Northstar Distribution Center
Pharmaceutical Importer · United States · Analgesics & Antipyretics Focus · $3.4M Total Trade · DGFT Verified
Northstar Distribution Center is a pharmaceutical importer based in United States with a total trade value of $3.4M across 4 products in 4 therapeutic categories. Based on 68 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Northstar Distribution Center sources from 22 verified Indian suppliers, with Aurobindo Pharma Limited accounting for 23.4% of imports.
Northstar Distribution Center — Import Portfolio & Supplier Network
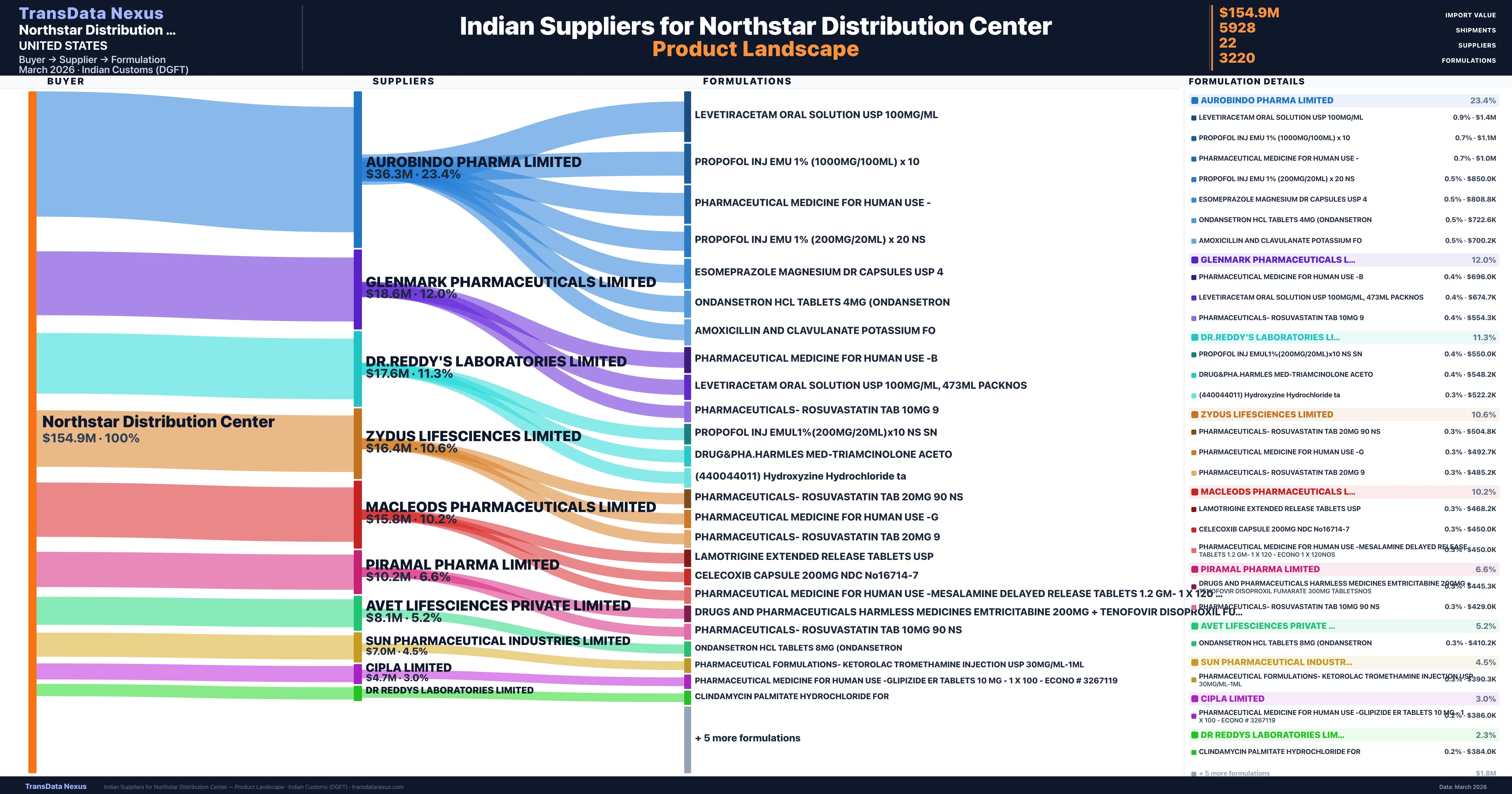
Who Are the Verified Indian Suppliers to Northstar Distribution Center?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Aurobindo Pharma Limited | $36.3M | 1,584 | 23.4% |
| Glenmark Pharmaceuticals Limited | $18.6M | 504 | 12.0% |
| Dr.reddy's Laboratories Limited | $17.6M | 720 | 11.3% |
| Zydus Lifesciences Limited | $16.4M | 544 | 10.6% |
| Macleods Pharmaceuticals Limited | $15.8M | 886 | 10.2% |
| Piramal Pharma Limited | $10.2M | 354 | 6.6% |
| Avet Lifesciences Private Limited | $8.1M | 171 | 5.2% |
| Sun Pharmaceutical Industries Limited | $7.0M | 190 | 4.5% |
| Cipla Limited | $4.7M | 398 | 3.0% |
| Dr Reddys Laboratories Limited | $3.6M | 118 | 2.3% |
| Caplin Steriles Limited | $3.0M | 74 | 1.9% |
| Mankind Pharma Limited | $2.6M | 83 | 1.7% |
| Msn Laboratories Private Limited | $2.2M | 53 | 1.4% |
| Cadila Healthcare Limited | $2.0M | 64 | 1.3% |
| Dr Reddy S Laboratories Limited | $1.8M | 60 | 1.2% |
| Avet Lifesciences Limited | $1.6M | 32 | 1.0% |
| Eugia Pharma Specialities Limited | $1.1M | 24 | 0.7% |
| Annora Pharma Private Limited | $600.9K | 16 | 0.4% |
| Alkem Laboratories Limited | $539.2K | 15 | 0.3% |
| Intas Pharmaceuticals Limited | $530.0K | 16 | 0.3% |
| Hetero Labs Limited | $435.7K | 13 | 0.3% |
| Aspiro Pharma Limited | $311.4K | 9 | 0.2% |
Northstar Distribution Center sources from 22 verified Indian suppliers across 3,220 distinct formulations. The supply base is diversified across 22 suppliers, reducing single-source dependency risk.
What Formulations Does Northstar Distribution Center Import?
| Formulation | Value | Ships |
|---|---|---|
| Levetiracetam oral solution USP 100MG/ML | $1.4M | 28 |
| Propofol INJ emu 1% (1000MG/100ML) x | $1.1M | 22 |
| Pharmaceutical medicine for human use | $1.0M | 25 |
