Movianto UK Limited
Pharmaceutical Importer · United Kingdom · Antihistamines & Allergy Focus · $20.7M Total Trade · DGFT Verified
Movianto UK Limited is a pharmaceutical importer based in United Kingdom with a total trade value of $20.7M across 5 products in 5 therapeutic categories. Based on 476 verified import shipments from Indian Customs (DGFT) records, Movianto UK Limited is the #1 buyer in 2 products including Fexofenadine, Bisacodyl. Movianto UK Limited sources from 3 verified Indian suppliers, with Dr.reddy's Laboratories Limited accounting for 43.3% of imports.
Movianto UK Limited — Import Portfolio & Supplier Network
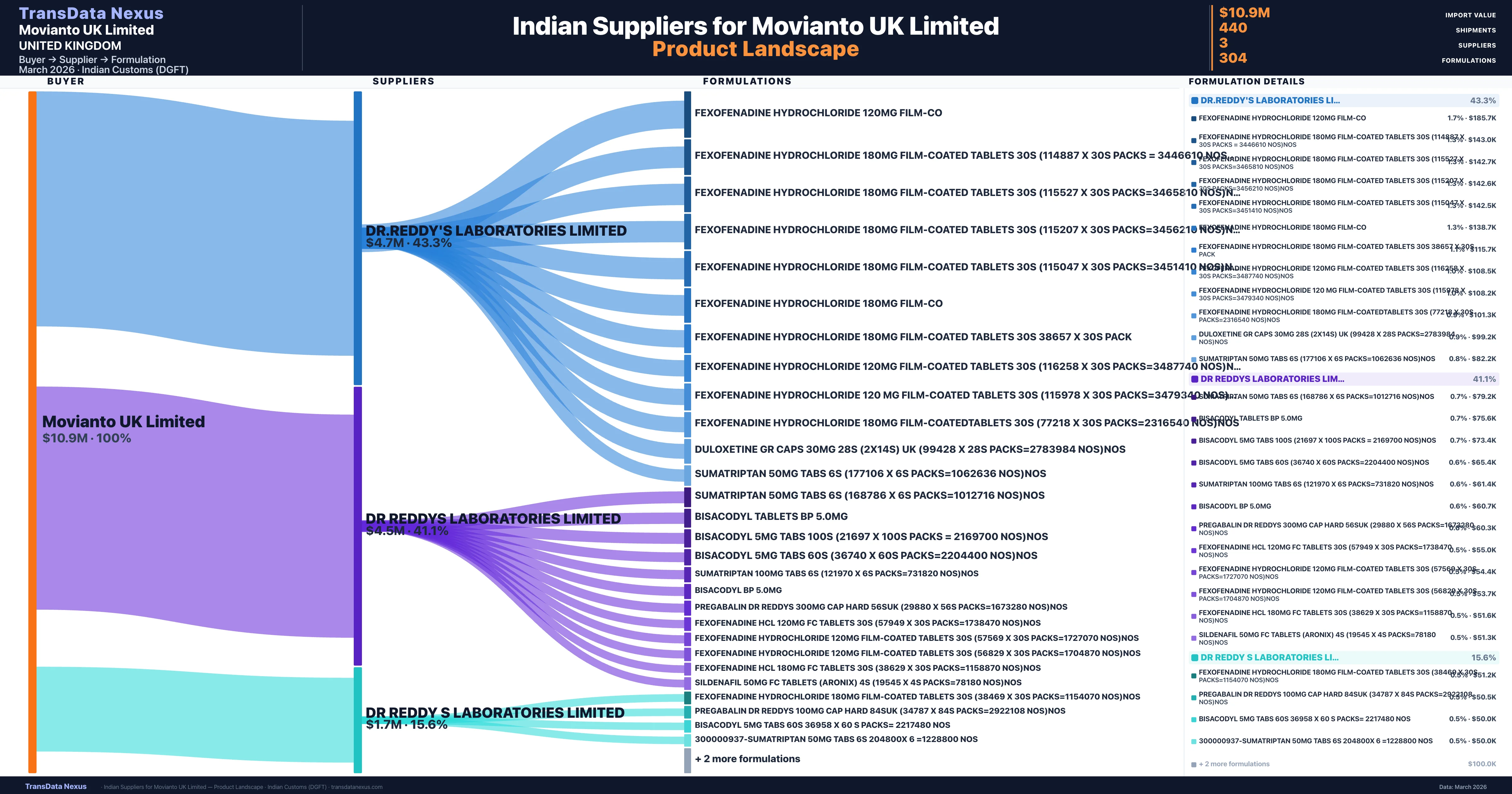
Who Are the Verified Indian Suppliers to Movianto UK Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Dr.reddy's Laboratories Limited | $4.7M | 241 | 43.3% |
| Dr Reddys Laboratories Limited | $4.5M | 144 | 41.1% |
| Dr Reddy S Laboratories Limited | $1.7M | 55 | 15.6% |
Movianto UK Limited sources from 3 verified Indian suppliers across 304 distinct formulations. Total import value: $10.9M across 440 shipments.
