Mylan Ireland Limited
Pharmaceutical Importer · Netherlands · Respiratory Focus · $54.2M Total Trade · DGFT Verified
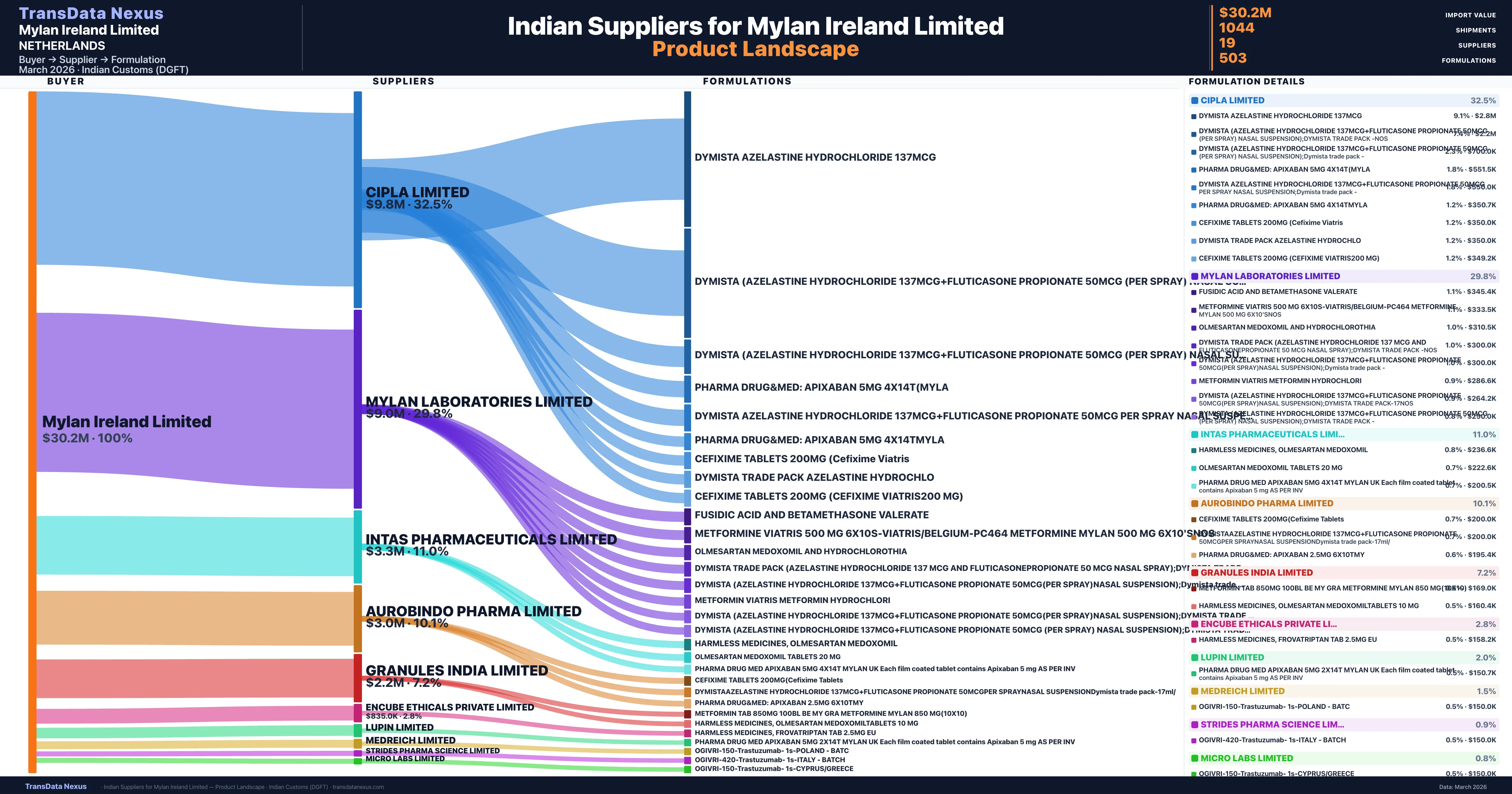
Mylan Ireland Limited is a pharmaceutical importer based in Netherlands with a total trade value of $54.2M across 10 products in 7 therapeutic categories. Based on 1,118 verified import shipments from Indian Customs (DGFT) records, Mylan Ireland Limited is the #1 buyer in 2 products including Fluticasone, Trastuzumab. Mylan Ireland Limited sources from 19 verified Indian suppliers, with Cipla Limited accounting for 32.5% of imports.
