Ranbaxy Farmaceutica Ltda :
Pharmaceutical Importer · Brazil · Advanced Diabetes Medications Focus · $5.8M Total Trade · DGFT Verified
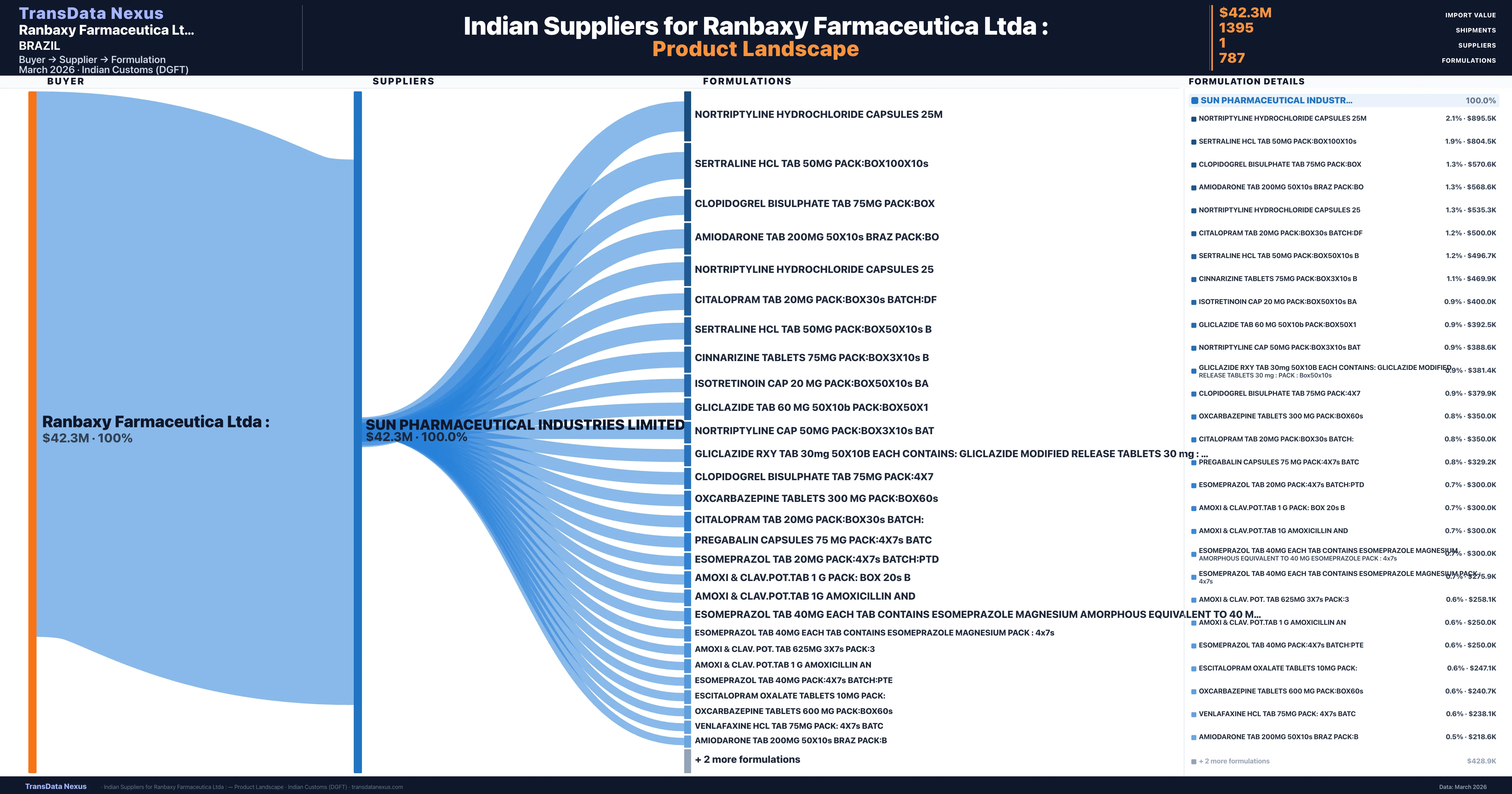
Ranbaxy Farmaceutica Ltda : is a pharmaceutical importer based in Brazil with a total trade value of $5.8M across 3 products in 3 therapeutic categories. Based on 146 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Ranbaxy Farmaceutica Ltda : sources from 1 verified Indian supplier, with Sun Pharmaceutical Industries Limited accounting for 100.0% of imports.
