Pharmachem Private Limited
Pharmaceutical Importer · Nepal · Advanced Oncology Focus · $282.4K Total Trade · DGFT Verified
Pharmachem Private Limited is a pharmaceutical importer based in Nepal with a total trade value of $282.4K across 4 products in 4 therapeutic categories. Based on 46 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Pharmachem Private Limited sources from 14 verified Indian suppliers, with Abbott India Limited accounting for 30.9% of imports.
Pharmachem Private Limited — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Pharmachem Private Limited?
Customs-verified supplier relationships from Indian DGFT records
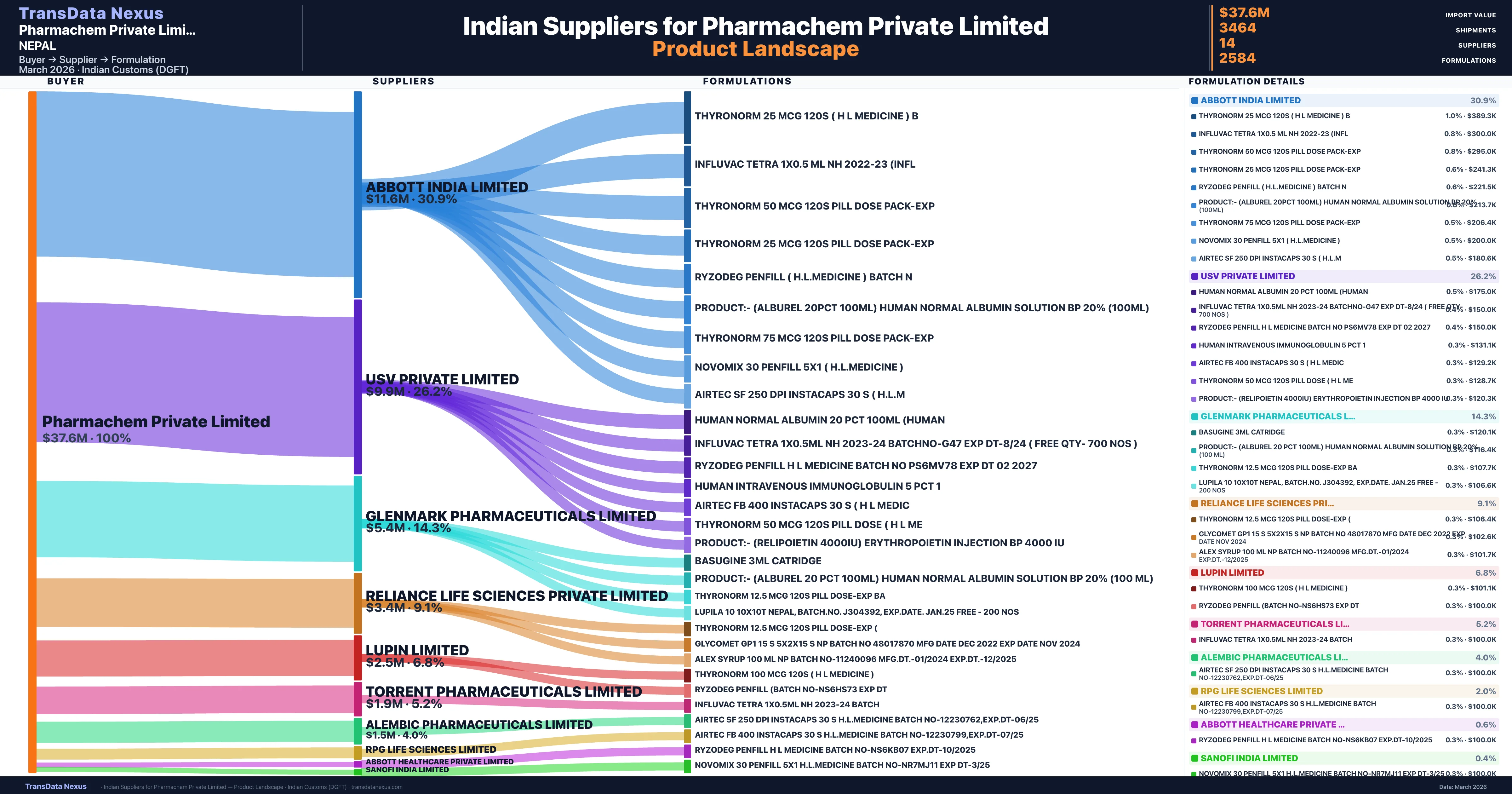
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Abbott India Limited | $11.6M | 1,011 | 30.9% |
| Usv Private Limited | $9.9M | 1,027 | 26.2% |
| Glenmark Pharmaceuticals Limited | $5.4M | 268 | 14.3% |
| Reliance Life Sciences Private Limited | $3.4M | 158 | 9.1% |
| Lupin Limited | $2.5M | 265 | 6.8% |
| Torrent Pharmaceuticals Limited | $1.9M | 289 | 5.2% |
| Alembic Pharmaceuticals Limited | $1.5M | 218 | 4.0% |
| Rpg Life Sciences Limited | $765.3K | 124 | 2.0% |
| Abbott Healthcare Private Limited | $233.6K | 39 | 0.6% |
| Sanofi India Limited | $134.8K | 20 | 0.4% |
| Shreya Life Sciences Private Limited | $133.5K | 26 | 0.4% |
| Abbott Health Care Private Limited | $37.1K | 7 | 0.1% |
| Tablets India Limited | $29.8K | 2 | 0.1% |
| Ban Labs Private Limited | $16.8K | 10 | 0.0% |
Pharmachem Private Limited sources from 14 verified Indian suppliers across 2,584 distinct formulations. The supply base is diversified across 14 suppliers, reducing single-source dependency risk.
