Ministry OF Health
Pharmaceutical Importer · Tanzania · Antiviral & HIV Medications Focus · $14.0M Total Trade · DGFT Verified
Ministry OF Health is a pharmaceutical importer based in Tanzania with a total trade value of $14.0M across 8 products in 5 therapeutic categories. Based on 323 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Ministry OF Health sources from 124 verified Indian suppliers, with Cipla Limited accounting for 21.2% of imports.
Ministry OF Health — Import Portfolio & Supplier Network
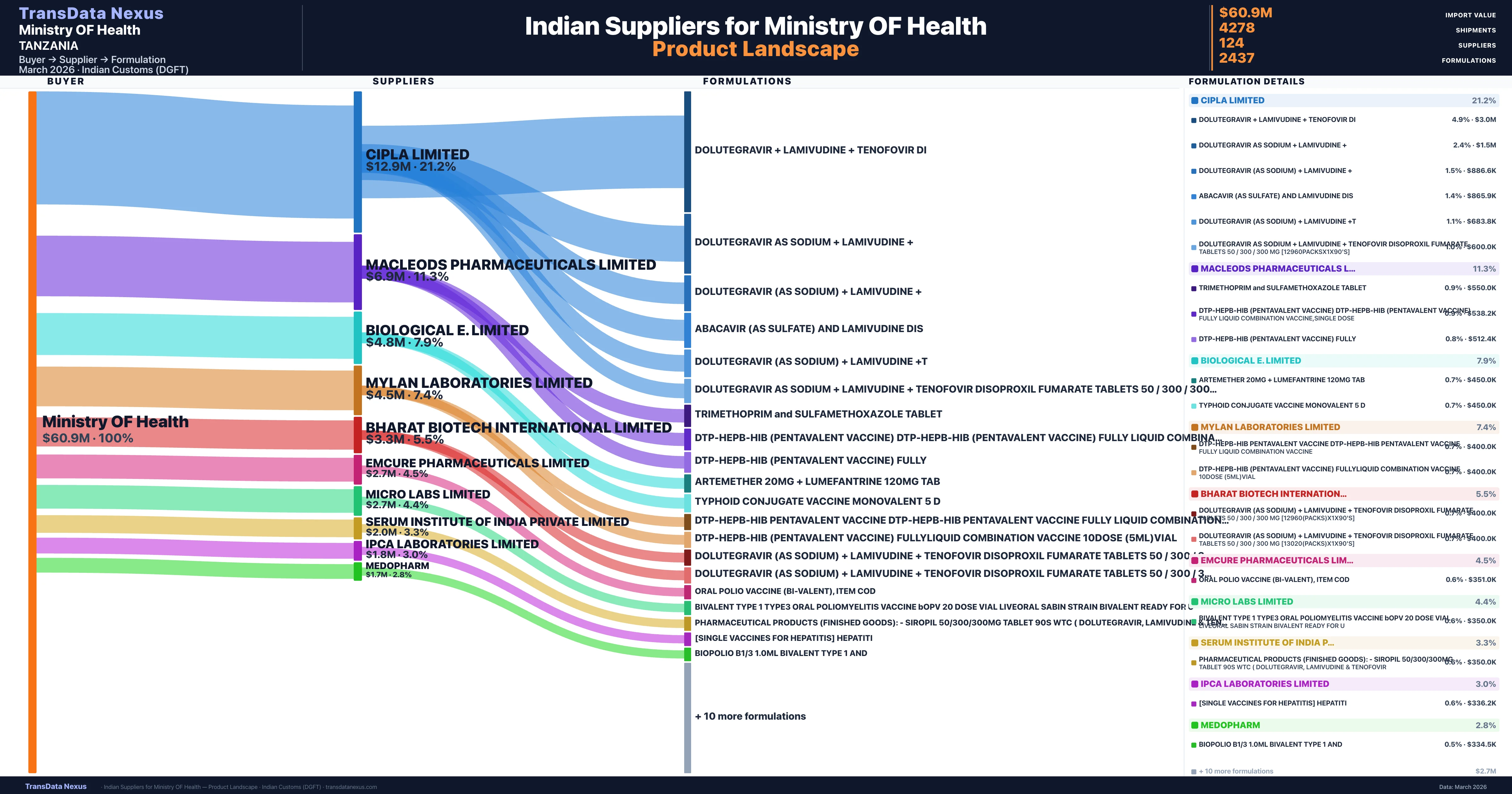
Who Are the Verified Indian Suppliers to Ministry OF Health?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $12.9M | 618 | 21.2% |
| Macleods Pharmaceuticals Limited | $6.9M | 328 | 11.3% |
| Biological E. Limited | $4.8M | 126 | 7.9% |
| Mylan Laboratories Limited | $4.5M | 191 | 7.4% |
| Bharat Biotech International Limited | $3.3M | 85 | 5.5% |
| Emcure Pharmaceuticals Limited | $2.7M | 67 | 4.5% |
| Micro Labs Limited | $2.7M | 79 | 4.4% |
| Serum Institute Of India Private Limited | $2.0M | 108 | 3.3% |
| Ipca Laboratories Limited | $1.8M | 52 | 3.0% |
| Medopharm | $1.7M | 48 | 2.8% |
| Biological E Limited | $1.5M | 48 | 2.5% |
| Aurobindo Pharma Limited | $1.4M | 72 | 2.3% |
| Lupin Limited | $1.4M | 32 | 2.3% |
| Strides Pharma Science Limited | $1.1M | 31 | 1.7% |
| Panacea Biotec Limited | $1.0M | 25 | 1.7% |
| Hetero Labs Limited | $793.5K | 36 | 1.3% |
| Oxford Laboratories Private Limited | $777.8K | 34 | 1.3% |
| Ajanta Pharma Limited | $587.4K | 22 | 1.0% |
| Intermed Laboratories Private Limited | $542.1K | 38 | 0.9% |
| Haffkine Bio Pharmaceutical Corporation Limited | $501.0K | 11 | 0.8% |
| Medicamen Biotech Limited | $494.9K | 25 | 0.8% |
| Sun Pharmaceutical Industries Limited | $485.2K | 24 | 0.8% |
| Hll Lifecare Limited | $476.4K | 23 | 0.8% |
| Maneesh Pharmaceuticals Limited | $400.0K | 8 | 0.7% |
