Piex
Pharmaceutical Importer · France · Vitamins & Supplements Focus · $1.4M Total Trade · DGFT Verified
Piex is a pharmaceutical importer based in France with a total trade value of $1.4M across 2 products in 2 therapeutic categories. Based on 33 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Piex sources from 24 verified Indian suppliers, with Impact Healthcare Private Limited accounting for 22.8% of imports.
Piex — Import Portfolio & Supplier Network
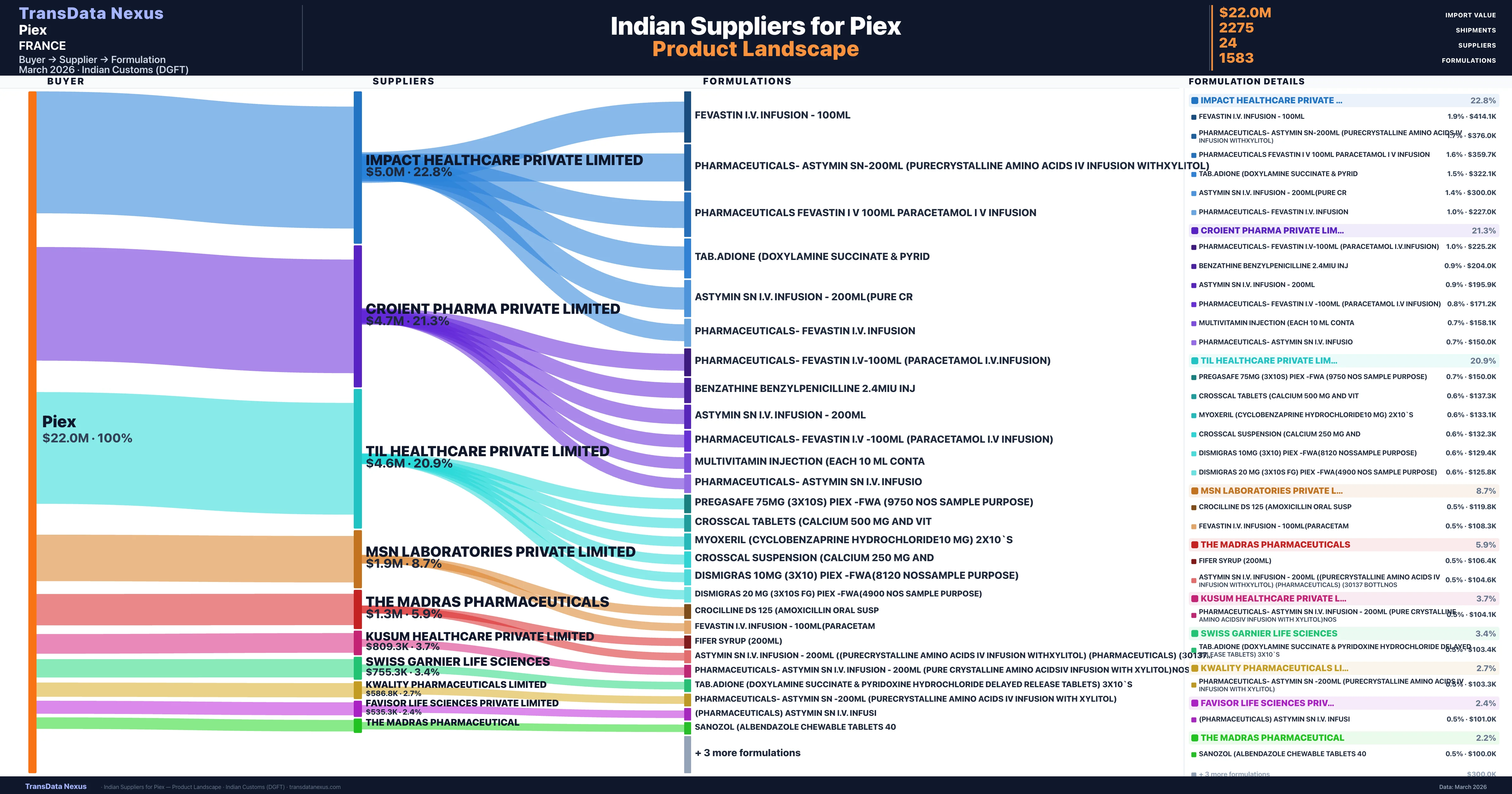
Who Are the Verified Indian Suppliers to Piex?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Impact Healthcare Private Limited | $5.0M | 802 | 22.8% |
| Croient Pharma Private Limited | $4.7M | 691 | 21.3% |
| Til Healthcare Private Limited | $4.6M | 156 | 20.9% |
| Msn Laboratories Private Limited | $1.9M | 78 | 8.7% |
| The Madras Pharmaceuticals | $1.3M | 56 | 5.9% |
| Kusum Healthcare Private Limited | $809.3K | 121 | 3.7% |
| Swiss Garnier Life Sciences | $755.3K | 117 | 3.4% |
| Kwality Pharmaceuticals Limited | $586.8K | 27 | 2.7% |
| Favisor Life Sciences Private Limited | $535.3K | 57 | 2.4% |
| The Madras Pharmaceutical | $474.4K | 12 | 2.2% |
| Medreich Limited | $251.1K | 12 | 1.1% |
| Brooks Laboratories Limited | $234.9K | 30 | 1.1% |
| Aishwarya Health Care | $214.5K | 15 | 1.0% |
| Indus Life Sciences Private Limited | $197.0K | 23 | 0.9% |
| Ind-swift Limited | $145.0K | 4 | 0.7% |
| Cyano Pharma Private Limited | $87.4K | 21 | 0.4% |
| Fiza Healthcare Private Limited | $70.8K | 32 | 0.3% |
| Ind Swift Limited | $50.0K | 2 | 0.2% |
