Nutra MED Packaging Inc
Pharmaceutical Importer · United States · Respiratory Focus · $2.4M Total Trade · DGFT Verified
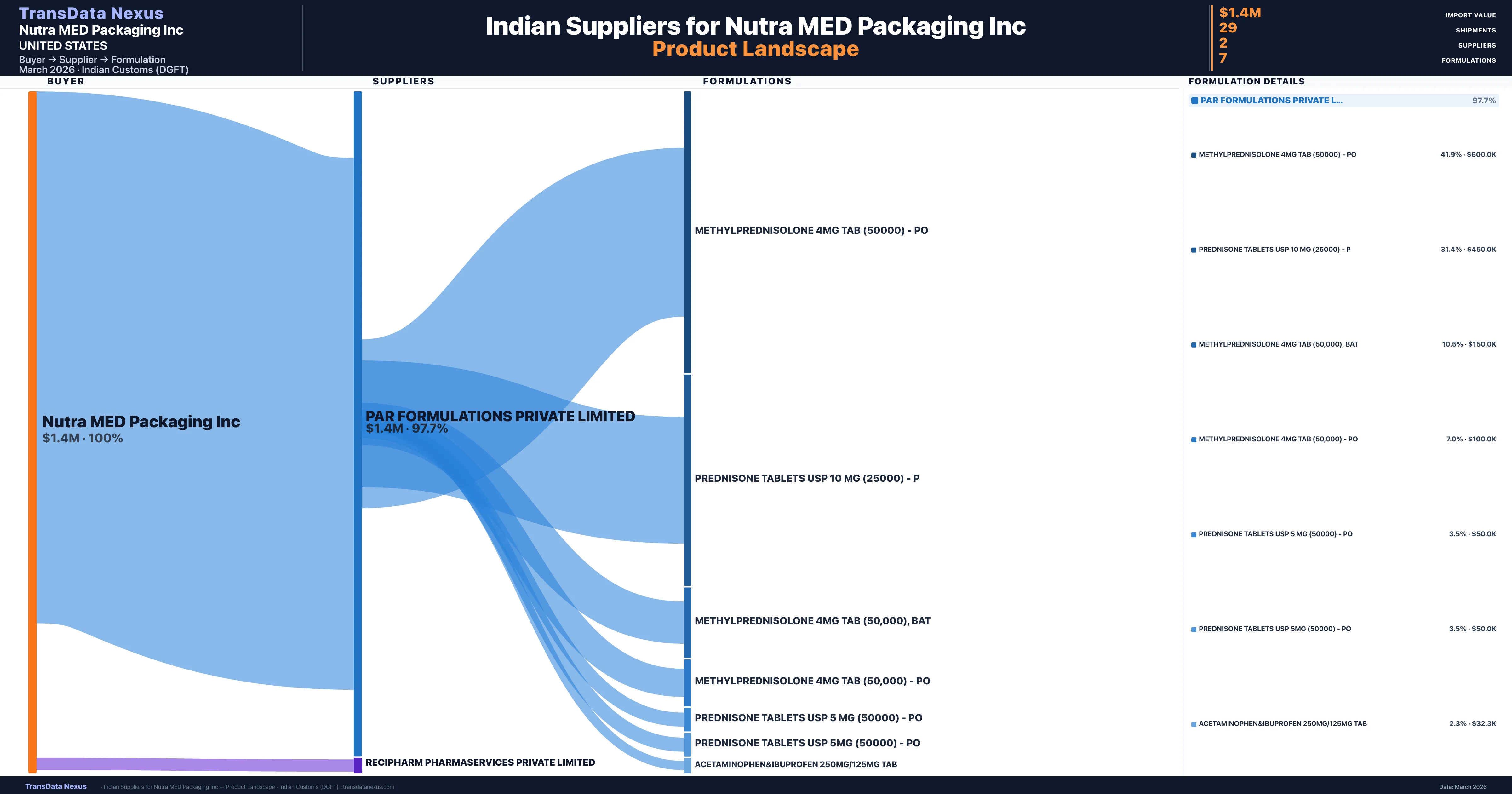
Nutra MED Packaging Inc is a pharmaceutical importer based in United States with a total trade value of $2.4M across 3 products in 2 therapeutic categories. Based on 47 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Nutra MED Packaging Inc sources from 2 verified Indian suppliers, with Par Formulations Private Limited accounting for 97.7% of imports.
Nutra MED Packaging Inc — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Nutra MED Packaging Inc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Par Formulations Private Limited | $1.4M | 28 | 97.7% |
| Recipharm Pharmaservices Private Limited | $32.3K | 1 | 2.3% |
Nutra MED Packaging Inc sources from 2 verified Indian suppliers across 7 distinct formulations. The sourcing is highly concentrated — Par Formulations Private Limited accounts for 97.7% of total imports, indicating a strategic single-source relationship.