| Vaman Healthcare And Exports Private Limited | $337.5K | 7 | 0.6% |
| S M B Corporation Of India | $283.4K | 17 | 0.5% |
| Venus Remedies Limited | $254.0K | 8 | 0.4% |
| Medwise Overseas Private Limited | $242.5K | 59 | 0.4% |
| Rainbow Life Sciences Private Limited | $234.3K | 7 | 0.4% |
| Andel Healthcare | $221.1K | 9 | 0.4% |
Ministry OF Health sources from 124 verified Indian suppliers across 2,437 distinct formulations. The supply base is diversified across 124 suppliers, reducing single-source dependency risk.
What Formulations Does Ministry OF Health Import?
| Formulation | Value | Ships |
|---|---|---|
| Dolutegravir + lamivudine + tenofovir | $3.0M | 66 |
| Dolutegravir as sodium + lamivudine | $1.5M | 31 |
| Dolutegravir (as sodium) + lamivudine | $886.6K | 19 |
| Abacavir (as sulfate) and lamivudine | $865.9K | 18 |
| Dolutegravir (as sodium) + lamivudine | $683.8K | 14 |
| Dolutegravir as sodium + lamivudine + tenofovir disoproxil fumarate tablets 50 / 300 / 300 MG | $600.0K | 12 |
| Trimethoprim and sulfamethoxazole | $550.0K | 11 |
| Dtp-hepb-hib (pentavalent vaccine) dtp-hepb-hib (pentavalent vaccine) fully liquid combination vaccine,single | $538.2K | 15 |
| Dtp-hepb-hib (pentavalent vaccine) | $512.4K | 11 |
| Artemether 20MG + lumefantrine 120MG TAB | $450.0K | 9 |
| Typhoid conjugate vaccine monovalent 5 | $450.0K | 9 |
| Dtp-hepb-hib pentavalent vaccine dtp-hepb-hib pentavalent vaccine fully liquid combination | $400.0K | 8 |
| Dtp-hepb-hib (pentavalent vaccine) fullyliquid combination vaccine 10dose (5ML | $400.0K | 8 |
| Dolutegravir (as sodium) + lamivudine + tenofovir disoproxil fumarate tablets 50 / 300 / 300 MG [12960(packs | $400.0K | 8 |
| Dolutegravir (as sodium) + lamivudine + tenofovir disoproxil fumarate tablets 50 / 300 / 300 MG [13020(packs | $400.0K | 8 |
Ministry OF Health imports 2,437 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Ministry OF Health Import?