What Formulations Does Movianto UK Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Fexofenadine hydrochloride 120MG | $185.7K | 5 |
| Fexofenadine hydrochloride 180MG Film-Coated tablets 30s (114887 x 30s packs = 3446610 nos | $143.0K | 3 |
| Fexofenadine hydrochloride 180MG Film-Coated tablets 30s (115527 x 30s packs=3465810 nos | $142.7K | 3 |
| Fexofenadine hydrochloride 180MG Film-Coated tablets 30s (115207 x 30s packs=3456210 nos | $142.6K | 3 |
| Fexofenadine hydrochloride 180MG Film-Coated tablets 30s (115047 x 30s packs=3451410 nos | $142.5K | 3 |
| Fexofenadine hydrochloride 180MG | $138.7K | 4 |
| Fexofenadine hydrochloride 180MG Film-Coated tablets 30s 38657 x 30s | $115.7K | 3 |
| Fexofenadine hydrochloride 120MG Film-Coated tablets 30s (116258 x 30s packs=3487740 nos | $108.5K | 3 |
| Fexofenadine hydrochloride 120 MG Film-Coated tablets 30s (115978 x 30s packs=3479340 nos | $108.2K | 3 |
| Fexofenadine hydrochloride 180MG film-coatedtablets 30s (77218 x 30s packs=2316540 nos | $101.3K | 3 |
| Duloxetine gr CAPS 30MG 28s (2x14s) uk (99428 x 28s packs=2783984 nos | $99.2K | 3 |
| Sumatriptan 50MG tabs 6s (177106 x 6s packs=1062636 nos | $82.2K | 3 |
| Sumatriptan 50MG tabs 6s (168786 x 6s packs=1012716 nos | $79.2K | 3 |
| Bisacodyl tablets BP 5.0MG | $75.6K | 2 |
| Bisacodyl 5MG tabs 100s (21697 x 100s packs = 2169700 nos | $73.4K | 3 |
Movianto UK Limited imports 304 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Movianto UK Limited Import?
Top Products by Import Value
Movianto UK Limited Therapeutic Categories — 5 Specializations
Movianto UK Limited imports across 5 therapeutic categories, with Antihistamines & Allergy (74.3%), Gastrointestinal (14.6%), Diabetes & Endocrine (8.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antihistamines & Allergy
1 products · 74.3% · $15.3M
Gastrointestinal
1 products · 14.6% · $3.0M
Diabetes & Endocrine
1 products · 8.0% · $1.6M
Cardiovascular
1 products · 1.9% · $400.0K
Advanced Antifungals
1 products · 1.2% · $250.0K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Fexofenadine | Antihistamines & Allergy | $15.3M | 307 | 8.4% | 1 |
| 2 | Bisacodyl | Gastrointestinal | $3.0M | 119 | 21.0% | 1 |
| 3 | Sitagliptin | Diabetes & Endocrine | $1.6M | 37 | 0.5% | 13 |
| 4 | Rivaroxaban | Cardiovascular | $400.0K | 8 | 0.8% | 17 |
| 5 | Voriconazole | Advanced Antifungals | $250.0K | 5 | 0.8% | 8 |
Movianto UK Limited imports 5 pharmaceutical products across 5 categories into United Kingdom totaling $20.7M. The company is the #1 buyer for 2 products: Fexofenadine, Bisacodyl.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Movianto UK Limited.
Request DemoMovianto UK Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Movianto UK Limited, established on 11 February 1976, is a prominent pharmaceutical importer and distributor headquartered in Bedford, United Kingdom. The company operates as a private limited entity, with its registered office located at Unit 1 Bedford Link Logistics Park, Bell Farm Way, Kempston, Bedford, MK43 9SS. (find-and-update.company-information.service.gov.uk) As of 2024, Movianto UK reported a turnover exceeding £54 million and employed over 1,300 individuals, reflecting its significant presence in the healthcare logistics sector.