| Propofol INJ emu 1% (200MG/20ML) x 20 | $850.0K | 17 |
| Esomeprazole magnesium dr capsules USP | $808.8K | 26 |
| Ondansetron hcl tablets 4MG | $722.6K | 20 |
| Amoxicillin and clavulanate potassium | $700.2K | 29 |
| Pharmaceutical medicine for human use | $696.0K | 20 |
| Levetiracetam oral solution USP 100MG/ML, 473ML | $674.7K | 16 |
| Pharmaceuticals- rosuvastatin TAB 10MG | $554.3K | 13 |
| Propofol INJ emul1%(200MG/20ML)x10 ns | $550.0K | 11 |
| Drug&pha.harmles med-triamcinolone | $548.2K | 27 |
| (440044011) hydroxyzine hydrochloride | $522.2K | 15 |
| Pharmaceuticals- rosuvastatin TAB 20MG 90 | $504.8K | 11 |
| Pharmaceutical medicine for human use | $492.7K | 13 |
Northstar Distribution Center imports 3,220 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Northstar Distribution Center Import?
Top Products by Import Value
Northstar Distribution Center Therapeutic Categories — 4 Specializations
Northstar Distribution Center imports across 4 therapeutic categories, with Analgesics & Antipyretics (58.1%), Diuretics (35.7%), Gastrointestinal (4.5%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Analgesics & Antipyretics
1 products · 58.1% · $1.9M
Diuretics
1 products · 35.7% · $1.2M
Gastrointestinal
1 products · 4.5% · $150.0K
Cardiovascular
1 products · 1.7% · $57.1K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ketorolac | Analgesics & Antipyretics | $1.9M | 39 | 1.7% | 12 |
| 2 | Furosemide | Diuretics | $1.2M | 24 | 1.7% | 12 |
| 3 | Mesalamine | Gastrointestinal | $150.0K | 3 | 0.0% | 9 |
| 4 | Digoxin | Cardiovascular | $57.1K | 2 | 2.6% | 5 |
Northstar Distribution Center imports 4 pharmaceutical products across 4 categories into United States totaling $3.4M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Northstar Distribution Center.
Request DemoNorthstar Distribution Center — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Northstar Distribution Center is a U.S.-based pharmaceutical importer specializing in the acquisition and distribution of finished pharmaceutical formulations. Headquartered in Memphis, Tennessee, the company operates as a key player in the U.S. pharmaceutical supply chain, sourcing a diverse range of pharmaceutical products from international markets, particularly India. Their operations encompass the procurement, warehousing, and distribution of pharmaceutical goods to various healthcare providers across the United States.