| Trugen Pharmaceuticals Private Limited | $27.3K | 7 | 0.1% |
| Unijules Life Sciences Limited | $14.7K | 3 | 0.1% |
| Brussels Laboratories Private Limited | $13.8K | 5 | 0.1% |
| Concept Pharmaceuticals Limited | $13.6K | 2 | 0.1% |
| Strava Healthcare Private Limited | $4.9K | 1 | 0.0% |
| Par Laboratories | $1.5K | 1 | 0.0% |
Piex sources from 24 verified Indian suppliers across 1,583 distinct formulations. The supply base is diversified across 24 suppliers, reducing single-source dependency risk.
What Formulations Does Piex Import?
| Formulation | Value | Ships |
|---|---|---|
| Fevastin i.v. infusion - 100ML | $414.1K | 12 |
| Pharmaceuticals- astymin sn-200ML (purecrystalline amino acids iv infusion withxylitol) | $376.0K | 8 |
| Pharmaceuticals fevastin i v 100ML paracetamol i v | $359.7K | 12 |
| Tab.adione (doxylamine succinate & | $322.1K | 8 |
| Astymin sn i.v. infusion - 200ML(pure | $300.0K | 6 |
| Pharmaceuticals- fevastin i.v. | $227.0K | 6 |
| Pharmaceuticals- fevastin i.v-100ML (paracetamol i.v.infusion) | $225.2K | 6 |
| Benzathine benzylpenicilline 2.4miu INJ | $204.0K | 6 |
| Astymin sn i.v. infusion - 200ML | $195.9K | 4 |
| Pharmaceuticals- fevastin i.v -100ML (paracetamol i.v infusion) | $171.2K | 9 |
| Multivitamin injection (Each 10 ML | $158.1K | 5 |
| Pharmaceuticals- astymin sn i.v. | $150.0K | 3 |
| Pregasafe 75MG (3x10s) piex -fwa (9750 nos sample purpose) | $150.0K | 3 |
| Crosscal tablets (calcium 500 MG and | $137.3K | 4 |
| Myoxeril (cyclobenzaprine hydrochloride10 MG) | $133.1K | 3 |
Piex imports 1,583 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Piex Import?