What Formulations Does Pharmachem Private Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Thyronorm 25 MCG 120s ( h l medicine ) | $389.3K | 31 |
| Influvac tetra 1x0.5 ML nh 2022-23 | $300.0K | 6 |
| Thyronorm 50 MCG 120s pill dose | $295.0K | 35 |
| Thyronorm 25 MCG 120s pill dose | $241.3K | 24 |
| Ryzodeg penfill ( h.l.medicine ) batch | $221.5K | 5 |
| Product:- (alburel 20pct 100ML) human normal albumin solution BP 20% (100ML) | $213.7K | 6 |
| Thyronorm 75 MCG 120s pill dose | $206.4K | 16 |
| Novomix 30 penfill 5x1 ( h.l.medicine ) | $200.0K | 4 |
| Airtec sf 250 dpi instacaps 30 s | $180.6K | 4 |
| Human normal albumin 20 pct 100ML | $175.0K | 4 |
| Influvac tetra 1x0.5ML nh 2023-24 batchno-g47 exp dt-8/24 ( free qty- 700 nos ) | $150.0K | 3 |
| Ryzodeg penfill h l medicine batch no ps6mv78 exp dt 02 | $150.0K | 3 |
| Human intravenous immunoglobulin 5 pct | $131.1K | 3 |
| Airtec fb 400 instacaps 30 s ( h l | $129.2K | 3 |
| Thyronorm 50 MCG 120s pill dose ( h l | $128.7K | 13 |
Pharmachem Private Limited imports 2,584 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Pharmachem Private Limited Import?
Top Products by Import Value
Pharmachem Private Limited Therapeutic Categories — 4 Specializations
Pharmachem Private Limited imports across 4 therapeutic categories, with Advanced Oncology (63.4%), Brand Names & OTC Products (21.5%), Biologics & Immunotherapy (8.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
1 products · 63.4% · $179.0K
Brand Names & OTC Products
1 products · 21.5% · $60.7K
Biologics & Immunotherapy
1 products · 8.0% · $22.7K
Diabetes & Endocrine
1 products · 7.1% · $20.0K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Rituximab | Advanced Oncology | $179.0K | 16 | 0.9% | 12 |
| 2 | Brufen | Brand Names & OTC Products | $60.7K | 21 | 1.4% | 12 |
| 3 | Adalimumab | Biologics & Immunotherapy | $22.7K | 4 | 1.9% | 4 |
| 4 | Liraglutide | Diabetes & Endocrine | $20.0K | 5 | 0.5% | 5 |
Pharmachem Private Limited imports 4 pharmaceutical products across 4 categories into Nepal totaling $282.4K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Pharmachem Private Limited.
Request DemoPharmachem Private Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Pharmachem Private Limited, established in 1988, is a prominent pharmaceutical importer and distributor based in Birgunj, Nepal. As a member of the diversified "KABRA" Group of Companies, Pharmachem operates within a conglomerate engaged in sectors such as banking and finance, automobiles, petrochemicals, LPG production, pharmaceuticals, FMCG marketing, and real estate. The group's investments and assets exceed NPR 10 billion across various parts of Nepal.