Movianto UK Limited is a subsidiary of Movianto Holding UK Limited, which serves as the holding company for Movianto's UK operations. The holding company was incorporated on 14 August 2012 and shares the same registered address in Bedford. (find-and-update.company-information.service.gov.uk) Movianto UK Limited plays a crucial role in the United Kingdom's pharmaceutical distribution network, offering comprehensive supply chain solutions tailored to the needs of pharmaceutical, veterinary, medical device, life sciences, and healthcare sectors. Their services encompass primary transport, storage, order fulfillment, manufacturing, financial services, customs management, and last-mile transport, ensuring efficient and compliant delivery of healthcare products across the UK.
2Distribution Network
Movianto UK Limited operates a robust distribution network with three certified warehouses strategically located in Bedford, Lutterworth, and Haydock. These facilities are equipped to handle a wide range of healthcare products, including pharmaceuticals, medical devices, and life sciences products, ensuring compliance with Good Distribution Practice (GDP) standards. The Bedford facility, situated at Unit 1 Bedford Link Logistics Park, Bell Farm Way, Kempston, Bedford, MK43 9SS, serves as the primary hub, offering advanced automation and a GMP-certified rework facility for secondary packaging.
The company's logistics capabilities include a fleet of GDP-compliant vehicles, providing temperature-controlled transport solutions to maintain product integrity throughout the supply chain. This infrastructure enables Movianto UK Limited to offer end-to-end logistics services, from primary transport to last-mile delivery, ensuring timely and secure distribution of healthcare products across the United Kingdom.
3Industry Role
Movianto UK Limited functions as a primary wholesaler and distributor within the United Kingdom's pharmaceutical supply chain. By importing finished pharmaceutical formulations from various global suppliers, including India, and distributing them to healthcare providers, pharmacies, and other stakeholders, Movianto UK ensures the availability of essential medical products across the UK. Their comprehensive services, encompassing storage, order fulfillment, and last-mile delivery, position them as a key intermediary between manufacturers and end-users in the healthcare sector.
Supplier Relationship Intelligence — Movianto UK Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Movianto UK Limited's sourcing strategy exhibits a high degree of concentration, particularly in its imports from India. Between 2022 and 2026, the company imported finished pharmaceutical formulations totaling $20.7 million USD across 476 shipments, with a portfolio concentration of 100% in the top five products. The primary products imported include Fexofenadine ($15.3 million, 8.4% share), Bisacodyl ($3.0 million, 21.0% share), Sitagliptin ($1.6 million, 0.5% share), Rivaroxaban ($400,000, 0.8% share), and Voriconazole ($250,000, 0.8% share).
This sourcing pattern indicates a strategic focus on specific therapeutic areas, notably Antihistamines & Allergy (74.3%), Gastrointestinal (14.6%), and Diabetes & Endocrine (8.0%). The company's dominance as the number one buyer in Fexofenadine and Bisacodyl suggests strong, stable relationships with suppliers in these categories. However, the reliance on a limited number of suppliers for these key products may pose risks related to supply chain disruptions or pricing volatility.
2Supply Chain Resilience
Movianto UK Limited's supply chain resilience is influenced by its concentrated sourcing from a few Indian suppliers. The company's imports from India are primarily sourced from three suppliers: DR.REDDY'S LABORATORIES LIMITED ($4.7 million, 43.3% share), DR REDDYS LABORATORIES LIMITED ($4.5 million, 41.1% share), and DR REDDY S LABORATORIES LIMITED ($1.7 million, 15.6% share).
This concentration may expose Movianto UK Limited to risks associated with supplier dependency, such as potential disruptions in supply or changes in supplier pricing strategies. To mitigate these risks, the company may consider diversifying its supplier base and establishing relationships with additional manufacturers to enhance supply chain resilience.
3Strategic Implications
The concentrated sourcing strategy of Movianto UK Limited, particularly its significant imports from India, positions the company as a major player in specific therapeutic categories within the UK market. This focus allows Movianto UK to leverage economies of scale and negotiate favorable terms with suppliers. For Indian exporters, this presents an opportunity to strengthen partnerships with Movianto UK by ensuring consistent product quality, reliable delivery schedules, and compliance with regulatory standards, thereby enhancing their position in the UK market.
