Piex Societe Pharmaceutique
Pharmaceutical Importer · France · Advanced Antibiotics Focus · $1.1M Total Trade · DGFT Verified
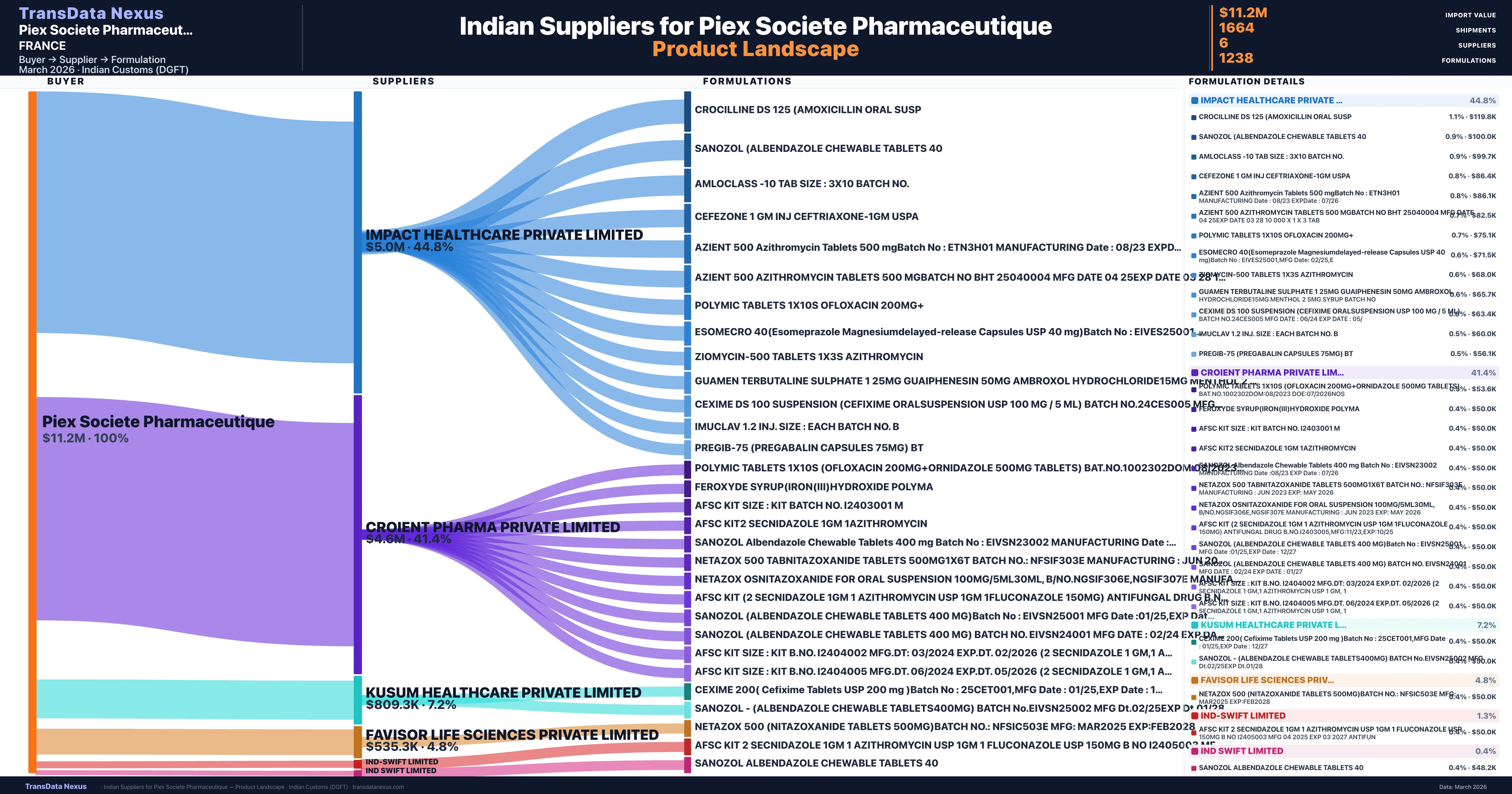
Piex Societe Pharmaceutique is a pharmaceutical importer based in France with a total trade value of $1.1M across 3 products in 3 therapeutic categories. Based on 32 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Piex Societe Pharmaceutique sources from 6 verified Indian suppliers, with Impact Healthcare Private Limited accounting for 44.8% of imports.