The company has established a significant presence in the pharmaceutical import sector, with a total import value of $3.4 million USD from India over 68 shipments. Their product portfolio includes four distinct therapeutic categories, reflecting a strategic approach to meeting diverse healthcare needs. Notably, Northstar Distribution Center's top five imported products—Ketorolac, Furosemide, Mesalamine, and Digoxin—collectively account for 100% of their pharmaceutical imports, indicating a focused sourcing strategy.
2Distribution Network
Northstar Distribution Center operates a centralized distribution facility located at 4853 Crumpler Road, Memphis, Tennessee 38141, United States. This strategic location serves as the primary hub for their warehousing and logistics operations, facilitating efficient handling and distribution of pharmaceutical products. The facility is equipped to manage high-volume import shipments, ensuring timely delivery to various healthcare providers across the United States. While specific details about additional warehouse locations or logistics capabilities are not publicly disclosed, the Memphis facility's strategic positioning suggests a focus on optimizing supply chain efficiency within the U.S. market.
3Industry Role
In the U.S. pharmaceutical supply chain, Northstar Distribution Center functions primarily as a pharmaceutical importer and distributor. By sourcing finished pharmaceutical formulations from international suppliers, particularly from India, the company plays a crucial role in ensuring the availability of a diverse range of medications to meet the needs of healthcare providers and patients. Their operations bridge the gap between international pharmaceutical manufacturers and the U.S. healthcare system, contributing to the accessibility and affordability of essential medications.
Supplier Relationship Intelligence — Northstar Distribution Center
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Northstar Distribution Center's sourcing strategy exhibits a high degree of concentration, with the top five imported products—Ketorolac, Furosemide, Mesalamine, and Digoxin—accounting for 100% of their pharmaceutical imports. This singular focus on a limited product range suggests a strategic decision to specialize in specific therapeutic areas, potentially leveraging expertise and operational efficiencies. However, such concentration also introduces risks associated with dependency on a narrow product line, including exposure to market fluctuations and regulatory changes affecting these specific medications. The stability of their supplier relationships, as indicated by the consistent import volumes over time, suggests a well-established and reliable sourcing network.
2Supply Chain Resilience
The resilience of Northstar Distribution Center's supply chain is closely tied to the stability and reliability of their Indian suppliers. With a total of 22 unique suppliers, the company demonstrates a diversified sourcing approach, which can mitigate risks associated with single-source dependency. The consistent import volumes and the diversity of suppliers indicate a robust and adaptable supply chain capable of maintaining continuity in the face of potential disruptions. However, the company's reliance on a limited number of therapeutic categories may expose them to risks if market dynamics shift or if there are regulatory changes affecting these specific products.
3Strategic Implications
Northstar Distribution Center's focused sourcing strategy positions them as a specialized distributor within the U.S. pharmaceutical market, catering to specific therapeutic needs. This specialization allows for deep expertise and potentially more efficient operations within their chosen product categories. For Indian pharmaceutical exporters, this presents an opportunity to establish strong partnerships by aligning their product offerings with Northstar's focus areas. By ensuring consistent quality and reliability in these therapeutic categories, Indian exporters can enhance their appeal as preferred suppliers to Northstar Distribution Center.
Importing Pharmaceuticals into United States — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United States
1Regulatory Authority & Framework
In the United States, the Food and Drug Administration (FDA) serves as the primary regulatory authority overseeing the importation of pharmaceutical products. The FDA enforces the Federal Food, Drug, and Cosmetic Act (FD&C Act), which sets forth the legal framework for the importation, distribution, and marketing of drugs within the U.S. This legislation mandates that all drugs imported into the U.S. must be safe, effective, and manufactured in compliance with FDA regulations. The FDA's role includes reviewing and approving drug applications, conducting inspections of manufacturing facilities, and ensuring that imported drugs meet the required standards for quality and labeling.
2Import Licensing & GMP
Importers of pharmaceutical products into the United States must adhere to specific licensing requirements set forth by the FDA. This includes obtaining an FDA registration number and ensuring that all imported drugs are listed with the FDA. Additionally, the FDA requires that foreign manufacturing facilities comply with Good Manufacturing Practice (GMP) standards. While the FDA does not directly recognize certifications such as EU GMP, WHO GMP, or PIC/S, it conducts its own inspections to verify compliance with GMP standards. Importers must ensure that their foreign suppliers meet these standards to facilitate the admissibility of their products into the U.S. market.