Pharmachem's primary role in Nepal's pharmaceutical distribution network is that of an importer and wholesaler. The company sources a wide range of finished pharmaceutical formulations from India, including tablets, capsules, syrups, and injections, to meet the diverse healthcare needs of the Nepali market. This strategic sourcing enables Pharmachem to provide a comprehensive portfolio of pharmaceutical products to its clientele.
2Distribution Network
Pharmachem Private Limited operates its main office in Birgunj, Nepal, and has a branch office in Kathmandu. The Kathmandu office, initially established for regulatory purposes, is slated to expand into full-fledged sales and distribution operations in the near future. This expansion aims to enhance the efficient and reliable distribution of life-saving medications in the capital city.
While specific details about warehouse locations and logistics capabilities are not publicly disclosed, Pharmachem's strategic presence in both Birgunj and Kathmandu suggests a distribution network designed to effectively serve the central and western regions of Nepal. The company's plans to expand its operations in Kathmandu indicate a commitment to strengthening its logistics and distribution infrastructure to meet the growing demand for pharmaceutical products across the country.
3Industry Role
In Nepal's pharmaceutical supply chain, Pharmachem Private Limited functions primarily as an importer and wholesaler. By sourcing a diverse array of finished pharmaceutical formulations from India, the company plays a crucial role in ensuring the availability of essential medications throughout Nepal. This position allows Pharmachem to bridge the gap between international pharmaceutical manufacturers and the Nepali healthcare system, facilitating access to a wide range of therapeutic options for healthcare providers and patients alike.
Supplier Relationship Intelligence — Pharmachem Private Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Pharmachem Private Limited's sourcing strategy is characterized by a diversified supplier base, with 14 unique Indian suppliers contributing to its total import value of $282,000 USD across 46 shipments. The top five suppliers—Abbott India Limited, USV Private Limited, Glenmark Pharmaceuticals Limited, Reliance Life Sciences Private Limited, and Lupin Limited—collectively account for approximately 87.3% of the total import value. This indicates a strategic choice to maintain strong relationships with key suppliers while mitigating risks associated with over-reliance on a single source.
The shipment data reveals a stable and consistent relationship between Pharmachem and its suppliers. The high number of shipments from these suppliers suggests a well-established and reliable supply chain, which is essential for maintaining the steady availability of pharmaceutical products in the Nepali market. This diversified sourcing approach not only reduces potential supply chain disruptions but also allows Pharmachem to leverage the strengths and specialties of various suppliers to meet the diverse needs of the healthcare sector in Nepal.
2Supply Chain Resilience
Pharmachem Private Limited's supply chain exhibits resilience through its diversified sourcing strategy and established relationships with multiple Indian pharmaceutical manufacturers. The company's import data indicates a consistent flow of shipments from a range of suppliers, suggesting a robust and dependable supply chain capable of adapting to market demands and potential disruptions.
While specific details about backup suppliers and formulation diversity are not publicly available, the existing supplier base and the company's plans to expand its distribution network in Kathmandu indicate a proactive approach to enhancing supply chain resilience. By strengthening its logistics capabilities and broadening its supplier relationships, Pharmachem aims to ensure a continuous and reliable supply of pharmaceutical products to meet the healthcare needs of the Nepali population.
3Strategic Implications
Pharmachem Private Limited's diversified sourcing pattern positions the company favorably within Nepal's pharmaceutical market. By maintaining strong relationships with multiple Indian suppliers, Pharmachem mitigates risks associated with supply chain disruptions and enhances its ability to offer a comprehensive range of pharmaceutical products. This strategic approach not only strengthens Pharmachem's competitive position but also contributes to the overall stability and reliability of the pharmaceutical supply chain in Nepal.
For Indian exporters seeking to become alternative suppliers to Pharmachem, understanding the company's sourcing preferences and establishing reliable supply chains will be crucial. Demonstrating the ability to meet Pharmachem's quality standards, regulatory requirements, and logistical needs will enhance the prospects of forming successful partnerships. Additionally, aligning with Pharmachem's strategic goals, such as expanding distribution networks and enhancing supply chain resilience, can further strengthen the collaboration between Indian exporters and Pharmachem.
Importing Pharmaceuticals into Nepal — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Nepal
1Regulatory Authority & Framework
In Nepal, the Department of Drug Administration (DDA) serves as the primary regulatory authority overseeing the pharmaceutical sector. The DDA is responsible for the regulation of drugs, including their manufacture, import, export, sale, distribution, and quality control. The legal framework governing pharmaceuticals in Nepal is primarily established by the Drug Act 2035 (1978), which provides comprehensive guidelines for the pharmaceutical industry. Additional regulations, such as the Drug Registration Regulation 2038 (1981) and the Drug Standard Regulation 2043 (1986), further detail the procedures and standards for drug registration and quality assurance.