Mylan Ireland Limited — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Mylan Ireland Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $9.8M | 324 | 32.5% |
| Mylan Laboratories Limited | $9.0M | 247 | 29.8% |
| Intas Pharmaceuticals Limited | $3.3M | 143 | 11.0% |
| Aurobindo Pharma Limited | $3.0M | 100 | 10.1% |
| Granules India Limited | $2.2M | 92 | 7.2% |
| Encube Ethicals Private Limited | $835.0K | 46 | 2.8% |
| Lupin Limited | $600.0K | 12 | 2.0% |
| Medreich Limited | $446.0K | 21 | 1.5% |
| Strides Pharma Science Limited | $286.6K | 8 | 0.9% |
| Micro Labs Limited | $230.3K | 9 | 0.8% |
| Cadila Healthcare Limited | $100.0K | 2 | 0.3% |
| Ind-swift Limited | $62.2K | 4 | 0.2% |
| Torrent Pharmaceuticals Limited | $60.4K | 13 | 0.2% |
| Aizant Drug Research Solutions Private Limited | $59.7K | 16 | 0.2% |
| Chowgule Fiberglass Ships Private Limited | $50.0K | 1 | 0.2% |
| Ind Swift Limited | $40.2K | 1 | 0.1% |
| Dr.reddy's Laboratories Limited | $37.0K | 2 | 0.1% |
| Dr Reddys Laboratories Limited | $24.8K | 2 | 0.1% |
| Dr Reddy S Laboratories Limited | $16.0K | 1 | 0.1% |
Mylan Ireland Limited sources from 19 verified Indian suppliers across 503 distinct formulations. The supply base is diversified across 19 suppliers, reducing single-source dependency risk.
What Formulations Does Mylan Ireland Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Dymista azelastine hydrochloride 137mcg | $2.8M | 55 |
| Dymista (azelastine hydrochloride 137mcg+fluticasone propionate 50mcg (per spray) nasal suspension);dymista trade Pack | $2.2M | 48 |
| Dymista (azelastine hydrochloride 137mcg+fluticasone propionate 50mcg (per spray) nasal suspension);dymista trade Pack | $700.0K | 14 |
| Pharma drug&med: apixaban 5MG | $551.5K | 21 |
| Dymista azelastine hydrochloride 137mcg+fluticasone propionate 50mcg per spray nasal suspension;dymista trade Pack | $550.0K | 11 |
| Pharma drug&med: apixaban 5MG | $350.7K | 13 |
| Cefixime tablets 200MG (cefixime | $350.0K | 7 |
| Dymista trade Pack azelastine | $350.0K | 7 |
| Cefixime tablets 200MG (cefixime viatris200 MG) | $349.2K | 14 |
| Fusidic acid and betamethasone | $345.4K | 10 |
| Metformine viatris 500 MG 6x10s-viatris/belgium-pc464 metformine mylan 500 MG | $333.5K | 18 |
| Olmesartan medoxomil and | $310.5K | 9 |
| Dymista trade Pack (azelastine hydrochloride 137 MCG and fluticasonepropionate 50 MCG nasal spray);dymista trade Pack | $300.0K | 6 |
| Dymista (azelastine hydrochloride 137mcg+fluticasone propionate 50mcg(per spray)nasal suspension);dymista trade Pack | $300.0K | 6 |
| Metformin viatris metformin | $286.6K | 8 |
Mylan Ireland Limited imports 503 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Mylan Ireland Limited Import?