Ministry OF Health Therapeutic Categories — 5 Specializations
Ministry OF Health imports across 5 therapeutic categories, with Antiviral & HIV Medications (48.2%), Other (32.5%), Nutritional Supplements (12.9%) representing the largest segments. The portfolio is concentrated — top 5 products = 98% of total imports.
Antiviral & HIV Medications
4 products · 48.2% · $6.8M
Other
1 products · 32.5% · $4.5M
Nutritional Supplements
1 products · 12.9% · $1.8M
Antimalarial & Antiparasitic
1 products · 6.1% · $850.0K
Medical Devices & Diagnostics
1 products · 0.4% · $50.7K
Import Portfolio — Top 8 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Vaccine | Other | $4.5M | 91 | 0.4% | 8 |
| 2 | Tenofovir | Antiviral & HIV Medications | $3.5M | 71 | 0.1% | 2 |
| 3 | Dolutegravir | Antiviral & HIV Medications | $3.0M | 59 | 0.1% | 2 |
| 4 | Sodium | Nutritional Supplements | $1.8M | 36 | 0.5% | 4 |
| 5 | Artemether | Antimalarial & Antiparasitic | $850.0K | 17 | 0.1% | 9 |
| 6 | Ritonavir | Antiviral & HIV Medications | $150.0K | 3 | 0.0% | 12 |
| 7 | Atazanavir | Antiviral & HIV Medications | $100.0K | 2 | 0.1% | 5 |
| 8 | Instrument | Medical Devices & Diagnostics | $50.7K | 44 | 3.7% | 5 |
Ministry OF Health imports 8 pharmaceutical products across 5 categories into Tanzania totaling $14.0M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Ministry OF Health.
Request DemoMinistry OF Health — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
The Ministry of Health (MoH) in Tanzania is the principal government agency responsible for overseeing the country's healthcare system, including the procurement and distribution of pharmaceutical products. As a government procurement entity, the MoH plays a pivotal role in ensuring the availability of essential medicines and medical supplies across Tanzania. The Ministry operates under the jurisdiction of the Tanzanian government, with its headquarters located in Dodoma, the capital city. It functions as a central authority coordinating with various stakeholders to manage the healthcare needs of the population.
In the pharmaceutical supply chain, the MoH's role is multifaceted. It collaborates with the Medical Stores Department (MSD), an autonomous agency under the Ministry, which is responsible for the procurement, storage, and distribution of medical supplies to public health facilities nationwide. The MoH also engages with international and local suppliers to source a wide range of pharmaceutical products, ensuring that public health facilities are adequately stocked with necessary medications. Through these efforts, the Ministry aims to enhance the quality and accessibility of healthcare services across the country.
2Distribution Network
The Ministry of Health's distribution network is extensive, encompassing various warehouses and logistics centers strategically located throughout Tanzania. These facilities are designed to store and manage a diverse array of pharmaceutical products, ensuring timely and efficient delivery to healthcare providers nationwide. The logistics capabilities include a combination of road, rail, and air transport, facilitating the movement of medical supplies from central warehouses to regional and district health facilities. This comprehensive distribution system is crucial for maintaining a steady supply of essential medicines and medical devices, particularly in remote and underserved areas.
Geographically, the Ministry's distribution network covers the entire Tanzanian territory, from urban centers to rural regions. This widespread coverage is essential for meeting the healthcare needs of the country's diverse population. Additionally, the Ministry collaborates with international partners and suppliers to import pharmaceutical products, further enhancing its capacity to provide quality healthcare services. The integration of these logistics and distribution strategies underscores the Ministry's commitment to improving health outcomes across Tanzania.