Importing Pharmaceuticals into United Kingdom — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Kingdom
1Regulatory Authority & Framework
In the United Kingdom, the Medicines and Healthcare products Regulatory Agency (MHRA) serves as the primary regulatory authority overseeing the importation and distribution of pharmaceutical products. The MHRA ensures that all medicinal products meet stringent safety, quality, and efficacy standards before they are authorized for sale in the UK. (cms.mhra.gov.uk)
Key legislation governing pharmaceutical imports includes the Human Medicines Regulations 2012 (SI 2012/1916), which consolidates and updates previous regulations related to the manufacture, distribution, and sale of medicinal products. This framework outlines the requirements for marketing authorization, Good Manufacturing Practice (GMP), and Good Distribution Practice (GDP) compliance. (cms.mhra.gov.uk)
For Indian generics to enter the UK market, they must undergo a marketing authorization process, which includes demonstrating bioequivalence to the reference product and compliance with GMP standards. The MHRA evaluates these submissions to ensure that imported generics meet the necessary criteria for safety and efficacy.
2Import Licensing & GMP
Import licensing requirements in the UK mandate that all pharmaceutical products, including generics, must be authorized by the MHRA before they can be imported and distributed. This process involves submitting detailed documentation, including evidence of GMP compliance, to ensure that imported products meet the required quality standards. (cms.mhra.gov.uk)
GMP certificates recognized by the MHRA include those issued by the European Union (EU GMP), the World Health Organization (WHO GMP), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). These certifications confirm that manufacturing facilities adhere to internationally accepted standards for pharmaceutical production. Additionally, wholesale distribution authorization is required for entities involved in the distribution of medicinal products, ensuring that all parties in the supply chain maintain appropriate standards of practice.
3Quality & Labeling
Pharmaceutical products imported into the UK must undergo batch testing to verify their quality, safety, and efficacy. This testing ensures that each batch meets the specifications outlined in the marketing authorization. Stability requirements are also enforced to confirm that products maintain their quality throughout their shelf life under specified storage conditions.
Labeling requirements stipulate that product information must be provided in English, including details such as the product name, dosage form, strength, route of administration, and storage conditions. Serialization mandates are in place to facilitate the traceability of pharmaceutical products throughout the supply chain, enhancing security and reducing the risk of counterfeit products entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, the UK government implemented several policy changes affecting pharmaceutical imports, including those from India. These changes focused on enhancing supply chain security, improving traceability through serialization, and updating GMP compliance requirements to align with international standards. Additionally, there was an increased emphasis on ensuring the quality and safety of imported generics, leading to more stringent testing and documentation requirements.
Movianto UK Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Movianto UK Limited's product category focus on Antihistamines & Allergy (74.3%), Gastrointestinal (14.6%), and Diabetes & Endocrine (8.0%) reflects a strategic alignment with prevalent health conditions in the UK. The significant import of Fexofenadine and Bisacodyl indicates a response to high demand in the antihistamine and gastrointestinal sectors, respectively. This focus allows Movianto UK to cater to a substantial portion of the UK's pharmaceutical needs, ensuring the availability of essential medications for a large patient population.
2Sourcing Profile
Movianto UK Limited's sourcing strategy emphasizes importing finished pharmaceutical formulations from India, particularly in the therapeutic areas of Antih
Frequently Asked Questions — Movianto UK Limited
What products does Movianto UK Limited import from India?
Movianto UK Limited imports 5 pharmaceutical products across 5 categories. Top imports: Fexofenadine ($15.3M), Bisacodyl ($3.0M), Sitagliptin ($1.6M), Rivaroxaban ($400.0K), Voriconazole ($250.0K).
Who supplies pharmaceuticals to Movianto UK Limited from India?
Movianto UK Limited sources from 3 verified Indian suppliers. The primary supplier is Dr.reddy's Laboratories Limited (43.3% of imports, $4.7M).
What is Movianto UK Limited's total pharmaceutical import value?
Movianto UK Limited's total pharmaceutical import value from India is $20.7M, based on 476 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Movianto UK Limited focus on?
Movianto UK Limited imports across 5 categories. The largest: Antihistamines & Allergy (74.3%), Gastrointestinal (14.6%), Diabetes & Endocrine (8.0%).
Get Full Movianto UK Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Movianto UK Limited identified across shipments using consignee name normalization, aggregating 4 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Movianto UK Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 476 individual customs records matching Movianto UK Limited.
- 5.Supplier Verification: Movianto UK Limited sources from 3 verified Indian suppliers across 304 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
5 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 4 company name variants from customs records. For current shipment-level data, contact TransData Nexus.