Nextpharma Logistics Gmbh
Pharmaceutical Importer · Germany · Diabetes & Endocrine Focus · $3.9M Total Trade · DGFT Verified
Nextpharma Logistics Gmbh is a pharmaceutical importer based in Germany with a total trade value of $3.9M across 3 products in 2 therapeutic categories. Based on 182 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Nextpharma Logistics Gmbh sources from 5 verified Indian suppliers, with Dr.reddy's Laboratories Limited accounting for 67.1% of imports.
Nextpharma Logistics Gmbh — Import Portfolio & Supplier Network
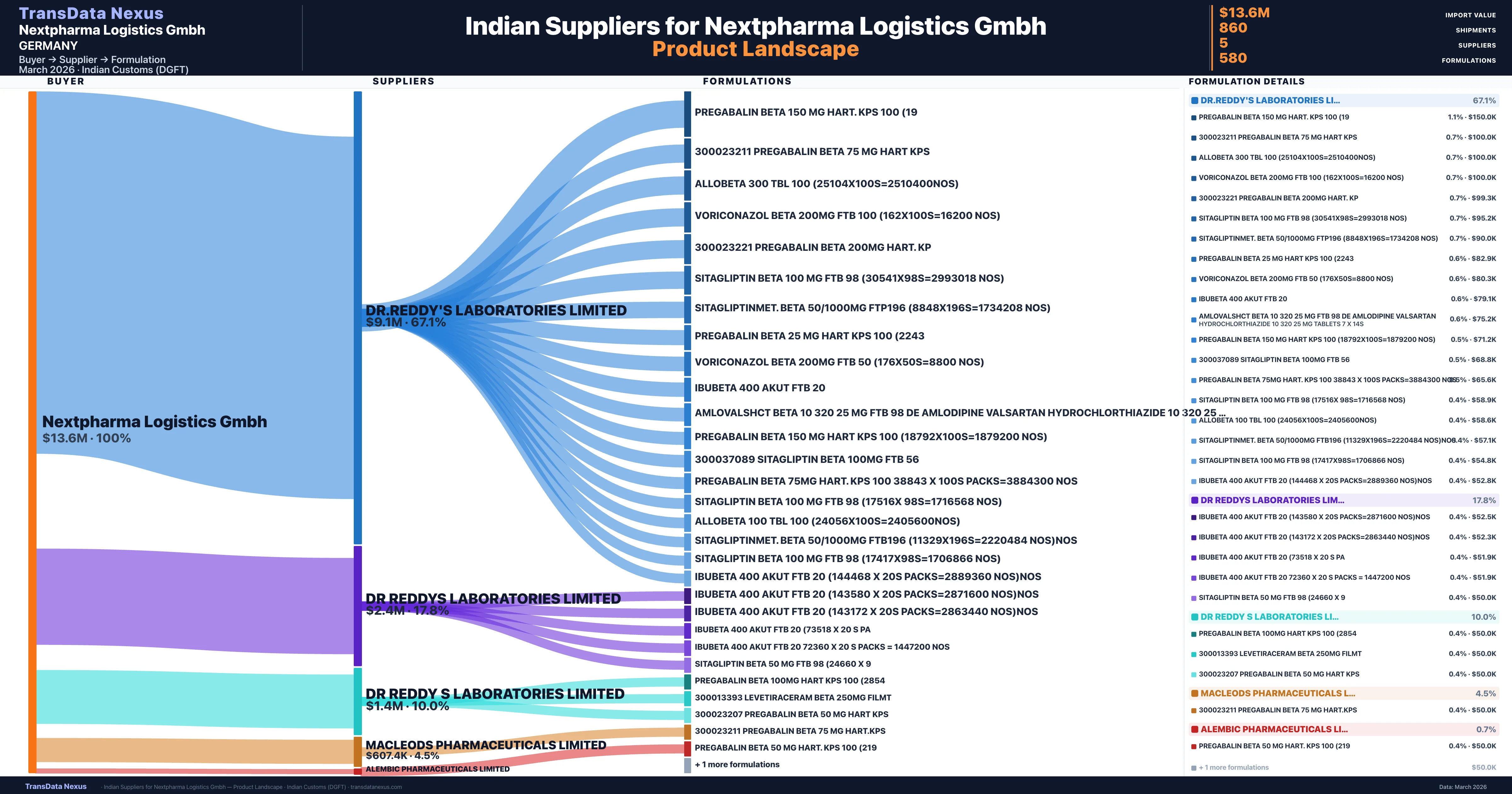
Who Are the Verified Indian Suppliers to Nextpharma Logistics Gmbh?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Dr.reddy's Laboratories Limited | $9.1M | 675 | 67.1% |
| Dr Reddys Laboratories Limited | $2.4M | 93 | 17.8% |
| Dr Reddy S Laboratories Limited | $1.4M | 47 | 10.0% |
| Macleods Pharmaceuticals Limited | $607.4K | 39 | 4.5% |
| Alembic Pharmaceuticals Limited | $94.9K | 6 | 0.7% |
Nextpharma Logistics Gmbh sources from 5 verified Indian suppliers across 580 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does Nextpharma Logistics Gmbh Import?