What Formulations Does Nutra MED Packaging Inc Import?
| Formulation | Value | Ships |
|---|---|---|
| Methylprednisolone 4MG TAB (50000) - | $600.0K | 12 |
| Prednisone tablets USP 10 MG (25000) - | $450.0K | 9 |
| Methylprednisolone 4MG TAB (50,000) | $150.0K | 3 |
| Methylprednisolone 4MG TAB (50,000) - | $100.0K | 2 |
| Prednisone tablets USP 5 MG (50000) - | $50.0K | 1 |
| Prednisone tablets USP 5MG (50000) - | $50.0K | 1 |
| Acetaminophen&ibuprofen 250MG/125MG TAB | $32.3K | 1 |
Nutra MED Packaging Inc imports 7 distinct pharmaceutical formulations. Showing top 7 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Nutra MED Packaging Inc Import?
Top Products by Import Value
Nutra MED Packaging Inc Therapeutic Categories — 2 Specializations
Nutra MED Packaging Inc imports across 2 therapeutic categories, with Respiratory (59.6%), Corticosteroids (40.4%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Respiratory
2 products · 59.6% · $1.4M
Corticosteroids
1 products · 40.4% · $950.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Methylprednisolone | Corticosteroids | $950.0K | 19 | 2.9% | 2 |
| 2 | Prednisolone | Respiratory | $850.0K | 17 | 0.9% | 6 |
| 3 | Prednisone | Respiratory | $550.0K | 11 | 2.0% | 4 |
Nutra MED Packaging Inc imports 3 pharmaceutical products across 2 categories into United States totaling $2.4M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Nutra MED Packaging Inc.
Request DemoNutra MED Packaging Inc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Nutra-Med Packaging Inc, established in 1999 by Mahesh Gupta, is a contract packaging company specializing in the pharmaceutical, health & wellness, and medical device sectors. Headquartered in Whippany, New Jersey, the company offers a comprehensive suite of packaging services, including bottling, blistering, and secondary packaging such as cartoning, shrink sleeving, heat seal carding, kitting, and retail pallet pack-out. These services cater to both prescription (Rx) and over-the-counter (OTC) products, as well as dietary supplements.
In February 2022, GenNx360 Capital Partners, a New York City-based private equity firm, invested in Nutra-Med Packaging. This partnership aims to support the company's growth initiatives and operational execution. As of January 2026, Hany Salama assumed the role of Chief Executive Officer, bringing over 20 years of global experience in healthcare, biopharma, consumer health, and life sciences.
2Distribution Network
Nutra-Med Packaging operates four centrally located facilities within a half-mile radius in New Jersey: Whippany, Hanover, and East Hanover. These facilities collectively encompass over 215,000 square feet, with 50,000 square feet dedicated to production and 123,000 square feet for warehousing of finished goods, bulk, and components. This strategic proximity enhances operational efficiency and facilitates rapid service delivery.
3Industry Role
Nutra-Med Packaging Inc functions as a contract packaging provider within the United States pharmaceutical supply chain. By offering specialized packaging services, the company supports pharmaceutical manufacturers in delivering their products to the market efficiently and in compliance with regulatory standards. This role is crucial for ensuring that pharmaceutical products are presented in a manner that maintains their integrity and meets consumer expectations.
Supplier Relationship Intelligence — Nutra MED Packaging Inc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Nutra-Med Packaging's sourcing from India is highly concentrated, with 97.7% of its imports originating from PAR Formulations Private Limited. This significant dependency on a single supplier may pose risks related to supply chain disruptions, quality control, and pricing negotiations. The limited number of shipments (47) and the dominance of a single supplier suggest a strategic choice to maintain consistency and quality, but it also indicates potential vulnerabilities in the supply chain.
2Supply Chain Resilience
The resilience of Nutra-Med Packaging's Indian supply chain appears limited due to the heavy reliance on a single supplier, PAR Formulations Private Limited. The absence of backup suppliers and the narrow range of imported formulations (7 unique products) suggest a lack of diversification. This concentration could lead to challenges in mitigating risks associated with supplier disruptions, regulatory changes, or quality issues. Additionally, the exposure to specific shipping routes and the regulatory compliance status of the sole supplier further impact the supply chain's robustness.
3Strategic Implications
Nutra-Med Packaging's concentrated sourcing strategy may offer benefits such as streamlined operations and stronger supplier relationships. However, it also exposes the company to significant risks, including supply chain disruptions and limited flexibility in product offerings. For Indian exporters, this presents an opportunity to diversify their client base by offering alternative products or services that align with Nutra-Med's needs, potentially reducing the company's dependency on a single supplier.
Importing Pharmaceuticals into United States — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United States
1Regulatory Authority & Framework
In the United States, the Food and Drug Administration (FDA) is the primary regulatory authority overseeing the importation of pharmaceutical products. Key legislation governing these imports includes the Federal Food, Drug, and Cosmetic Act (FDCA), which sets standards for drug safety, efficacy, and labeling. The FDA's Center for Drug Evaluation and Research (CDER) is responsible for evaluating and approving new drugs, including those imported from foreign markets.