For Indian pharmaceutical exporters, understanding and complying with these regulations is essential for ensuring the legal importation and sale of their products in Nepal. The DDA's guidelines encompass various aspects, including product registration, quality standards, and labeling requirements, all of which must be met to facilitate the smooth entry of pharmaceutical products into the Nepali market.
2Import Licensing & GMP
Import licensing in Nepal is governed by the Department of Drug Administration (DDA), which requires all pharmaceutical products to be registered before they can be imported and sold. The registration process involves submitting detailed documentation, including product information, manufacturing details, and quality control measures, to ensure compliance with Nepal's regulatory standards.
Good Manufacturing Practice (GMP) certification is a critical component of the import licensing process. The DDA recognizes GMP certifications from reputable international bodies, such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian pharmaceutical exporters seeking to supply products to Pharmachem Private Limited must ensure that their manufacturing facilities hold valid GMP certifications from these recognized organizations. Additionally, obtaining wholesale distribution authorization from the DDA is necessary for the legal distribution of pharmaceutical products within Nepal.
3Quality & Labeling
Pharmaceutical products imported into Nepal are subject to stringent quality control measures. Batch testing is mandatory to verify the safety, efficacy, and quality of each batch before it reaches the market. Stability studies are also required to ensure that products maintain their intended quality throughout their shelf life. Labeling requirements stipulate that product labels must be in the Nepali language, providing essential information such as dosage instructions, side effects, and storage conditions. Serialization mandates may also be implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Nepal's pharmaceutical regulatory framework has undergone several updates to strengthen the quality and safety of imported drugs. The Department of Drug Administration (DDA) has introduced more stringent requirements for product registration, including comprehensive documentation and evidence of compliance with international quality standards. Additionally, there has been an increased emphasis on post-market surveillance to monitor the performance of pharmaceutical products in the market. These regulatory changes aim to enhance the overall quality of pharmaceuticals available to the Nepali population and ensure that imported products meet the highest standards of safety and efficacy.
Pharmachem Private Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Pharmachem Private Limited's product strategy focuses on importing a diverse range of pharmaceutical formulations across various therapeutic categories. The company's top five imported products—Rituximab, Brufen, Adalimumab, Liraglutide, and others—indicate a strategic emphasis on critical therapeutic areas such as oncology, pain management, immunology, and endocrinology. This selection aligns with the growing healthcare needs in Nepal, where there is an increasing demand for advanced treatments in these areas.
The market demand for these products is driven by factors such as an aging population, rising prevalence of chronic diseases, and greater awareness of advanced treatment options among healthcare providers and patients. By focusing on these therapeutic areas, Pharmachem aims to address significant health challenges in Nepal and contribute to improving patient outcomes through access to essential medications.
2Sourcing Profile
Pharmachem Private Limited's sourcing strategy is centered on importing finished pharmaceutical formulations from India, a country known for its robust pharmaceutical manufacturing sector. The company's portfolio includes a wide array of products, indicating a preference for sourcing generic drugs that offer cost-effective alternatives to branded medications. This approach allows Pharmachem to provide a comprehensive range of therapeutic options to the Nepali market while maintaining affordability.
India's pharmaceutical
Frequently Asked Questions — Pharmachem Private Limited
What products does Pharmachem Private Limited import from India?
Pharmachem Private Limited imports 4 pharmaceutical products across 4 categories. Top imports: Rituximab ($179.0K), Brufen ($60.7K), Adalimumab ($22.7K), Liraglutide ($20.0K).
Who supplies pharmaceuticals to Pharmachem Private Limited from India?
Pharmachem Private Limited sources from 14 verified Indian suppliers. The primary supplier is Abbott India Limited (30.9% of imports, $11.6M).
What is Pharmachem Private Limited's total pharmaceutical import value?
Pharmachem Private Limited's total pharmaceutical import value from India is $282.4K, based on 46 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Pharmachem Private Limited focus on?
Pharmachem Private Limited imports across 4 categories. The largest: Advanced Oncology (63.4%), Brand Names & OTC Products (21.5%), Biologics & Immunotherapy (8.0%).
Get Full Pharmachem Private Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Pharmachem Private Limited identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Pharmachem Private Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 46 individual customs records matching Pharmachem Private Limited.
- 5.Supplier Verification: Pharmachem Private Limited sources from 14 verified Indian suppliers across 2,584 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.