Prince Farma SA .
Pharmaceutical Importer · Angola · Brand Names & OTC Products Focus · $120.5K Total Trade · DGFT Verified
Prince Farma SA . is a pharmaceutical importer based in Angola with a total trade value of $120.5K across 2 products in 2 therapeutic categories. Based on 4 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Prince Farma SA . sources from 1 verified Indian supplier, with Lykis Limited accounting for 100.0% of imports.
Prince Farma SA . — Import Portfolio & Supplier Network
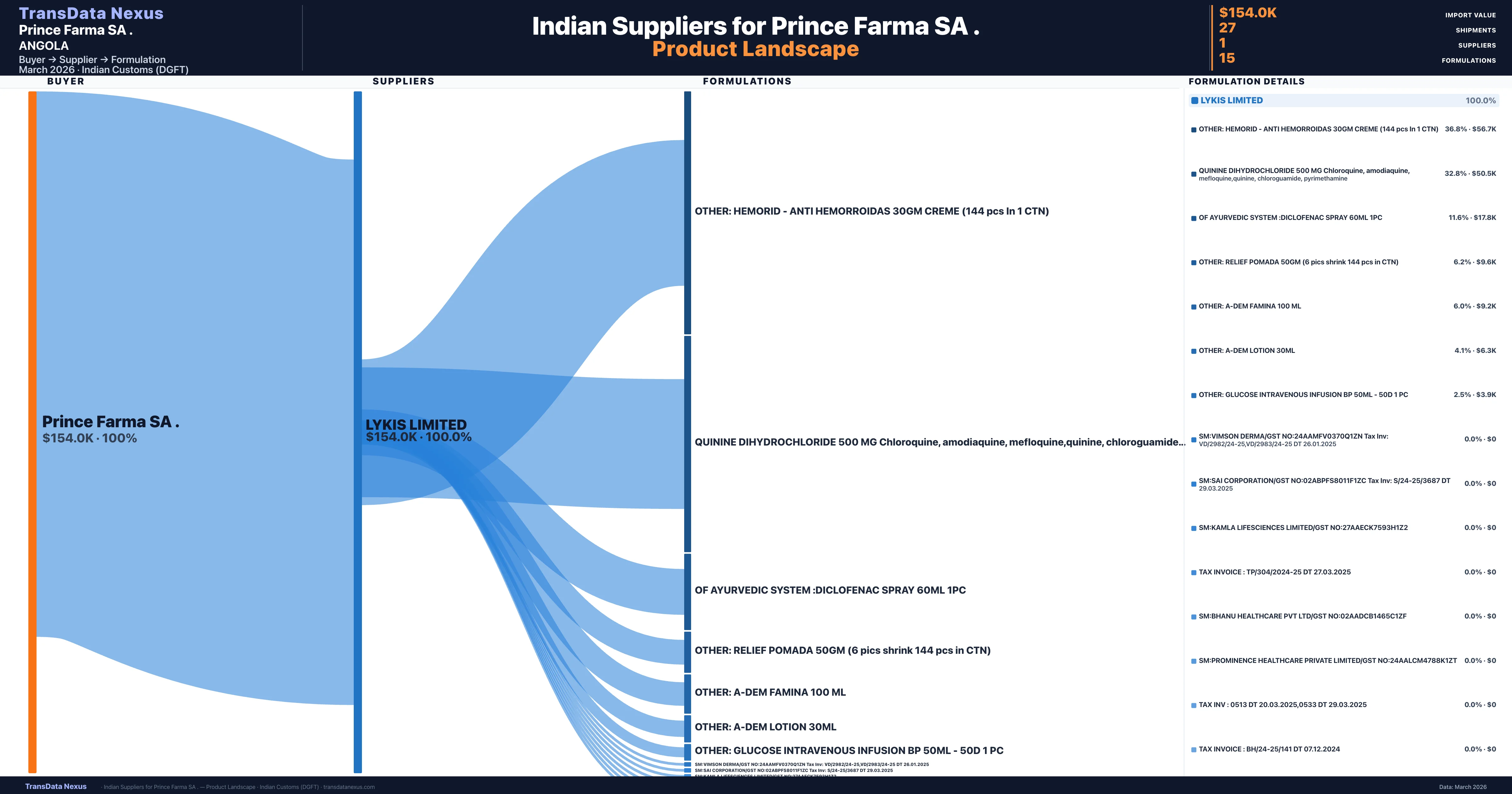
Who Are the Verified Indian Suppliers to Prince Farma SA .?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Lykis Limited | $154.0K | 27 | 100.0% |
Prince Farma SA . sources from 1 verified Indian supplier across 15 distinct formulations. The sourcing is highly concentrated — Lykis Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Prince Farma SA . Import?