3Industry Role
In Tanzania's pharmaceutical supply chain, the Ministry of Health serves as a central procurement and regulatory authority. While it does not function as a wholesaler or distributor in the traditional sense, it plays a critical role in coordinating the procurement of pharmaceutical products for public health facilities. The Ministry's collaboration with the Medical Stores Department (MSD) ensures that medicines and medical supplies are efficiently distributed to hospitals, clinics, and other healthcare providers. This centralized approach aims to streamline the supply chain, reduce costs, and improve the availability of essential medicines across the country.
The Ministry's role also extends to policy development and regulatory oversight. It works closely with the Tanzania Medicines and Medical Devices Authority (TMDA) to establish guidelines and regulations governing the importation, distribution, and use of pharmaceutical products. This collaboration ensures that all imported medicines meet the required quality standards and are safe for public consumption. Through these efforts, the Ministry contributes to the overall stability and effectiveness of Tanzania's pharmaceutical supply chain.
Supplier Relationship Intelligence — Ministry OF Health
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The Ministry of Health's sourcing strategy reveals a significant concentration in its supplier base, particularly from Indian pharmaceutical manufacturers. Over the period from 2022 to 2026, the Ministry imported a total of $14.0 million worth of finished pharmaceutical formulations from India, encompassing 323 shipments across eight products in five therapeutic categories. This indicates a strong reliance on Indian suppliers for a substantial portion of its pharmaceutical needs.
The top five imported products—Vaccine, Tenofovir, Dolutegravir, Sodium, and Artemether—account for 97.9% of the total import value. This high concentration suggests that the Ministry's procurement strategy is heavily dependent on a narrow range of products and suppliers. Such dependency can pose risks, including potential supply chain disruptions due to geopolitical factors, regulatory changes, or manufacturing issues in the supplier's country.
However, the extensive number of shipments (323) and the involvement of 124 unique suppliers indicate a level of diversification within the supplier base. This diversification may help mitigate some risks associated with supplier concentration. Nonetheless, the Ministry may consider exploring additional suppliers and diversifying its product portfolio to enhance supply chain resilience and reduce potential vulnerabilities.
2Supply Chain Resilience
The resilience of the Ministry of Health's supply chain, particularly concerning imports from India, is influenced by several factors. The Ministry's reliance on a diverse set of 124 unique suppliers for 323 shipments suggests a level of diversification that can help mitigate risks associated with supply chain disruptions. This approach allows for flexibility in sourcing and can provide alternative options in case of issues with a particular supplier.
However, the high concentration of import value in the top five products—Vaccine, Tenofovir, Dolutegravir, Sodium, and Artemether—indicates that the Ministry's supply chain is heavily dependent on a narrow range of products. This concentration can pose challenges if there are disruptions in the production or availability of these specific products. To enhance supply chain resilience, the Ministry may consider diversifying its product portfolio and exploring additional suppliers, including those from other countries, to reduce dependency on a single source.
Additionally, the Ministry's collaboration with the Medical Stores Department (MSD) and adherence to regulatory guidelines set by the Tanzania Medicines and Medical Devices Authority (TMDA) contribute to the overall stability and compliance of the supply chain. Ensuring that all imported products meet the required quality standards and are registered with the TMDA is essential for maintaining a resilient and effective supply chain.
3Strategic Implications
The Ministry of Health's sourcing pattern, characterized by a significant reliance on Indian pharmaceutical manufacturers and a concentrated product portfolio, has several strategic implications. For the Ministry, this approach offers cost advantages due to competitive pricing and the availability of a wide range of generic medications from India. However, the heavy dependence on a limited number of products and suppliers may expose the Ministry to risks such as supply chain disruptions, regulatory changes, or geopolitical tensions affecting trade routes.