Piex Societe Pharmaceutique — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Piex Societe Pharmaceutique?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Impact Healthcare Private Limited | $5.0M | 802 | 44.8% |
| Croient Pharma Private Limited | $4.6M | 678 | 41.4% |
| Kusum Healthcare Private Limited | $809.3K | 121 | 7.2% |
| Favisor Life Sciences Private Limited | $535.3K | 57 | 4.8% |
| Ind-swift Limited | $145.0K | 4 | 1.3% |
| Ind Swift Limited | $50.0K | 2 | 0.4% |
Piex Societe Pharmaceutique sources from 6 verified Indian suppliers across 1,238 distinct formulations. The supply base is diversified across 6 suppliers, reducing single-source dependency risk.
What Formulations Does Piex Societe Pharmaceutique Import?
| Formulation | Value | Ships |
|---|---|---|
| Crocilline ds 125 (amoxicillin oral | $119.8K | 9 |
| Sanozol (albendazole chewable tablets | $100.0K | 2 |
| Amloclass -10 TAB size : 3x10 batch no. | $99.7K | 2 |
| Cefezone 1 GM INJ ceftriaxone-1GM | $86.4K | 6 |
| Azient 500 azithromycin tablets 500 mgbatch no : etn3h01 manufacturing date : 08/23 expdate | $86.1K | 2 |
| Azient 500 azithromycin tablets 500 mgbatch no bht 25040004 mfg date 04 25exp date 03 28 10 000 x 1 x 3 TAB | $82.5K | 3 |
| Polymic tablets 1x10s ofloxacin | $75.1K | 6 |
| Esomecro 40(esomeprazole magnesiumdelayed-release capsules USP 40 MG)batch no : eives25001,mfg date: 02/25 | $71.5K | 2 |
| Ziomycin-500 tablets 1x3s | $68.0K | 6 |
| Guamen terbutaline sulphate 1 25MG guaiphenesin 50MG ambroxol hydrochloride15mg menthol 2 5MG syrup batch | $65.7K | 4 |
| Cexime ds 100 suspension (cefixime oralsuspension USP 100 MG / 5 ML) batch no.24ces005 mfg date : 06/24 exp date | $63.4K | 2 |
| Imuclav 1.2 INJ. size : Each batch no. | $60.0K | 4 |
| Pregib-75 (pregabalin capsules 75MG) | $56.1K | 7 |
| Polymic tablets 1x10s (ofloxacin 200MG+ornidazole 500MG tablets) bat.no.1002302dom:08/2023 | $53.6K | 3 |
| Feroxyde syrup(iron(iii)hydroxide | $50.0K | 1 |
Piex Societe Pharmaceutique imports 1,238 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Piex Societe Pharmaceutique Import?
Top Products by Import Value
Piex Societe Pharmaceutique Therapeutic Categories — 3 Specializations
Piex Societe Pharmaceutique imports across 3 therapeutic categories, with Advanced Antibiotics (58.5%), Antimalarial & Antiparasitic (39.2%), Respiratory & OTC (2.3%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
1 products · 58.5% · $670.9K
Antimalarial & Antiparasitic
1 products · 39.2% · $450.0K
Respiratory & OTC
1 products · 2.3% · $25.9K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Secnidazole | Advanced Antibiotics | $670.9K | 19 | 1.4% | 5 |
| 2 | Albendazole | Antimalarial & Antiparasitic | $450.0K | 9 | 0.6% | 14 |
| 3 | Terbutaline | Respiratory & OTC | $25.9K | 4 | 0.7% | 13 |
Piex Societe Pharmaceutique imports 3 pharmaceutical products across 3 categories into France totaling $1.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Piex Societe Pharmaceutique.
Request DemoPiex Societe Pharmaceutique — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Piex Societe Pharmaceutique, established in 1979 by Robert Jamain, is a French company specializing in the import and distribution of pharmaceutical products. Headquartered in Les Ulis, France, the company has developed a significant presence in the pharmaceutical sector, particularly focusing on the African market. In July 2017, Piex was acquired by a group of investors led by LBO France, which included AfricInvest, BNP Paribas Développement, Bpifrance, and Trocadéro Capital Partners. This acquisition aimed to enhance Piex's capabilities in distributing health and wellness products across Africa, the Middle East, and Asia.
