiwanuka Street. Additionally, the company has expanded its reach to the northern region with a branch in Lira. This widespread presence enables Royal Pharma 2011 Limited to effectively distribute pharmaceutical products throughout the country.
3Industry Role
Royal Pharma 2011 Limited plays a multifaceted role in Uganda's pharmaceutical supply chain. As a primary wholesaler and importer, the company sources pharmaceutical products from various international suppliers, including Indian manufacturers. It also engages in retail distribution through its network of branches, serving a diverse clientele that includes government hospitals, private clinics, NGOs, and educational institutions. This comprehensive approach positions Royal Pharma 2011 Limited as a significant contributor to the accessibility and distribution of pharmaceutical products in Uganda.
Supplier Relationship Intelligence — Royal Pharma 2011 Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Royal Pharma 2011 Limited's sourcing strategy exhibits a high degree of concentration, particularly in its imports from India. The company has imported a total of $8.4 million USD worth of pharmaceutical products from India across 169 shipments, focusing on two primary therapeutic categories: Advanced Antibiotics and Antimalarial & Antiparasitic. The top five products imported include Ceftriaxone ($6.8 million, 2.4% share) and Artesunate ($1.7 million, 1.0% share), indicating a strategic emphasis on these medications. The reliance on a limited number of products and suppliers suggests a deliberate sourcing strategy aimed at ensuring product availability and quality. However, this concentration also introduces potential risks related to supply chain disruptions and dependency on specific suppliers.
2Supply Chain Resilience
The resilience of Royal Pharma 2011 Limited's supply chain is closely tied to its sourcing practices. With a portfolio concentration where the top five products account for 100% of imports, the company faces challenges in diversifying its product range. The heavy reliance on a select group of suppliers, particularly from India, may expose the company to risks associated with geopolitical tensions, regulatory changes, or logistical disruptions affecting these suppliers. To enhance supply chain resilience, it would be prudent for Royal Pharma 2011 Limited to explore alternative suppliers and diversify its product offerings, thereby mitigating potential risks and ensuring a more robust supply chain.
3Strategic Implications
Royal Pharma 2011 Limited's concentrated sourcing pattern positions it as a dominant player in the Ugandan market for specific pharmaceutical products, particularly Advanced Antibiotics and Antimalarial & Antiparasitic medications. This strategic focus allows the company to leverage economies of scale and establish strong relationships with key suppliers. For Indian exporters, this presents an opportunity to become alternative suppliers by offering competitive pricing, reliable delivery schedules, and adherence to quality standards. Diversifying the supplier base can also provide Royal Pharma 2011 Limited with leverage to negotiate better terms and reduce dependency on a limited number of suppliers.
Importing Pharmaceuticals into Uganda — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Uganda
1Regulatory Authority & Framework
The National Drug Authority (NDA) is the primary regulatory body overseeing the pharmaceutical sector in Uganda. Established under the National Drug Policy and Authority Act (Cap 206), the NDA is responsible for regulating the manufacture, importation, exportation, sale, and supply of drugs to ensure they meet consistent high standards of quality, safety, and efficacy. The NDA's mandate includes the creation and maintenance of a national list of essential drugs, which is revised periodically to reflect the country's healthcare needs. Additionally, the NDA provides guidelines for the importation and exportation of drugs, ensuring that all pharmaceutical products entering the Ugandan market comply with established standards.