| Formulation | Value | Ships |
|---|---|---|
| Other: hemorid - anti hemorroidas 30GM creme (144 pcs in 1 ctn) | $56.7K | 2 |
| Quinine dihydrochloride 500 MG chloroquine, amodiaquine, mefloquine,quinine, chloroguamide | $50.5K | 2 |
| Of ayurvedic system :diclofenac spray 60ML | $17.8K | 2 |
| Other: relief pomada 50GM (6 pics shrink 144 pcs in ctn) | $9.6K | 2 |
| Other: a-dem famina 100 ML | $9.2K | 1 |
| Other: a-dem lotion 30ML | $6.3K | 1 |
| Other: glucose intravenous infusion BP 50ML - 50d 1 | $3.9K | 2 |
| Sm:vimson derma/gst no:24aamfv0370q1zn tax inv: vd/2982/24-25,vd/2983/24-25 dt | $0 | 1 |
| Sm:sai corporation/gst no:02abpfs8011f1zc tax inv: s/24-25/3687 dt | $0 | 2 |
| Sm:kamla lifesciences limited/gst | $0 | 2 |
| Tax invoice : tp/304/2024-25 dt | $0 | 2 |
| Sm:bhanu healthcare pvt ltd/gst | $0 | 2 |
| Sm:prominence healthcare private limited/gst | $0 | 2 |
| Tax inv : 0513 dt 20.03.2025,0533 dt | $0 | 2 |
| Tax invoice : bh/24-25/141 dt | $0 | 2 |
Prince Farma SA . imports 15 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Prince Farma SA . Import?
Top Products by Import Value
Prince Farma SA . Therapeutic Categories — 2 Specializations
Prince Farma SA . imports across 2 therapeutic categories, with Brand Names & OTC Products (58.1%), Antimalarial & Antiparasitic (41.9%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Brand Names & OTC Products
1 products · 58.1% · $70.0K
Antimalarial & Antiparasitic
1 products · 41.9% · $50.5K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Soframycin | Brand Names & OTC Products | $70.0K | 2 | 11.7% | 4 |
| 2 | Mefloquine | Antimalarial & Antiparasitic | $50.5K | 2 | 2.1% | 6 |
Prince Farma SA . imports 2 pharmaceutical products across 2 categories into Angola totaling $120.5K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Prince Farma SA ..
Request DemoPrince Farma SA . — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Prince Farma SA is a leading pharmaceutical company based in Luanda, Angola, specializing in the distribution, sales, and marketing of pharmaceutical products. Established in 2005, the company has built a strong reputation for providing high-quality and affordable healthcare solutions across Angola. Prince Farma SA operates as a wholesaler and distributor, collaborating with multinational companies to supply a diverse range of medicines and healthcare products. The company is ISO 9001:2015 certified, reflecting its commitment to quality management systems.
The company's mission is to make lives healthier by ensuring the availability of quality and affordable medicines for all. Prince Farma SA offers a broad spectrum of products, including generic and branded medicines, surgical items, and hospital equipment. Its portfolio spans various therapeutic areas, such as analgesics, antipyretics, anti-inflammatory agents, antimalarials, and cardiovascular drugs. By collaborating with WHO-GMP certified, EU-GMP certified, and ISO standard companies, Prince Farma SA ensures that its products meet international quality standards.
