The Tanzania Medicines and Medical Devices Authority (TMDA) is the primary regulatory body overseeing the importation, exportation, and distribution of pharmaceutical products in Tanzania. Established under the Tanzania Medicines and Medical Devices Act, Cap. 219, TMDA is responsible for ensuring that all medicines and medical devices meet the required quality, safety, and efficacy standards before they enter the Tanzanian market. This includes the registration of pharmaceutical products, licensing of manufacturing and importing facilities, and monitoring of market compliance.
Key legislation governing pharmaceutical imports includes the Tanzania Medicines and Medical Devices Act, Cap. 219, and the Tanzania Food, Drugs and Cosmetics (Registration of Premises, Importation and Exportation of Pharmaceutical Products and Raw Materials) Regulations, 2015. These regulations outline the procedures and requirements for importing pharmaceutical products, including the necessity for importers to register their premises with TMDA and obtain import permits for each consignment. The guidelines also specify conditions related to product shelf-life, labeling, and compliance with Good Manufacturing Practice (GMP) standards.
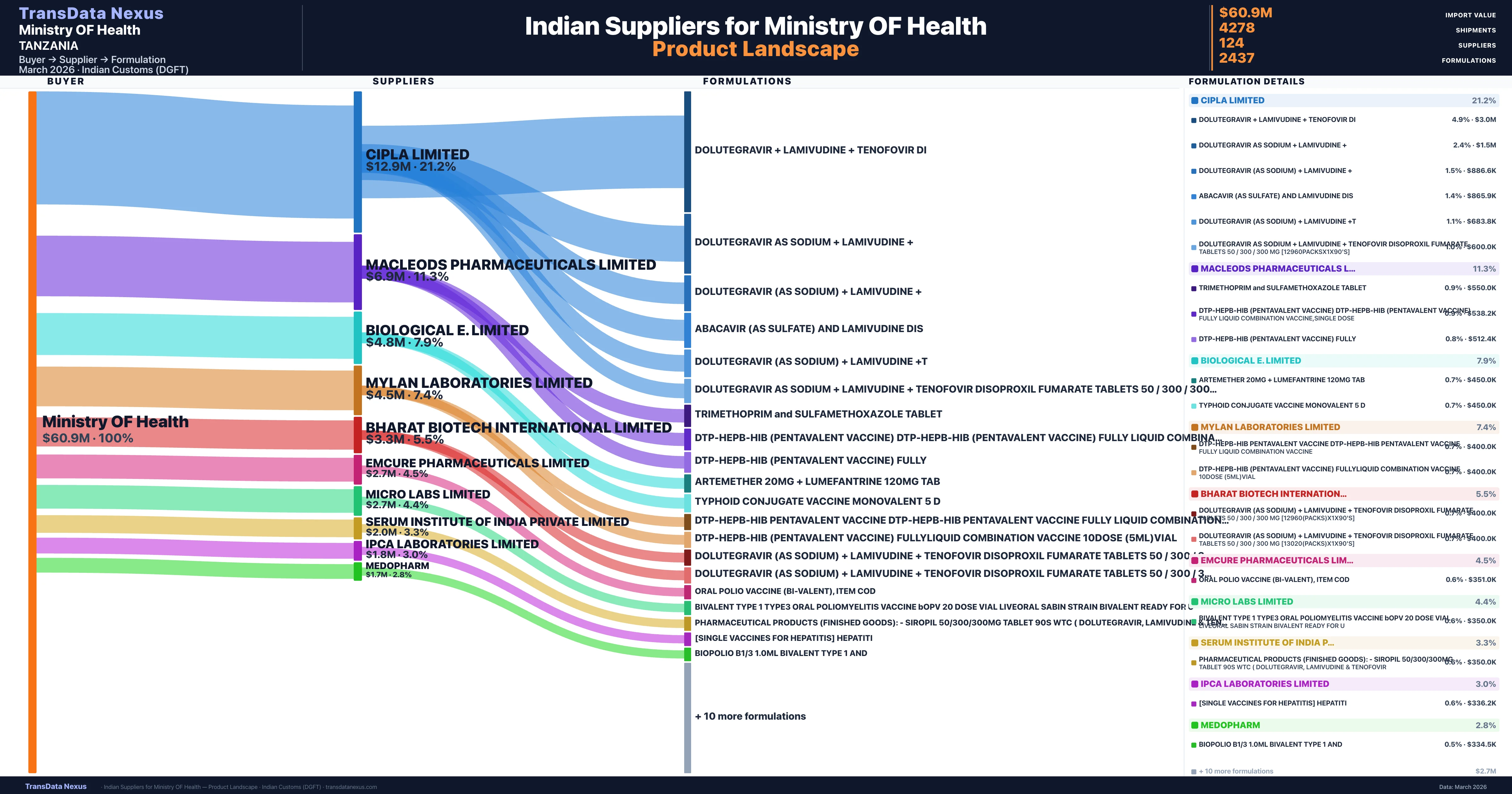
For Indian pharmaceutical exporters, understanding and adhering to these regulatory frameworks is crucial. Ensuring that products are registered with TMDA, comply with Tanzanian labeling requirements, and meet GMP standards recognized by TMDA will facilitate smoother market entry and acceptance. Additionally, staying informed about any updates or changes in Tanzanian pharmaceutical regulations is essential for maintaining compliance and sustaining business relationships with Tanzanian importers like the Ministry of Health.