2Import Licensing & GMP
To import pharmaceutical products into Uganda, entities must obtain an import license from the National Drug Authority (NDA). This license is valid for one year and is only effective within the calendar year in which it is issued. Applicants must also hold a valid license to operate a pharmacy. Before importing each consignment, a licensed importer must apply to the NDA for a Verification Certificate, ensuring that the products meet the required standards. Regarding Good Manufacturing Practice (GMP), the NDA requires that foreign manufacturing sites pass an inspection prior to the approval of the marketing authorization application. This inspection assesses compliance with global regulatory requirements, including those of the World Health Organization (WHO). The NDA's Directorate of Inspectorate & Enforcement Services is responsible for inspecting and authorizing manufacturers of various pharmaceutical products, ensuring that medicines supplied in Uganda meet consistent high standards of quality, safety, and efficacy. (nda.or.ug)
3Quality & Labeling
Pharmaceutical products imported into Uganda must undergo batch testing to ensure they meet the required quality standards. The National Drug Authority (NDA) conducts inspections and sampling at ports of entry to verify the quality, safety, and efficacy of imported drugs. Stability requirements are also enforced to ensure that products maintain their intended quality throughout their shelf life. Labeling must comply with NDA regulations, which include requirements for labeling language, typically English, and may include other local languages. Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the market. These measures are part of the NDA's comprehensive approach to maintaining high standards in the pharmaceutical supply chain. (nda.or.ug)
4Recent Regulatory Changes
Between 2024 and 2026, the National Drug Authority (NDA) has implemented several policy changes affecting pharmaceutical imports into Uganda. These include stricter enforcement of import licensing requirements, enhanced inspection protocols at ports of entry, and updated guidelines for the verification of applications for importation of drugs. The NDA has also revised the national list of essential drugs to better align with the country's evolving healthcare needs. These regulatory changes aim to improve the quality, safety, and efficacy of pharmaceutical products available in Uganda, ensuring that they meet international standards. (nda.or.ug)
Royal Pharma 2011 Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Royal Pharma 2011 Limited's product strategy focuses on two primary therapeutic areas: Advanced Antibiotics and Antimalarial & Antiparasitic medications. The company's imports are heavily concentrated in these categories, with the top five products accounting for 100% of its imports. This strategic focus is likely driven by the high demand for these medications in Uganda, where infectious diseases such as bacterial infections and malaria are prevalent. By concentrating on these therapeutic areas, Royal Pharma 2011 Limited aims to address critical healthcare needs and establish a strong market presence in these segments.
2Sourcing Profile
Royal Pharma 2011 Limited's sourcing strategy is heavily reliant on imports from India, with a total import value of $8.4 million USD across 169 shipments. The company imports a total of 442 unique pharmaceutical formulations, indicating a diverse product range within its focus areas. This sourcing strategy allows Royal Pharma 2011 Limited to leverage India's manufacturing capabilities and cost advantages while ensuring a steady supply of essential medications. However, the concentration of imports from a single country may expose the company to risks associated with geopolitical tensions, regulatory changes, or logistical disruptions affecting Indian suppliers.
3Market Positioning
Based on its product mix, Royal Pharma 2011 Limited primarily serves the wholesale distribution segment of the Ugandan pharmaceutical market. The company's focus on Advanced Antibiotics and Antimalarial & Antiparasitic medications positions it to meet the needs of government hospitals, private clinics, NGOs, and educational institutions. By concentrating on these therapeutic areas, Royal Pharma 2011 Limited addresses critical healthcare needs and establishes a strong market presence in these segments. The company's extensive distribution network further enhances its ability to serve a wide range of clients across Uganda.
Frequently Asked Questions — Royal Pharma 2011 Limited
What products does Royal Pharma 2011 Limited import from India?
Royal Pharma 2011 Limited imports 2 pharmaceutical products across 2 categories. Top imports: Ceftriaxone ($6.8M), Artesunate ($1.7M).
Who supplies pharmaceuticals to Royal Pharma 2011 Limited from India?
Royal Pharma 2011 Limited sources from 24 verified Indian suppliers. The primary supplier is Innova Captab Limited (50.8% of imports, $4.1M).
What is Royal Pharma 2011 Limited's total pharmaceutical import value?
Royal Pharma 2011 Limited's total pharmaceutical import value from India is $8.4M, based on 169 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Royal Pharma 2011 Limited focus on?
Royal Pharma 2011 Limited imports across 2 categories. The largest: Advanced Antibiotics (79.9%), Antimalarial & Antiparasitic (20.1%).
Get Full Royal Pharma 2011 Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Royal Pharma 2011 Limited identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Royal Pharma 2011 Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 169 individual customs records matching Royal Pharma 2011 Limited.
- 5.Supplier Verification: Royal Pharma 2011 Limited sources from 24 verified Indian suppliers across 442 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.