| Formulation | Value | Ships |
|---|---|---|
| Pregabalin beta 150 MG hart. kps 100 | $150.0K | 3 |
| 300023211 pregabalin beta 75 MG hart | $100.0K | 2 |
| Allobeta 300 tbl 100 (25104x100s=2510400nos) | $100.0K | 2 |
| Voriconazol beta 200MG ftb 100 (162x100s=16200 nos) | $100.0K | 2 |
| 300023221 pregabalin beta 200MG hart. | $99.3K | 3 |
| Sitagliptin beta 100 MG ftb 98 (30541x98s=2993018 nos) | $95.2K | 3 |
| Sitagliptinmet. beta 50/1000MG ftp196 (8848x196s=1734208 nos) | $90.0K | 2 |
| Pregabalin beta 25 MG hart kps 100 | $82.9K | 2 |
| Voriconazol beta 200MG ftb 50 (176x50s=8800 nos) | $80.3K | 2 |
| Ibubeta 400 akut ftb | $79.1K | 2 |
| Amlovalshct beta 10 320 25 MG ftb 98 de amlodipine valsartan hydrochlorthiazide 10 320 25 MG tablets 7 x 14s | $75.2K | 2 |
| Pregabalin beta 150 MG hart kps 100 (18792x100s=1879200 nos) | $71.2K | 2 |
| 300037089 sitagliptin beta 100MG ftb | $68.8K | 2 |
| Pregabalin beta 75MG hart. kps 100 38843 x 100s packs=3884300 | $65.6K | 2 |
| Sitagliptin beta 100 MG ftb 98 (17516x 98s=1716568 nos) | $58.9K | 3 |
Nextpharma Logistics Gmbh imports 580 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Nextpharma Logistics Gmbh Import?
Top Products by Import Value
Nextpharma Logistics Gmbh Therapeutic Categories — 2 Specializations
Nextpharma Logistics Gmbh imports across 2 therapeutic categories, with Diabetes & Endocrine (71.6%), Cardiovascular (28.4%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Diabetes & Endocrine
1 products · 71.6% · $2.8M
Cardiovascular
2 products · 28.4% · $1.1M
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Sitagliptin | Diabetes & Endocrine | $2.8M | 160 | 3.7% | 7 |
| 2 | Amlodipine | Cardiovascular | $550.0K | 11 | 0.0% | 7 |
| 3 | Valsartan | Cardiovascular | $550.0K | 11 | 0.3% | 4 |
Nextpharma Logistics Gmbh imports 3 pharmaceutical products across 2 categories into Germany totaling $3.9M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Nextpharma Logistics Gmbh.
Request DemoNextpharma Logistics Gmbh — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
NextPharma Logistics GmbH, established in 1992, was a prominent pharmaceutical logistics company headquartered in Bielefeld, North Rhine-Westphalia, Germany. The company specialized in providing comprehensive logistics services tailored for the healthcare sector, including storage, distribution, and transportation of pharmaceutical, medical, cosmetic, and dietary products. Serving clients across Germany, Austria, Switzerland, and beyond, NextPharma Logistics was recognized as one of the leading pharmaceutical logistics providers in the D-A-CH region.
In October 2025, NextPharma announced the sale of its logistics arm to Cencora, Inc., a global pharmaceutical solutions organization. This strategic divestment aimed to refocus NextPharma's efforts on its core competencies as a contract development and manufacturing organization (CDMO), positioning the company as Europe's leading pure-play CDMO. The transaction was completed in December 2025.