Top Products by Import Value
Piex Therapeutic Categories — 2 Specializations
Piex imports across 2 therapeutic categories, with Vitamins & Supplements (74.1%), Advanced Antibiotics (25.9%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Vitamins & Supplements
1 products · 74.1% · $1.1M
Advanced Antibiotics
1 products · 25.9% · $366.8K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Calcium | Vitamins & Supplements | $1.1M | 21 | 0.1% | 17 |
| 2 | Benzylpenicillin | Advanced Antibiotics | $366.8K | 12 | 9.5% | 3 |
Piex imports 2 pharmaceutical products across 2 categories into France totaling $1.4M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Piex — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Piex, officially known as Société Pharmaceutique d’Import-Export, is a French company specializing in the distribution of pharmaceutical and healthcare products. Established in 1979 by Robert Jamain, Piex has evolved into a significant player in the pharmaceutical sector, focusing primarily on the African market. The company's headquarters are located at 3-7 Avenue du Cap Horn, Les Ulis, France.
In July 2017, Piex underwent a strategic acquisition led by LBO France, in partnership with AfricInvest Europe, BNP Paribas Développement, BPI, and Trocadéro Capital Partners. This acquisition aimed to enhance Piex's capabilities in distributing genuine medicines and parapharmaceuticals across Africa, the Middle East, and Asia.
Piex operates as an independent distributor, serving as a crucial link between pharmaceutical manufacturers and healthcare providers, particularly in Sub-Saharan Africa. The company offers a comprehensive range of services, including marketing, regulatory affairs, and logistics, ensuring efficient access to quality health and wellness products.
2Distribution Network
Piex's distribution network is strategically designed to serve a wide array of markets, with a primary focus on Sub-Saharan Africa. The company manages a substantial inventory, offering over 1,500 product references tailored to meet the diverse needs of the region.
Logistically, Piex operates a network of warehouses equipped to handle a variety of products, including medicines, food supplements, and hygiene products. The company emphasizes compliance with Good Distribution Practices (GDP) and is recognized as an Authorized Economic Operator by French Customs, ensuring secure and efficient logistics operations.
3Industry Role
Within France's pharmaceutical supply chain, Piex functions as a specialized distributor, focusing on the export of pharmaceutical and healthcare products to various international markets, particularly in Africa and Asia. The company's role is pivotal in bridging the gap between manufacturers and end-users, ensuring the availability of quality health products in regions with limited access.
Supplier Relationship Intelligence — Piex
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Piex's sourcing strategy reveals a high concentration of imports from India, with a total import value of $1.4 million USD across 33 shipments. The top two imported products are Calcium ($1.1 million, 0.1% share) and Benzylpenicillin ($367,000, 9.5% share). This concentration indicates a strategic choice to source specific formulations from India, likely due to cost-effectiveness and product availability.
The shipment data, with 33 entries, suggests a stable and ongoing relationship with Indian suppliers. However, the lack of diversification in sourcing could pose risks related to supply chain disruptions or regulatory changes in India. Diversifying suppliers and products may enhance resilience and mitigate potential risks.
2Supply Chain Resilience
Piex's supply chain resilience is closely tied to its sourcing strategy from India. The company's reliance on a limited number of suppliers for specific formulations may expose it to risks such as supply disruptions or regulatory changes in India. To enhance resilience, Piex could consider diversifying its supplier base and product range, thereby reducing dependency on a single source.
3Strategic Implications
Piex's concentrated sourcing pattern positions it as a key player in distributing specific pharmaceutical products from India to African markets. For Indian exporters, this presents an opportunity to strengthen partnerships with Piex by offering a broader range of products and ensuring consistent supply to meet the growing demand in Africa.