Ranbaxy Farmaceutica Ltda : — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Ranbaxy Farmaceutica Ltda :?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Sun Pharmaceutical Industries Limited | $42.3M | 1,395 | 100.0% |
Ranbaxy Farmaceutica Ltda : sources from 1 verified Indian supplier across 787 distinct formulations. The sourcing is highly concentrated — Sun Pharmaceutical Industries Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Ranbaxy Farmaceutica Ltda : Import?
| Formulation | Value | Ships |
|---|---|---|
| Nortriptyline hydrochloride capsules | $895.5K | 22 |
| Sertraline hcl TAB 50MG Pack:box100x10s | $804.5K | 17 |
| Clopidogrel bisulphate TAB 75MG | $570.6K | 13 |
| Amiodarone TAB 200MG 50x10s braz | $568.6K | 13 |
| Nortriptyline hydrochloride capsules | $535.3K | 11 |
| Citalopram TAB 20MG Pack:box30s | $500.0K | 10 |
| Sertraline hcl TAB 50MG Pack:box50x10s | $496.7K | 16 |
| Cinnarizine tablets 75MG Pack:box3x10s | $469.9K | 10 |
| Isotretinoin cap 20 MG Pack:box50x10s | $400.0K | 8 |
| Gliclazide TAB 60 MG 50x10b | $392.5K | 8 |
| Nortriptyline cap 50MG Pack:box3x10s | $388.6K | 8 |
| Gliclazide rxy TAB 30MG 50x10b Each contains: gliclazide modified release tablets 30 MG : Pack : box50x10s | $381.4K | 10 |
| Clopidogrel bisulphate TAB 75MG | $379.9K | 8 |
| Oxcarbazepine tablets 300 MG Pack:box60s | $350.0K | 7 |
| Citalopram TAB 20MG Pack:box30s | $350.0K | 7 |
Ranbaxy Farmaceutica Ltda : imports 787 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Ranbaxy Farmaceutica Ltda : Import?
Top Products by Import Value
Ranbaxy Farmaceutica Ltda : Therapeutic Categories — 3 Specializations
Ranbaxy Farmaceutica Ltda : imports across 3 therapeutic categories, with Advanced Diabetes Medications (39.3%), Vitamins & Supplements (32.1%), Nutritional Supplements (28.6%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Diabetes Medications
1 products · 39.3% · $2.3M
Vitamins & Supplements
1 products · 32.1% · $1.9M
Nutritional Supplements
1 products · 28.6% · $1.6M
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Gliclazide | Advanced Diabetes Medications | $2.3M | 76 | 0.8% | 9 |
| 2 | Magnesium | Vitamins & Supplements | $1.9M | 37 | 0.5% | 16 |
| 3 | Sodium | Nutritional Supplements | $1.6M | 33 | 0.5% | 7 |
Ranbaxy Farmaceutica Ltda : imports 3 pharmaceutical products across 3 categories into Brazil totaling $5.8M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Ranbaxy Farmaceutica Ltda :.
Request DemoRanbaxy Farmaceutica Ltda : — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Ranbaxy Farmaceutica Ltda. is a Brazilian pharmaceutical company specializing in the wholesale distribution of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. Established on November 4, 1993, the company is headquartered in Rio de Janeiro, Brazil. It operates under the legal structure of a Sociedade Empresária Limitada (Limited Liability Company), with a capital social of R$ 17,366,914.00.
The company's primary activity is the wholesale distribution of human-use pharmaceuticals, as classified under the CNAE code 46.44-3/01. Ranbaxy Farmaceutica Ltda. has expanded its operations with additional facilities in Barueri, São Paulo, and Goiânia, Goiás, indicating a strategic approach to enhance its distribution capabilities across Brazil. (econodata.com.br)
2Distribution Network
Ranbaxy Farmaceutica Ltda. has established a robust distribution network with multiple facilities across Brazil. The main office is located at Rua Francisco de Souza Melo, 252, Cordovil, Rio de Janeiro, RJ, 21.010-410. Additional facilities include a branch in Barueri, São Paulo, situated at Alameda Tocantins, 125, Alphaville Industrial, and another in Goiânia, Goiás, at Avenida Doutor Napoleão R. Laureano, 2821, Fazenda Planície. (econodata.com.br)
These strategically located warehouses enable Ranbaxy Farmaceutica Ltda. to effectively manage logistics and ensure timely delivery of pharmaceutical products throughout Brazil. The company's distribution network is designed to support its extensive import operations, facilitating the efficient movement of goods from suppliers to end-users across the country.