2Distribution Network
Prior to its acquisition, NextPharma Logistics GmbH operated a robust distribution network with multiple facilities across the D-A-CH region. The company's headquarters were located in Bielefeld, North Rhine-Westphalia, Germany. Additional sites included Werne and Holzwickede in Germany, Garching bei München, and Schaffhausen in Switzerland. These strategically positioned facilities enabled NextPharma Logistics to offer efficient and reliable logistics solutions to its diverse clientele.
3Industry Role
NextPharma Logistics GmbH played a pivotal role in the pharmaceutical supply chain as a specialized logistics service provider. The company focused on the storage, distribution, and transportation of pharmaceutical products, ensuring compliance with stringent industry standards and regulations. By offering tailored logistics solutions, NextPharma Logistics supported various stakeholders in the healthcare sector, including pharmacies, hospitals, laboratories, and pharmaceutical wholesalers.
Supplier Relationship Intelligence — Nextpharma Logistics Gmbh
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
NextPharma Logistics GmbH's sourcing strategy was characterized by a high degree of concentration, with a significant portion of its imports originating from a single supplier, DR.REDDY'S LABORATORIES LIMITED. This supplier accounted for approximately 67.1% of the total import value, amounting to $9.1 million across 675 shipments. Such a concentrated sourcing approach can present risks, including potential supply chain disruptions and dependency on a single supplier. However, the substantial volume of transactions suggests a stable and long-term relationship, which may mitigate some of these risks. The remaining imports were diversified among other suppliers, including DR REDDYS LABORATORIES LIMITED, DR REDDY S LABORATORIES LIMITED, MACLEODS PHARMACEUTICALS LIMITED, and ALEMBIC PHARMACEUTICALS LIMITED.
2Supply Chain Resilience
The resilience of NextPharma Logistics GmbH's supply chain was influenced by its sourcing concentration. While the primary supplier, DR.REDDY'S LABORATORIES LIMITED, provided a significant volume of products, the company's diversified supplier base, including DR REDDYS LABORATORIES LIMITED, DR REDDY S LABORATORIES LIMITED, MACLEODS PHARMACEUTICALS LIMITED, and ALEMBIC PHARMACEUTICALS LIMITED, contributed to mitigating potential risks associated with single-source dependency. The company's ability to manage a diverse range of formulations, with 580 unique formulations imported, indicates a level of flexibility and adaptability in its supply chain operations. However, the reliance on a single supplier for a substantial portion of imports suggests that any disruptions affecting this supplier could have a significant impact on the company's operations.
3Strategic Implications
The concentrated sourcing pattern of NextPharma Logistics GmbH, particularly its significant reliance on DR.REDDY'S LABORATORIES LIMITED, has strategic implications for the company's competitive position. While the strong relationship with a major supplier may offer benefits such as favorable terms and consistent product quality, it also exposes the company to risks associated with supply chain disruptions. For Indian exporters seeking to become alternative suppliers, this presents an opportunity to diversify NextPharma Logistics' supplier base by offering competitive pricing, reliable delivery schedules, and high-quality products. Demonstrating compliance with international quality standards and the ability to meet the company's specific logistical requirements would be crucial in establishing a successful partnership.
Importing Pharmaceuticals into Germany — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Germany
1Regulatory Authority & Framework
In Germany, the primary regulatory authority overseeing pharmaceutical imports is the Federal Institute for Drugs and Medical Devices (BfArM). The key legislation governing pharmaceutical imports includes the German Medicines Act (AMG), which outlines the requirements for the importation, distribution, and marketing of medicinal products. The marketing authorization pathway for Indian generics involves obtaining approval from the European Medicines Agency (EMA) or through national procedures, ensuring that imported generics meet the safety, efficacy, and quality standards set by European regulations.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products in Germany stipulate that all medicinal products must be authorized for sale within the European Union. This involves obtaining a marketing authorization from the EMA or a national competent authority. Good Manufacturing Practice (GMP) certificates recognized by the European Union, such as EU GMP, WHO GMP, or PIC/S, are essential for ensuring product quality and safety. Additionally, wholesale distribution authorization is required for entities involved in the distribution of medicinal products, ensuring compliance with regulatory standards and maintaining the integrity of the pharmaceutical supply chain.