2Distribution Network
Prince Farma SA has developed an extensive distribution network to ensure the efficient delivery of pharmaceutical products throughout Angola. The company operates 12 distribution outlets strategically located in key provinces, including Cabinda, Uige, Luanda, Malanje, Benguela, Huambo, Huila, and Lunda Sul. This widespread presence enables Prince Farma SA to effectively serve a broad clientele across the country.
In addition to its domestic distribution capabilities, Prince Farma SA collaborates with renowned logistics firms to facilitate timely and reliable deliveries. The company also owns a fleet of controlled-temperature vehicles, ensuring that products requiring specific storage conditions are transported appropriately. This comprehensive logistics infrastructure underscores Prince Farma SA's commitment to maintaining product quality and meeting customer expectations.
3Industry Role
Prince Farma SA plays a pivotal role in Angola's pharmaceutical supply chain as a primary wholesaler and distributor. By sourcing a wide array of pharmaceutical products from international manufacturers, the company ensures the availability of essential medicines and healthcare products within the Angolan market. Its extensive distribution network and partnerships with multinational companies enable Prince Farma SA to meet the diverse healthcare needs of the population.
The company's strategic focus on quality and affordability has positioned it as a trusted partner for healthcare providers and patients alike. Through its commitment to excellence and customer satisfaction, Prince Farma SA continues to strengthen its position in the pharmaceutical industry, contributing significantly to the health and well-being of the Angolan population.
Supplier Relationship Intelligence — Prince Farma SA .
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Prince Farma SA's sourcing strategy exhibits a high degree of concentration, with a total import value of $121,000 USD from India across four shipments. This concentration is further emphasized by the fact that the top two imported products—Soframycin and Mefloquine—account for 11.7% and 2.1% of the total import value, respectively. Notably, LYKIS LIMITED is the sole verified Indian supplier, accounting for 100% of the shipments.
This single-source dependency may present risks, such as supply chain disruptions due to geopolitical issues, regulatory changes, or operational challenges faced by the supplier. However, it also indicates a strategic choice by Prince Farma SA to establish a focused and potentially more efficient procurement process with a trusted supplier. The stability of this relationship is crucial for maintaining a consistent supply of the imported products.
The shipment data reveals a consistent pattern of imports from LYKIS LIMITED, suggesting a stable and ongoing partnership. This stability is essential for ensuring the uninterrupted availability of key pharmaceutical products in the Angolan market. Nonetheless, diversifying the supplier base could mitigate potential risks associated with over-reliance on a single source.
2Supply Chain Resilience
Prince Farma SA's supply chain resilience is closely tied to its exclusive partnership with LYKIS LIMITED, the sole verified Indian supplier. This singular sourcing strategy may expose the company to risks such as supply disruptions, price fluctuations, and potential challenges in meeting regulatory requirements. The absence of backup suppliers for the imported formulations further underscores this vulnerability.
The limited diversity in imported formulations, with only 15 unique products, suggests a narrow product range that may not fully address the diverse healthcare needs of the Angolan population. Additionally, the reliance on a single shipping route and the lack of alternative suppliers could impact the company's ability to adapt to changes in the global pharmaceutical supply chain.
To enhance supply chain resilience, Prince Farma SA could consider diversifying its supplier base and expanding the range of imported formulations. Engaging with multiple suppliers and incorporating a broader spectrum of products would provide greater flexibility and security, ensuring a more robust and responsive supply chain capable of meeting the evolving healthcare demands of Angola.
3Strategic Implications
Prince Farma SA's concentrated sourcing pattern, characterized by a sole supplier and a limited product range, has significant strategic implications. While this approach may streamline procurement processes and foster strong supplier relationships, it also exposes the company to potential risks associated with supply chain disruptions and market fluctuations.
For Indian exporters, the existing sourcing pattern presents both challenges and opportunities. The exclusive relationship between Prince Farma SA and LYKIS LIMITED may limit entry points for new suppliers. However, the company's openness to high-quality, affordable pharmaceutical products suggests that there may be opportunities to introduce complementary products or alternative formulations that align with Prince Farma SA's mission and market needs.