Piex's product portfolio encompasses over 2,000 items, including pharmaceuticals, personal hygiene products, formula milk, and medical devices. The company serves a diverse clientele, ranging from wholesalers and pharmacies to hospitals, ensuring a comprehensive distribution network. In 2023, Piex expanded its operations by acquiring Apexfarma and its subsidiary Cross Pharm, further strengthening its value chain and reinforcing its position in the African market.
2Distribution Network
Piex Societe Pharmaceutique operates a robust distribution network with a significant presence in Africa, Asia, and the Middle East. While specific warehouse locations within France are not publicly disclosed, the company's strategic acquisitions, such as Apexfarma and Cross Pharm, have bolstered its logistics capabilities. These expansions have enhanced Piex's ability to manage and distribute a wide range of health and wellness products efficiently across various regions.
3Industry Role
In France's pharmaceutical supply chain, Piex Societe Pharmaceutique functions primarily as a pharmaceutical importer and distributor. The company imports finished pharmaceutical formulations from countries like India and distributes them to various stakeholders, including wholesalers, pharmacies, and hospitals. Piex's strategic focus on the African market underscores its role in bridging the gap between pharmaceutical manufacturers and healthcare providers in regions with significant demand for quality health products.
Supplier Relationship Intelligence — Piex Societe Pharmaceutique
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Piex Societe Pharmaceutique's sourcing strategy reveals a high concentration on a limited number of suppliers. The company has imported a total of 1,528 shipments from India, with the majority of these shipments originating from six key suppliers. This sourcing pattern indicates a strategic choice to establish strong, long-term relationships with select suppliers, ensuring consistent product quality and supply reliability. However, such concentration also presents potential risks, including dependency on a few suppliers and exposure to supply chain disruptions. The shipment data suggests a stable relationship with these suppliers, as evidenced by the consistent volume of imports over the years.
2Supply Chain Resilience
Piex Societe Pharmaceutique's supply chain resilience is bolstered by its partnerships with multiple Indian suppliers, providing a degree of diversification. The company's import data indicates a diverse range of formulations, with 1,238 unique formulations imported. This diversity suggests that Piex is not overly reliant on a single product line, enhancing its ability to adapt to market changes and demand fluctuations. While the company has established relationships with key suppliers, the absence of publicly available information on backup suppliers and specific shipping routes limits a comprehensive assessment of its supply chain resilience. Additionally, the regulatory compliance of key suppliers is not detailed in the available data, which is crucial for ensuring consistent product quality and adherence to international standards.
3Strategic Implications
Piex Societe Pharmaceutique's sourcing pattern, characterized by a concentrated supplier base and a diverse product portfolio, positions the company to leverage strong supplier relationships for consistent product quality and supply reliability. For Indian exporters, this presents an opportunity to become alternative suppliers by meeting Piex's quality standards and demonstrating the ability to fulfill large-scale orders. However, potential suppliers must be prepared to navigate the complexities of the French and African pharmaceutical markets, including regulatory requirements and distribution logistics. Establishing a partnership with Piex could lead to access to a broad distribution network and a foothold in emerging markets.
Importing Pharmaceuticals into France — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for France
1Regulatory Authority & Framework
In France, the primary regulatory authority overseeing pharmaceutical imports is the French National Agency for Medicines and Health Products Safety (ANSM). The ANSM is responsible for ensuring the safety, efficacy, and quality of health products available in the French market. Key legislation governing pharmaceutical imports includes the Public Health Code, which outlines the requirements for marketing authorization, importation, and distribution of medicinal products. For Indian generics to be marketed in France, they must undergo a centralized marketing authorization process through the European Medicines Agency (EMA), which evaluates the quality, safety, and efficacy of medicinal products. Once authorized by the EMA, the product can be marketed across all EU member states, including France.
2Import Licensing & GMP
Import licensing requirements in France stipulate that pharmaceutical products must be authorized for sale by the ANSM. This involves obtaining a marketing authorization, which is granted based on the evaluation of the product's quality, safety, and efficacy. Good Manufacturing Practice (GMP) certification is a critical component of this process. The EMA recognizes GMP certifications from various regulatory bodies, including the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian manufacturers seeking to export to France must ensure that their facilities comply with these recognized GMP standards. Additionally, wholesale distribution authorization is required for entities involved in the distribution of pharmaceutical products within France.