3Quality & Labeling
Pharmaceutical products imported into Germany must undergo batch testing to verify their quality, safety, and efficacy. Stability requirements ensure that products maintain their intended quality throughout their shelf life. Labeling must be in German and include essential information such as the product name, active ingredients, dosage form, and instructions for use. Serialization mandates require unique identifiers on packaging to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Germany implemented several regulatory changes affecting pharmaceutical imports. These included stricter compliance requirements for GMP certifications, enhanced batch testing protocols, and updated labeling and serialization standards to improve product traceability and safety. Additionally, there was an increased emphasis on sustainability, with regulations encouraging the reduction of carbon emissions and the adoption of environmentally friendly practices within the pharmaceutical supply chain.
Nextpharma Logistics Gmbh — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
NextPharma Logistics GmbH's product category focus on diabetes and endocrine (71.6%) and cardiovascular (28.4%) therapeutic areas aligns with the significant market demand for these medications. The substantial import value of sitagliptin ($2.8 million) and amlodipine ($550,000) indicates a strategic emphasis on high-demand products within these categories. This focus suggests that NextPharma Logistics aimed to address prevalent health conditions in Germany, catering to a large patient population and meeting the needs of healthcare providers.
2Sourcing Profile
NextPharma Logistics GmbH's sourcing strategy was predominantly centered on generic drugs, with a significant portion of imports originating from India. The company's preference for finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, indicates a focus on ready-to-market products that meet European quality standards. India's established pharmaceutical manufacturing capabilities and adherence to international quality certifications made it a key supplier for NextPharma Logistics, facilitating efficient procurement and distribution within the European market.
3Market Positioning
Based on its product mix, NextPharma Logistics GmbH primarily served the wholesale distribution segment of the German pharmaceutical market. By importing and distributing a range of pharmaceutical products, the company played a crucial role in ensuring the availability of essential medications to pharmacies, hospitals, and other healthcare providers across Germany. The focus on high-demand therapeutic areas further underscores its position in addressing critical healthcare needs within the country.
Seller's Guide — How to Become a Supplier to Nextpharma Logistics Gmbh
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
The concentrated sourcing pattern of NextPharma Logistics GmbH presents an opportunity for new Indian suppliers to diversify the company's supplier base. By offering competitive pricing, reliable delivery schedules, and high-quality products, Indian exporters can position themselves as viable alternatives to existing suppliers. Demonstrating compliance with international quality standards and the ability to meet the company's specific logistical requirements would be crucial in establishing a successful partnership.
2Requirements & Qualifications
Indian exporters seeking to supply NextPharma Logistics GmbH and the German market must obtain marketing authorization from the European Medicines Agency (EMA) or a national competent authority. Possessing GMP certificates recognized by the European Union, such as EU GMP, WHO GMP, or PIC/S, is essential to ensure product quality and safety. Additionally, compliance with German labeling requirements, including German language labeling and serialization mandates, is necessary to meet regulatory standards.
3How to Approach
To build a relationship with NextPharma Logistics GmbH, Indian exporters should initiate contact by providing detailed product information, including quality certifications and compliance with European standards. Participating in relevant tenders and industry events can enhance visibility and demonstrate commitment to the European market. Developing a comprehensive regulatory filing strategy, including obtaining necessary marketing authorizations and GMP certifications, is crucial. Setting realistic
Frequently Asked Questions — Nextpharma Logistics Gmbh
What products does Nextpharma Logistics Gmbh import from India?
Nextpharma Logistics Gmbh imports 3 pharmaceutical products across 2 categories. Top imports: Sitagliptin ($2.8M), Amlodipine ($550.0K), Valsartan ($550.0K).
Who supplies pharmaceuticals to Nextpharma Logistics Gmbh from India?
Nextpharma Logistics Gmbh sources from 5 verified Indian suppliers. The primary supplier is Dr.reddy's Laboratories Limited (67.1% of imports, $9.1M).
What is Nextpharma Logistics Gmbh's total pharmaceutical import value?
Nextpharma Logistics Gmbh's total pharmaceutical import value from India is $3.9M, based on 182 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Nextpharma Logistics Gmbh focus on?
Nextpharma Logistics Gmbh imports across 2 categories. The largest: Diabetes & Endocrine (71.6%), Cardiovascular (28.4%).
Get Full Nextpharma Logistics Gmbh Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Nextpharma Logistics Gmbh identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Nextpharma Logistics Gmbh's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 182 individual customs records matching Nextpharma Logistics Gmbh.
- 5.Supplier Verification: Nextpharma Logistics Gmbh sources from 5 verified Indian suppliers across 580 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.