Mylan Ireland Limited Therapeutic Categories — 7 Specializations
Mylan Ireland Limited imports across 7 therapeutic categories, with Respiratory (44.7%), Ayurvedic & Herbal Products (19.5%), Advanced Oncology (16.4%) representing the largest segments. The portfolio is concentrated — top 5 products = 96% of total imports.
Respiratory
1 products · 44.7% · $24.3M
Ayurvedic & Herbal Products
1 products · 19.5% · $10.6M
Advanced Oncology
2 products · 16.4% · $8.9M
Cardiovascular
1 products · 11.7% · $6.3M
Advanced Antibiotics
3 products · 6.5% · $3.5M
Advanced Diabetes Medications
1 products · 0.9% · $500.0K
Gastrointestinal
1 products · 0.2% · $115.1K
Import Portfolio — Top 10 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Fluticasone | Respiratory | $24.3M | 485 | 8.6% | 1 |
| 2 | Ras | Ayurvedic & Herbal Products | $10.6M | 211 | 1.8% | 2 |
| 3 | Trastuzumab | Advanced Oncology | $8.8M | 176 | 7.0% | 1 |
| 4 | Apixaban | Cardiovascular | $6.3M | 127 | 3.5% | 3 |
| 5 | Cefixime | Advanced Antibiotics | $1.8M | 54 | 1.0% | 10 |
| 6 | Flucloxacillin | Advanced Antibiotics | $1.6M | 48 | 4.2% | 4 |
| 7 | Dapagliflozin | Advanced Diabetes Medications | $500.0K | 10 | 1.0% | 3 |
| 8 | Sulfasalazine | Gastrointestinal | $115.1K | 3 | 3.6% | 8 |
| 9 | Epirubicin | Advanced Oncology | $100.0K | 2 | 1.6% | 4 |
| 10 | Minocycline | Advanced Antibiotics | $100.0K | 2 | 0.4% | 8 |
Mylan Ireland Limited imports 10 pharmaceutical products across 7 categories into Netherlands totaling $54.2M. The company is the #1 buyer for 2 products: Fluticasone, Trastuzumab.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Mylan Ireland Limited.
Request DemoMylan Ireland Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Mylan Ireland Limited is a pharmaceutical importer and buyer based in the Netherlands, specializing in the acquisition of finished pharmaceutical formulations from India. As a subsidiary of Viatris, a global healthcare company formed in 2020 through the merger of Mylan and Upjohn, Mylan Ireland Limited plays a pivotal role in the distribution of generic and specialty pharmaceuticals within the European market.
The company's operations are integral to the Netherlands' pharmaceutical supply chain, serving as a key conduit for the importation of a diverse range of medicinal products. By sourcing high-quality generics from India, Mylan Ireland Limited contributes to the availability and affordability of essential medications across Europe.
2Distribution Network
Mylan Ireland Limited's distribution network is strategically designed to ensure efficient delivery of pharmaceutical products throughout the Netherlands and beyond. While specific warehouse locations are not publicly disclosed, the company collaborates with established logistics partners to facilitate the timely importation and distribution of medicines. This network is supported by robust logistics capabilities, including compliance with the European guidelines on Good Distribution Practice (GDP) of medicinal products for human use. (business.gov.nl)
The company's geographic coverage extends throughout the Netherlands, ensuring that imported pharmaceutical products are accessible to a wide range of healthcare providers and patients. Additionally, Mylan Ireland Limited's affiliation with Viatris enhances its ability to leverage a broader European distribution network, facilitating the efficient movement of products across the continent.
3Industry Role
In the Netherlands' pharmaceutical supply chain, Mylan Ireland Limited functions primarily as a pharmaceutical importer and buyer. The company is not involved in manufacturing or direct sales to end-users but focuses on sourcing and importing finished pharmaceutical formulations from India. This role positions Mylan Ireland Limited as a critical intermediary, ensuring the availability of a wide array of generic and specialty medicines within the Dutch market.