3Industry Role
Ranbaxy Farmaceutica Ltda. plays a significant role in Brazil's pharmaceutical supply chain as a primary wholesaler. The company imports a diverse range of finished pharmaceutical formulations from India, including advanced diabetes medications, vitamins, and nutritional supplements. By sourcing these products, Ranbaxy Farmaceutica Ltda. contributes to the availability of essential medications in the Brazilian market, catering to various therapeutic needs.
The company's operations are characterized by a high degree of supplier concentration, primarily importing from a single supplier, SUN PHARMACEUTICAL INDUSTRIES LIMITED. This sourcing strategy underscores Ranbaxy Farmaceutica Ltda.'s reliance on established partnerships to meet the pharmaceutical demands of the Brazilian market.
Supplier Relationship Intelligence — Ranbaxy Farmaceutica Ltda :
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Ranbaxy Farmaceutica Ltda.'s sourcing strategy exhibits a high degree of concentration, with 100% of its pharmaceutical imports originating from SUN PHARMACEUTICAL INDUSTRIES LIMITED. This exclusive partnership indicates a strategic choice to maintain a streamlined supply chain and potentially negotiate favorable terms.
However, such dependency on a single supplier introduces risks related to supply chain disruptions, price fluctuations, and potential regulatory changes affecting the supplier. The stability of this relationship is crucial for Ranbaxy Farmaceutica Ltda.'s operations, and any challenges faced by SUN PHARMACEUTICAL INDUSTRIES LIMITED could directly impact the company's ability to meet market demands.
2Supply Chain Resilience
The resilience of Ranbaxy Farmaceutica Ltda.'s supply chain is closely tied to the stability and reliability of SUN PHARMACEUTICAL INDUSTRIES LIMITED. Given the exclusive nature of this partnership, the company may face challenges in diversifying its supplier base. The absence of backup suppliers could pose risks in the event of supply chain disruptions, such as production delays or logistical issues.
Additionally, the company's focus on a limited range of formulations, with 787 unique formulations imported, suggests a specialized product portfolio. While this specialization can enhance operational efficiency, it may also limit flexibility in adapting to changing market demands or regulatory requirements.
3Strategic Implications
Ranbaxy Farmaceutica Ltda.'s concentrated sourcing strategy positions it to leverage strong relationships with its supplier, potentially securing favorable pricing and consistent product quality. This approach can enhance the company's competitive edge in the Brazilian pharmaceutical market.
For Indian exporters, the exclusive partnership with SUN PHARMACEUTICAL INDUSTRIES LIMITED presents an opportunity to explore alternative supply channels to Ranbaxy Farmaceutica Ltda. Diversifying the supplier base could mitigate risks associated with supply chain disruptions and open avenues for new business relationships.
Importing Pharmaceuticals into Brazil — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Brazil
1Regulatory Authority & Framework
In Brazil, the regulation of pharmaceutical products is overseen by the Agência Nacional de Vigilância Sanitária (ANVISA), the National Health Surveillance Agency. ANVISA is responsible for ensuring the safety, efficacy, and quality of health-related products, including pharmaceuticals. (gov.br)
Key legislation governing pharmaceutical imports includes Law No. 9,782/1999, which established ANVISA, and Law No. 6,360/1976, which regulates the manufacture and marketing of pharmaceutical products. These laws outline the requirements for product registration, importation, and distribution within Brazil.
2Import Licensing & GMP
Import licensing in Brazil requires that pharmaceutical products be registered with ANVISA before they can be marketed. This process involves submitting detailed documentation, including evidence of Good Manufacturing Practice (GMP) compliance. ANVISA recognizes GMP certifications from various international standards, such as EU GMP, WHO GMP, and PIC/S, facilitating the importation of products manufactured under these guidelines. (gov.br)
Wholesalers like Ranbaxy Farmaceutica Ltda. must obtain authorization from ANVISA to distribute pharmaceutical products. This authorization ensures that the company adheres to regulatory standards concerning storage, handling, and distribution, maintaining the integrity and safety of the pharmaceutical supply chain.