3Quality & Labeling
Pharmaceutical products imported into France must undergo batch testing to verify their quality and compliance with regulatory standards. Stability requirements are also enforced to ensure that products maintain their efficacy and safety throughout their shelf life. Labeling must be in French and include essential information such as the product's name, active ingredients, dosage form, and instructions for use. Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the market. These requirements are part of the EU Falsified Medicines Directive, which aims to protect public health by ensuring the integrity of the pharmaceutical supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, several policy changes have impacted the importation of Indian pharmaceuticals into France. The ANSM has implemented stricter guidelines for the evaluation of marketing authorization applications, emphasizing the need for comprehensive clinical data and robust quality control measures. Additionally, the EMA has updated its GMP guidelines to align with international standards, requiring Indian manufacturers to demonstrate compliance with these enhanced criteria. These changes aim to ensure that imported generics meet the highest standards of quality, safety, and efficacy, thereby protecting public health and maintaining the integrity of the French pharmaceutical market.
Piex Societe Pharmaceutique — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Piex Societe Pharmaceutique's focus on importing advanced antibiotics, antimalarial and antiparasitic agents, and respiratory and over-the-counter (OTC) products aligns with the significant health challenges faced in the African market. Advanced antibiotics are crucial in combating bacterial infections, while antimalarial and antiparasitic agents are essential in regions where these diseases are endemic. Respiratory and OTC products address common health issues prevalent in the target markets. The market demand for these therapeutic areas is driven by the high incidence of infectious diseases and the need for accessible and effective treatments. By focusing on these categories, Piex aims to meet critical health needs and establish a strong presence in the pharmaceutical distribution sector.
2Sourcing Profile
Piex Societe Pharmaceutique's sourcing strategy emphasizes the importation of generic drugs, particularly from India, which is known for its robust pharmaceutical manufacturing capabilities. The company's preference for finished pharmaceutical formulations, such as tablets, capsules, syrups, and injections, indicates a focus on ready-to-market products that can be efficiently distributed. India's compliance with international GMP standards and its competitive pricing make it an attractive sourcing destination for Piex. This strategy allows Piex to offer a diverse range of quality products to meet the health needs of its target markets.
3Market Positioning
Based on its product mix, Piex Societe Pharmaceutique primarily serves the wholesale distribution segment of the French market. The company's focus on importing and distributing pharmaceutical products positions it as a key player in supplying wholesalers, pharmacies, and hospitals with essential medications and health products. Piex's strategic emphasis on the African market further indicates its role in addressing the health needs of regions with significant demand for quality pharmaceutical products.
Seller's Guide — How to Become a Supplier to Piex Societe Pharmaceutique
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with Piex Societe Pharmaceutique. The company's established relationships with key Indian suppliers and its focus on importing finished pharmaceutical formulations suggest a preference for suppliers who can meet stringent quality standards and regulatory requirements. Potential suppliers should be prepared to demonstrate compliance with international GMP standards, provide comprehensive product documentation, and ensure reliable supply capabilities. By meeting these criteria, new Indian suppliers can position themselves as viable partners for Piex, potentially gaining access to a broad distribution network and a foothold in emerging markets.
Frequently Asked Questions — Piex Societe Pharmaceutique
What products does Piex Societe Pharmaceutique import from India?
Piex Societe Pharmaceutique imports 3 pharmaceutical products across 3 categories. Top imports: Secnidazole ($670.9K), Albendazole ($450.0K), Terbutaline ($25.9K).
Who supplies pharmaceuticals to Piex Societe Pharmaceutique from India?
Piex Societe Pharmaceutique sources from 6 verified Indian suppliers. The primary supplier is Impact Healthcare Private Limited (44.8% of imports, $5.0M).
What is Piex Societe Pharmaceutique's total pharmaceutical import value?
Piex Societe Pharmaceutique's total pharmaceutical import value from India is $1.1M, based on 32 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Piex Societe Pharmaceutique focus on?
Piex Societe Pharmaceutique imports across 3 categories. The largest: Advanced Antibiotics (58.5%), Antimalarial & Antiparasitic (39.2%), Respiratory & OTC (2.3%).
Get Full Piex Societe Pharmaceutique Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Piex Societe Pharmaceutique identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Piex Societe Pharmaceutique's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 32 individual customs records matching Piex Societe Pharmaceutique.
- 5.Supplier Verification: Piex Societe Pharmaceutique sources from 6 verified Indian suppliers across 1,238 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.