2Import Licensing & GMP
Importers of pharmaceutical products into the United States must ensure that their foreign suppliers are registered with the FDA and that their facilities comply with Good Manufacturing Practice (GMP) standards. The FDA recognizes various GMP certifications, including those from the European Union (EU GMP), World Health Organization (WHO GMP), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers are also required to obtain an import license and ensure that all imported drugs are properly labeled and meet FDA standards.
3Quality & Labeling
Imported pharmaceutical products must adhere to FDA labeling requirements, which include accurate and truthful information in English. This encompasses details such as drug identity, strength, dosage form, and directions for use. Batch testing and stability studies are essential to ensure product quality and efficacy. Additionally, serialization mandates require that each saleable unit of prescription drugs be assigned a unique serial number to enhance traceability and prevent counterfeit products from entering the supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, the FDA has implemented several policy changes affecting the importation of pharmaceutical products from India. These include stricter enforcement of GMP compliance, enhanced scrutiny of drug labeling and packaging, and the introduction of new serialization requirements to combat counterfeit drugs. Importers must stay informed about these changes to ensure continued compliance and avoid potential disruptions in their supply chains.
Nutra MED Packaging Inc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Nutra-Med Packaging's focus on respiratory and corticosteroid products aligns with market demands for treatments addressing respiratory conditions and inflammation. The significant import values of methylprednisolone, prednisolone, and prednisone indicate a strategic emphasis on these therapeutic areas, likely driven by consistent demand and the company's expertise in packaging such formulations.
2Sourcing Profile
Nutra-Med Packaging's sourcing strategy is heavily concentrated on generic drug formulations, particularly corticosteroids. The company's reliance on a single supplier in India suggests a preference for established relationships and consistent product quality. However, this approach may limit flexibility and expose the company to risks associated with supply chain disruptions or regulatory changes affecting the sole supplier.
3Market Positioning
Based on its product mix, Nutra-Med Packaging serves a segment of the United States market that includes retail pharmacies, hospitals, and government tenders. The company's focus on packaging services for both prescription and over-the-counter products positions it to meet the needs of various stakeholders in the pharmaceutical supply chain, ensuring that medications are delivered to end-users in optimal condition.
Seller's Guide — How to Become a Supplier to Nutra MED Packaging Inc
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Nutra-Med Packaging by offering alternative products or services that align with the company's packaging capabilities and market focus. Diversifying Nutra-Med's supplier base could mitigate risks associated with single-source dependency and enhance supply chain resilience. Identifying gaps in Nutra-Med's current sourcing, such as the need for additional product formulations or packaging solutions, presents avenues for collaboration.
2Requirements & Qualifications
Indian exporters seeking to supply Nutra-Med Packaging and the U.S. market must ensure that their manufacturing facilities are registered with the FDA and comply with recognized GMP standards, such as EU GMP, WHO GMP, or PIC/S. Additionally, products must meet FDA labeling requirements, including accurate and truthful information in English. Compliance with serialization mandates is also essential to facilitate traceability and prevent counterfeit products.
3How to Approach
Indian exporters should initiate engagement with Nutra-Med Packaging by demonstrating their compliance with FDA regulations and GMP standards. Participating in industry trade shows, establishing direct communication channels, and presenting product portfolios that align with Nutra-Med's packaging capabilities can facilitate relationship building. Understanding the regulatory filing process and timelines for FDA approvals is crucial for effective collaboration. Patience and persistence are key, as establishing new supplier relationships can be a time-consuming process.
Frequently Asked Questions — Nutra MED Packaging Inc
What products does Nutra MED Packaging Inc import from India?
Nutra MED Packaging Inc imports 3 pharmaceutical products across 2 categories. Top imports: Methylprednisolone ($950.0K), Prednisolone ($850.0K), Prednisone ($550.0K).
Who supplies pharmaceuticals to Nutra MED Packaging Inc from India?
Nutra MED Packaging Inc sources from 2 verified Indian suppliers. The primary supplier is Par Formulations Private Limited (97.7% of imports, $1.4M).
What is Nutra MED Packaging Inc's total pharmaceutical import value?
Nutra MED Packaging Inc's total pharmaceutical import value from India is $2.4M, based on 47 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Nutra MED Packaging Inc focus on?
Nutra MED Packaging Inc imports across 2 categories. The largest: Respiratory (59.6%), Corticosteroids (40.4%).
Get Full Nutra MED Packaging Inc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Nutra MED Packaging Inc identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Nutra MED Packaging Inc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 47 individual customs records matching Nutra MED Packaging Inc.
- 5.Supplier Verification: Nutra MED Packaging Inc sources from 2 verified Indian suppliers across 7 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.