For Indian exporters, the Ministry's procurement strategy presents both opportunities and challenges. The existing strong presence of Indian suppliers indicates a well-established relationship and a preference for Indian products within the Ministry's procurement processes. This established relationship can facilitate smoother negotiations and potential for increased market share. However, the high concentration in product categories and suppliers suggests that Indian exporters may face challenges in introducing new products or diversifying their offerings within the Ministry's procurement framework.
To enhance their competitive position, Indian exporters may consider diversifying their product portfolios to align with the Ministry's needs, ensuring compliance with Tanzanian regulatory standards, and exploring opportunities for direct engagement with the Ministry to understand procurement priorities and requirements.
Importing Pharmaceuticals into Tanzania — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Tanzania
1Regulatory Authority & Framework
The Tanzania Medicines and Medical Devices Authority (TMDA) is the primary regulatory body overseeing the importation, exportation, and distribution of pharmaceutical products in Tanzania. Established under the Tanzania Medicines and Medical Devices Act, Cap. 219, TMDA is responsible for ensuring that all medicines and medical devices meet the required quality, safety, and efficacy standards before they enter the Tanzanian market. This includes the registration of pharmaceutical products, licensing of manufacturing and importing facilities, and monitoring of market compliance.
Key legislation governing pharmaceutical imports includes the Tanzania Medicines and Medical Devices Act, Cap. 219, and the Tanzania Food, Drugs and Cosmetics (Registration of Premises, Importation and Exportation of Pharmaceutical Products and Raw Materials) Regulations, 2015. These regulations outline the procedures and requirements for importing pharmaceutical products, including the necessity for importers to register their premises with TMDA and obtain import permits for each consignment. The guidelines also specify conditions related to product shelf-life, labeling, and compliance with Good Manufacturing Practice (GMP) standards.
For Indian pharmaceutical exporters, understanding and adhering to these regulatory frameworks is crucial. Ensuring that products are registered with TMDA, comply with Tanzanian labeling requirements, and meet GMP standards recognized by TMDA will facilitate smoother market entry and acceptance. Additionally, staying informed about any updates or changes in Tanzanian pharmaceutical regulations is essential for maintaining compliance and sustaining business relationships with Tanzanian importers like the Ministry of Health.
2Import Licensing & GMP
Import licensing in Tanzania is governed by the Tanzania Medicines and Medical Devices Authority (TMDA). Entities intending to import pharmaceutical products must register their premises with TMDA and obtain an import permit for each consignment. The application process involves submitting relevant documentation, including a proforma invoice and evidence of compliance with TMDA's guidelines. Importers are also required to import products through authorized Ports of Entry (PoE) as specified by TMDA. For controlled drugs, additional requirements apply, including obtaining special permits and adhering to specific importation procedures.
TMDA recognizes Good Manufacturing Practice (GMP) certificates from various international standards, including EU GMP, WHO GMP, and PIC/S. Pharmaceutical products imported into Tanzania must be manufactured in facilities that comply with these recognized GMP standards. This ensures that imported medicines meet the required quality, safety, and efficacy standards. For Indian exporters, obtaining GMP certification from recognized bodies and ensuring that manufacturing facilities
Frequently Asked Questions — Ministry OF Health
What products does Ministry OF Health import from India?
Ministry OF Health imports 8 pharmaceutical products across 5 categories. Top imports: Vaccine ($4.5M), Tenofovir ($3.5M), Dolutegravir ($3.0M), Sodium ($1.8M), Artemether ($850.0K).
Who supplies pharmaceuticals to Ministry OF Health from India?
Ministry OF Health sources from 124 verified Indian suppliers. The primary supplier is Cipla Limited (21.2% of imports, $12.9M).
What is Ministry OF Health's total pharmaceutical import value?
Ministry OF Health's total pharmaceutical import value from India is $14.0M, based on 323 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Ministry OF Health focus on?
Ministry OF Health imports across 5 categories. The largest: Antiviral & HIV Medications (48.2%), Other (32.5%), Nutritional Supplements (12.9%).
Get Full Ministry OF Health Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Ministry OF Health identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Ministry OF Health's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 323 individual customs records matching Ministry OF Health.
- 5.Supplier Verification: Ministry OF Health sources from 124 verified Indian suppliers across 2,437 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
8 Products Tracked
5 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.