To capitalize on these opportunities, Indian exporters should focus on demonstrating product quality, regulatory compliance, and the ability to meet the specific requirements of the Angolan market. Building relationships with Prince Farma SA and understanding their strategic objectives will be crucial steps in establishing a successful partnership.
Importing Pharmaceuticals into Angola — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Angola
1Regulatory Authority & Framework
In Angola, the pharmaceutical sector is regulated by the Regulatory Agency for Medicines and Healthcare Technologies (ARMED), operating under the Ministry of Health. ARMED is responsible for regulating and supervising the sector, licensing pharmaceuticals and health technologies for human use, ensuring the quality of healthcare products and services entering Angola, and safeguarding public health.
Key legislation governing pharmaceutical imports includes the General Law of Medicines and Medical Devices (2019), which establishes guidelines for the regulation of medicines and medical devices in Angola. This law outlines the requirements for importing medicines, including the need for special authorization from the Ministry of Health for products not approved or unavailable in the country but essential for saving lives.
The marketing authorization pathway for Indian generics involves obtaining approval from ARMED, which includes submitting comprehensive documentation such as certificates of analysis, certificates of free sale, and compliance with international quality standards. This process ensures that imported generics meet the safety, efficacy, and quality standards required for the Angolan market.
2Import Licensing & GMP
Import licensing requirements in Angola are governed by Presidential Decree No. 202/21 of 26 August 2021, which aims to streamline the licensing process for distribution points. This decree facilitates the entry of new players into the market by reducing bureaucratic hurdles, thereby promoting competition and improving the availability of pharmaceutical products.
Angola recognizes Good Manufacturing Practice (GMP) certificates from reputable international bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their products are manufactured in facilities holding these certifications to comply with Angolan regulations.
Wholesale distribution authorization is required for companies like Prince Farma SA to operate legally within Angola. This authorization involves demonstrating compliance with regulatory standards, including product quality, storage conditions, and distribution practices, to ensure the safety and efficacy of pharmaceutical products in the market.
3Quality & Labeling
Imported pharmaceutical products are subject to batch testing and stability requirements to ensure their safety and efficacy. Angolan authorities may conduct inspections and testing to verify compliance with established standards. Manufacturers and importers must provide necessary documentation and samples as part of this process.
Labeling requirements include providing information in Portuguese, the official language of Angola. Labels must include details such as product name, active ingredients, dosage form, strength, manufacturer information, batch number, expiration date, and storage conditions. Serialization mandates may also be in place to enhance traceability and prevent counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, Angola has implemented policy changes affecting pharmaceutical imports, including the establishment of ARMED and the enactment of Presidential Decree No. 202/21. These changes aim to improve regulatory oversight, streamline licensing processes, and enhance the quality and safety of imported pharmaceutical products.
Frequently Asked Questions — Prince Farma SA .
What products does Prince Farma SA . import from India?
Prince Farma SA . imports 2 pharmaceutical products across 2 categories. Top imports: Soframycin ($70.0K), Mefloquine ($50.5K).
Who supplies pharmaceuticals to Prince Farma SA . from India?
Prince Farma SA . sources from 1 verified Indian suppliers. The primary supplier is Lykis Limited (100.0% of imports, $154.0K).
What is Prince Farma SA .'s total pharmaceutical import value?
Prince Farma SA .'s total pharmaceutical import value from India is $120.5K, based on 4 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Prince Farma SA . focus on?
Prince Farma SA . imports across 2 categories. The largest: Brand Names & OTC Products (58.1%), Antimalarial & Antiparasitic (41.9%).
Get Full Prince Farma SA . Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Prince Farma SA . identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Prince Farma SA .'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 4 individual customs records matching Prince Farma SA ..
- 5.Supplier Verification: Prince Farma SA . sources from 1 verified Indian suppliers across 15 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.