Importing Pharmaceuticals into France — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for France
1Regulatory Authority & Framework
In France, the primary regulatory authority overseeing pharmaceutical imports is the Agence Nationale de la Sécurité du Médicament et des Produits de Santé (ANSM). The ANSM is responsible for ensuring the safety, efficacy, and quality of health products available in the French market.
Key legislation governing pharmaceutical imports includes the Public Health Code, which outlines the requirements for marketing authorization, importation, and distribution of medicinal products. The marketing authorization pathway for Indian generics involves obtaining a Marketing Authorization (MA) from the ANSM, ensuring compliance with French and European Union standards.
2Import Licensing & GMP
Import licensing requirements in France stipulate that all pharmaceutical products must have a valid Marketing Authorization (MA) issued by the ANSM. Good Manufacturing Practice (GMP) certificates recognized by the European Union, such as EU GMP, WHO GMP, or PIC/S, are mandatory for suppliers. Additionally, wholesale distribution authorization is required for entities involved in the distribution of medicinal products within France.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure compliance with French and European quality standards. Stability requirements are enforced to guarantee product efficacy and safety throughout its shelf life. Labeling must be in French, providing clear information on usage, dosage, and storage. Serialization mandates are implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, France has implemented several regulatory changes affecting pharmaceutical imports. These include stricter compliance with EU regulations on Good Distribution Practices (GDP), enhanced requirements for serialization and traceability, and updated standards for marketing authorization procedures. These changes aim to strengthen the safety and integrity of the pharmaceutical supply chain.
Piex — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Piex's focus on importing Calcium and Benzylpenicillin aligns with the therapeutic needs of the African market, where there is a significant demand for essential medicines. The company's strategy to import these specific products suggests a targeted approach to address prevalent health issues in the regions it serves.
2Sourcing Profile
Piex's sourcing strategy emphasizes importing generic drugs from India, particularly in the categories of Vitamins & Supplements and Advanced Antibiotics. This approach leverages India's manufacturing capabilities and cost advantages, ensuring a steady supply of essential medicines to meet the needs of the African market.
3Market Positioning
Based on its product mix, Piex serves the wholesale distribution segment of the French market, focusing on exporting pharmaceutical products to African countries. The company's role is crucial in ensuring the availability of essential medicines and health products in regions with limited access to such resources.
Seller's Guide — How to Become a Supplier to Piex
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Piex, especially those offering products that complement the existing portfolio. Identifying gaps in Piex's current sourcing, such as additional therapeutic categories or formulations, could lead to mutually beneficial partnerships.
2Requirements & Qualifications
Indian exporters aiming to supply Piex and the French market must ensure their products have valid Marketing Authorization from the ANSM. Compliance with recognized GMP standards, such as EU GMP, WHO GMP, or PIC/S, is essential. Additionally, adherence to French labeling requirements, including French language labeling and serialization mandates, is mandatory.
3How to Approach
Indian exporters should initiate contact with Piex by providing detailed product information, including compliance with French regulatory standards. Participating in relevant tenders and aligning with Piex's strategic goals can enhance the partnership potential. Understanding the regulatory filing process and expected timelines for marketing authorization in France is crucial for a successful collaboration.
Frequently Asked Questions — Piex
What products does Piex import from India?
Piex imports 2 pharmaceutical products across 2 categories. Top imports: Calcium ($1.1M), Benzylpenicillin ($366.8K).
Who supplies pharmaceuticals to Piex from India?
Piex sources from 24 verified Indian suppliers. The primary supplier is Impact Healthcare Private Limited (22.8% of imports, $5.0M).
What is Piex's total pharmaceutical import value?
Piex's total pharmaceutical import value from India is $1.4M, based on 33 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Piex focus on?
Piex imports across 2 categories. The largest: Vitamins & Supplements (74.1%), Advanced Antibiotics (25.9%).
Get Full Piex Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Piex identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Piex's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 33 individual customs records matching Piex.
- 5.Supplier Verification: Piex sources from 24 verified Indian suppliers across 1,583 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.