By importing these products, Mylan Ireland Limited supports the Dutch healthcare system by enhancing the accessibility and affordability of essential medications. The company's operations are aligned with the European Union's objectives of promoting competition and reducing healthcare costs through the availability of generic medicines.
Supplier Relationship Intelligence — Mylan Ireland Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Mylan Ireland Limited's sourcing strategy exhibits a high degree of concentration, with the top five products accounting for 95.5% of the total import value from India. This concentration is particularly notable in the respiratory and advanced oncology therapeutic categories, where the company imports significant volumes of fluticasone and trastuzumab. Such a focused sourcing approach may indicate a strategic decision to concentrate on high-demand, high-value products, potentially leveraging economies of scale and supplier relationships.
However, this concentration also introduces risks associated with dependency on a limited number of products and suppliers. Any disruptions in the supply chain for these key products could have a disproportionate impact on Mylan Ireland Limited's operations. The company's reliance on a diverse set of Indian suppliers, including CIPLA Limited and Mylan Laboratories Limited, suggests an effort to mitigate such risks by not depending on a single source. Nonetheless, the overall high concentration underscores the importance of robust supply chain management and contingency planning.
2Supply Chain Resilience
Mylan Ireland Limited's supply chain resilience is supported by its diversified sourcing strategy, engaging multiple Indian suppliers to mitigate risks associated with single-source dependency. The company's importation of 503 unique pharmaceutical formulations indicates a broad product portfolio, which can help buffer against market fluctuations and supply disruptions.
However, the high concentration in the top five products suggests that while the company has a diverse supplier base, it remains heavily reliant on a narrow range of products. This focus could expose Mylan Ireland Limited to vulnerabilities if there are disruptions in the supply of these key products. To enhance supply chain resilience, the company may consider expanding its product range and further diversifying its supplier base to reduce dependency on specific products and suppliers.
3Strategic Implications
Mylan Ireland Limited's concentrated sourcing pattern positions it as a dominant player in specific therapeutic areas within the Netherlands' pharmaceutical market, particularly in respiratory and oncology segments. This focus allows the company to leverage supplier relationships and achieve economies of scale, potentially enhancing its competitive edge in these high-demand areas.
For Indian exporters, this concentration presents both opportunities and challenges. Suppliers of fluticasone and trastuzumab may find a stable and substantial market in Mylan Ireland Limited, fostering long-term partnerships. Conversely, the limited diversification in Mylan Ireland Limited's product portfolio may necessitate strategic efforts to introduce new products and expand market share within the Netherlands.
Importing Pharmaceuticals into Netherlands — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Netherlands
1Regulatory Authority & Framework
In the Netherlands, the Medicines Evaluation Board (MEB) serves as the primary regulatory authority overseeing the authorization and supervision of medicinal products. The MEB ensures that all pharmaceutical products meet the necessary safety, efficacy, and quality standards before they are marketed. (english.cbg-meb.nl)
Key legislation governing pharmaceutical imports includes the Medicines Act (Geneesmiddelenwet), which outlines the requirements for the importation, distribution, and sale of medicinal products. Additionally, the Commodities Act (Warenwet) provides general rules on public health, product safety, fairness of trade, and proper information, applicable to all consumer products, including medicines. (business.gov.nl)
The marketing authorization pathway for Indian generics involves obtaining a parallel import marketing authorization from the MEB. This process requires demonstrating that the imported product is therapeutically equivalent to an already authorized product in the Netherlands and complies with all regulatory standards. (english.cbg-meb.nl)
2Import Licensing & GMP
Importers of medicinal products into the Netherlands must obtain a manufacturer's permit if they prepare or import medicines from outside the European Economic Area (EEA). This permit is issued by Farmatec, an executive body of the Ministry of Health, Welfare and Sport (VWS). (business.gov.nl)
Good Manufacturing Practice (GMP) certification is essential for ensuring the quality and safety of pharmaceutical products. The Netherlands recognizes GMP certificates from the European Union, the World Health Organization (WHO), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their suppliers hold valid GMP certifications from these recognized bodies to comply with Dutch regulations. (business.gov.nl)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. This testing ensures that the products meet the standards set by the MEB and are safe for consumer use. (business.gov.nl)
Stability requirements are also critical, as they determine the shelf life and storage conditions of pharmaceutical products. Manufacturers must provide stability data to demonstrate that their products remain effective and safe throughout their intended shelf life.