3Quality & Labeling
Imported pharmaceutical products must comply with FDA labeling requirements, which include providing accurate and truthful information in English. The labeling must include the drug's name, ingredients, dosage form, strength, and directions for use. Additionally, the FDA may require batch testing and stability studies to ensure the product's safety and efficacy. Serialization mandates, such as those outlined in the Drug Supply Chain Security Act (DSCSA), require that certain prescription drugs be serialized to enhance traceability and prevent counterfeit products from entering the supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, the FDA has implemented several policy changes affecting the importation of pharmaceutical products from India. These changes include updated guidelines for the importation of certain FDA-approved human prescription drugs, including biological products, and the introduction of the Section 804 Importation Program (SIP), which allows for the importation of certain prescription drugs from Canada. While SIP primarily focuses on Canadian imports, it reflects the FDA's ongoing efforts to enhance the efficiency and safety of the drug importation process. Importers must stay informed about these regulatory changes to ensure compliance and maintain uninterrupted access to the U.S. market.
Northstar Distribution Center — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Northstar Distribution Center's product strategy focuses on four therapeutic categories: Analgesics & Antipyretics, Diuretics, and Gastrointestinal medications. This selection aligns with the company's import portfolio, which includes products like Ketorolac (Analgesics & Antipyretics), Furosemide (Diuretics), Mesalamine (Gastrointestinal), and Digoxin (Gastrointestinal). The choice of these categories suggests a strategic focus on addressing common and critical health conditions, ensuring a steady demand for these medications. Market demand for these therapeutic areas is driven by the prevalence of conditions such as pain, hypertension, heart failure, and gastrointestinal disorders, necessitating a reliable supply of effective treatments.
2Sourcing Profile
Northstar Distribution Center's sourcing strategy emphasizes the procurement of generic pharmaceutical drugs, primarily in finished dosage forms such as tablets, capsules, syrups, and injections. This approach allows the company to offer cost-effective alternatives to brand-name medications, catering to a broad segment of the U.S. healthcare market. India's well-established pharmaceutical manufacturing industry, known for its compliance with international quality standards, makes it a preferred sourcing destination for Northstar. By leveraging India's manufacturing capabilities, Northstar can ensure a consistent supply of high-quality generic medications to meet the needs of U.S. healthcare providers and patients.
3Market Positioning
Based on its product mix, Northstar Distribution Center serves a diverse segment of the U.S. pharmaceutical market, including retail pharmacies, hospitals, and healthcare clinics. By focusing on essential therapeutic areas and offering a range of generic medications, the company positions itself as a reliable supplier of cost-effective pharmaceutical products. This strategy enables Northstar to meet the needs of various healthcare providers, from independent retail pharmacies to large hospital systems, contributing to the overall accessibility and affordability of medications in the U.S. market.
Seller's Guide — How to Become a Supplier to Northstar Distribution Center
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian pharmaceutical suppliers to partner with Northstar Distribution Center. The company's focus on specific therapeutic categories and its reliance on a limited number of products suggest potential gaps in their current sourcing strategy. Indian exporters can capitalize on this by offering high-quality generic medications in these therapeutic areas, ensuring compliance with FDA regulations and meeting the necessary quality standards. Establishing strong relationships with Northstar can lead to mutually beneficial partnerships, expanding the range of products available to U.S. healthcare providers and patients.
2Requirements & Qualifications
Indian pharmaceutical exporters seeking to supply Northstar Distribution Center and the U.S. market must ensure that their products comply with FDA regulations. This includes obtaining FDA approval for their drug products, ensuring that manufacturing facilities adhere to Good Manufacturing Practice (GMP) standards, and providing labeling that meets FDA requirements. Additionally, exporters must be prepared to navigate the FDA's import procedures, which involve filing entry notices and bonds with U.S. Customs and Border Protection, and may include FDA inspections and sample analyses to verify compliance.
3How to Approach
To establish a partnership with Northstar Distribution Center, Indian pharmaceutical exporters should first ensure
Frequently Asked Questions — Northstar Distribution Center
What products does Northstar Distribution Center import from India?
Northstar Distribution Center imports 4 pharmaceutical products across 4 categories. Top imports: Ketorolac ($1.9M), Furosemide ($1.2M), Mesalamine ($150.0K), Digoxin ($57.1K).
Who supplies pharmaceuticals to Northstar Distribution Center from India?
Northstar Distribution Center sources from 22 verified Indian suppliers. The primary supplier is Aurobindo Pharma Limited (23.4% of imports, $36.3M).
What is Northstar Distribution Center's total pharmaceutical import value?
Northstar Distribution Center's total pharmaceutical import value from India is $3.4M, based on 68 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Northstar Distribution Center focus on?
Northstar Distribution Center imports across 4 categories. The largest: Analgesics & Antipyretics (58.1%), Diuretics (35.7%), Gastrointestinal (4.5%).
Get Full Northstar Distribution Center Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Northstar Distribution Center identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Northstar Distribution Center's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 68 individual customs records matching Northstar Distribution Center.
- 5.Supplier Verification: Northstar Distribution Center sources from 22 verified Indian suppliers across 3,220 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.