3Quality & Labeling
Pharmaceutical products imported into Brazil are subject to batch testing and stability requirements to ensure their safety and efficacy. ANVISA mandates that imported drugs undergo quality control procedures, including testing for purity, potency, and stability, to meet Brazilian standards. (gov.br)
Labeling requirements stipulate that product information be provided in Portuguese, including dosage instructions, indications, contraindications, and storage conditions. Serialization mandates are also in place to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Brazil implemented several regulatory changes affecting pharmaceutical imports. ANVISA introduced stricter guidelines for product registration, emphasizing the need for comprehensive clinical data and GMP compliance. Additionally, new labeling requirements were enforced, mandating the inclusion of QR codes for product verification. These changes aim to enhance product traceability and consumer safety.
Ranbaxy Farmaceutica Ltda : — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Ranbaxy Farmaceutica Ltda.'s focus on advanced diabetes medications, vitamins, and nutritional supplements aligns with the growing demand for chronic disease management and preventive healthcare in Brazil. The prevalence of diabetes and other lifestyle-related conditions has increased, driving the need for effective therapeutic options.
The company's import strategy reflects an understanding of market needs, positioning itself to provide essential medications that address prevalent health issues. By sourcing these products from India, Ranbaxy Farmaceutica Ltda. leverages cost-effective manufacturing while meeting the therapeutic demands of the Brazilian population.
2Sourcing Profile
Ranbaxy Farmaceutica Ltda. employs a sourcing strategy that emphasizes the importation of generic drugs, focusing on finished pharmaceutical formulations. This approach allows the company to offer affordable therapeutic options to the Brazilian market. The preference for formulations that address chronic conditions, such as diabetes, indicates a strategic alignment with prevalent health concerns.
India's established pharmaceutical manufacturing capabilities and adherence to international GMP standards make it a suitable partner for Ranbaxy Farmaceutica Ltda.'s procurement needs. The company's sourcing profile reflects a commitment to quality, affordability, and responsiveness to market demands.
3Market Positioning
Based on its product mix, Ranbaxy Farmaceutica Ltda. serves the Brazilian pharmaceutical market through wholesale distribution channels. By importing and distributing a range of pharmaceutical products, the company plays a pivotal role in ensuring the availability of essential medications across the country.
The focus on chronic disease management and preventive healthcare positions Ranbaxy Farmaceutica Ltda. to cater to both retail pharmacies and hospital suppliers, addressing the needs of a diverse patient population.
Seller's Guide — How to Become a Supplier to Ranbaxy Farmaceutica Ltda :
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Ranbaxy Farmaceutica Ltda. by offering complementary products that align with the company's existing portfolio. Introducing innovative formulations or addressing unmet therapeutic needs could enhance the company's product offerings and market competitiveness.
Identifying gaps in Ranbaxy Farmaceutica Ltda.'s current sourcing, such as the need for additional suppliers to mitigate risks associated with single-source dependency, presents an opportunity for Indian exporters to establish partnerships and diversify the company's
Frequently Asked Questions — Ranbaxy Farmaceutica Ltda :
What products does Ranbaxy Farmaceutica Ltda : import from India?
Ranbaxy Farmaceutica Ltda : imports 3 pharmaceutical products across 3 categories. Top imports: Gliclazide ($2.3M), Magnesium ($1.9M), Sodium ($1.6M).
Who supplies pharmaceuticals to Ranbaxy Farmaceutica Ltda : from India?
Ranbaxy Farmaceutica Ltda : sources from 1 verified Indian suppliers. The primary supplier is Sun Pharmaceutical Industries Limited (100.0% of imports, $42.3M).
What is Ranbaxy Farmaceutica Ltda :'s total pharmaceutical import value?
Ranbaxy Farmaceutica Ltda :'s total pharmaceutical import value from India is $5.8M, based on 146 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Ranbaxy Farmaceutica Ltda : focus on?
Ranbaxy Farmaceutica Ltda : imports across 3 categories. The largest: Advanced Diabetes Medications (39.3%), Vitamins & Supplements (32.1%), Nutritional Supplements (28.6%).
Get Full Ranbaxy Farmaceutica Ltda : Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Ranbaxy Farmaceutica Ltda : identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Ranbaxy Farmaceutica Ltda :'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 146 individual customs records matching Ranbaxy Farmaceutica Ltda :.
- 5.Supplier Verification: Ranbaxy Farmaceutica Ltda : sources from 1 verified Indian suppliers across 787 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.