Labeling requirements include providing information in Dutch, ensuring that consumers and healthcare professionals can understand the product's usage, dosage, and potential side effects. Serialization mandates may also apply, requiring unique identifiers on packaging to enhance traceability and prevent counterfeit products.
4Recent Regulatory Changes
Effective from 1 July 2024, significant changes have been implemented regarding the declaration of pharmaceutical products and active substances for import into the Netherlands. Importers are now required to include a fictitious document code in their declarations, corresponding to their marketing authorization or API registration status. This change aims to streamline import procedures and enhance regulatory oversight.
Mylan Ireland Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Mylan Ireland Limited's focus on importing products in the respiratory, Ayurvedic & herbal, and advanced oncology therapeutic categories aligns with market demand for these treatments. The significant importation of fluticasone, a corticosteroid used in respiratory conditions, and trastuzumab, a monoclonal antibody for advanced oncology, indicates a strategic emphasis on high-demand, high-value products. This strategy caters to prevalent health concerns and leverages the company's expertise in these therapeutic areas.
The company's importation of Ayurvedic and herbal products reflects a growing consumer interest in alternative and complementary medicine. By incorporating these products, Mylan Ireland Limited diversifies its portfolio and meets the evolving preferences of the Dutch market.
2Sourcing Profile
Mylan Ireland Limited's sourcing strategy is centered on importing generic drugs from India, focusing on finished pharmaceutical formulations. This approach allows the company to offer a broad range of products without the complexities associated with raw material procurement. India's robust pharmaceutical manufacturing sector, known for its adherence to international quality standards, makes it a reliable source for these formulations.
The company's preference for finished formulations aligns with its role as an importer and distributor, facilitating efficient market entry and distribution within the Netherlands. By sourcing from India, Mylan Ireland Limited leverages cost advantages and a diverse product range, enhancing its competitiveness in the European market.
3Market Positioning
Based on its product mix, Mylan Ireland Limited serves multiple segments of the Netherlands market, including retail pharmacies
Frequently Asked Questions — Mylan Ireland Limited
What products does Mylan Ireland Limited import from India?
Mylan Ireland Limited imports 10 pharmaceutical products across 7 categories. Top imports: Fluticasone ($24.3M), Ras ($10.6M), Trastuzumab ($8.8M), Apixaban ($6.3M), Cefixime ($1.8M).
Who supplies pharmaceuticals to Mylan Ireland Limited from India?
Mylan Ireland Limited sources from 19 verified Indian suppliers. The primary supplier is Cipla Limited (32.5% of imports, $9.8M).
What is Mylan Ireland Limited's total pharmaceutical import value?
Mylan Ireland Limited's total pharmaceutical import value from India is $54.2M, based on 1,118 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Mylan Ireland Limited focus on?
Mylan Ireland Limited imports across 7 categories. The largest: Respiratory (44.7%), Ayurvedic & Herbal Products (19.5%), Advanced Oncology (16.4%).
Get Full Mylan Ireland Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Mylan Ireland Limited identified across shipments using consignee name normalization, aggregating 3 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Mylan Ireland Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 1,118 individual customs records matching Mylan Ireland Limited.
- 5.Supplier Verification: Mylan Ireland Limited sources from 19 verified Indian suppliers across 503 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
10 Products Tracked
7 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 3 company name variants from customs records. For current shipment-level data, contact TransData Nexus.