Ministere DE Sante Publique Sg/msp
Pharmaceutical Importer · Niger · Antiviral & HIV Medications Focus · $200.0K Total Trade · DGFT Verified
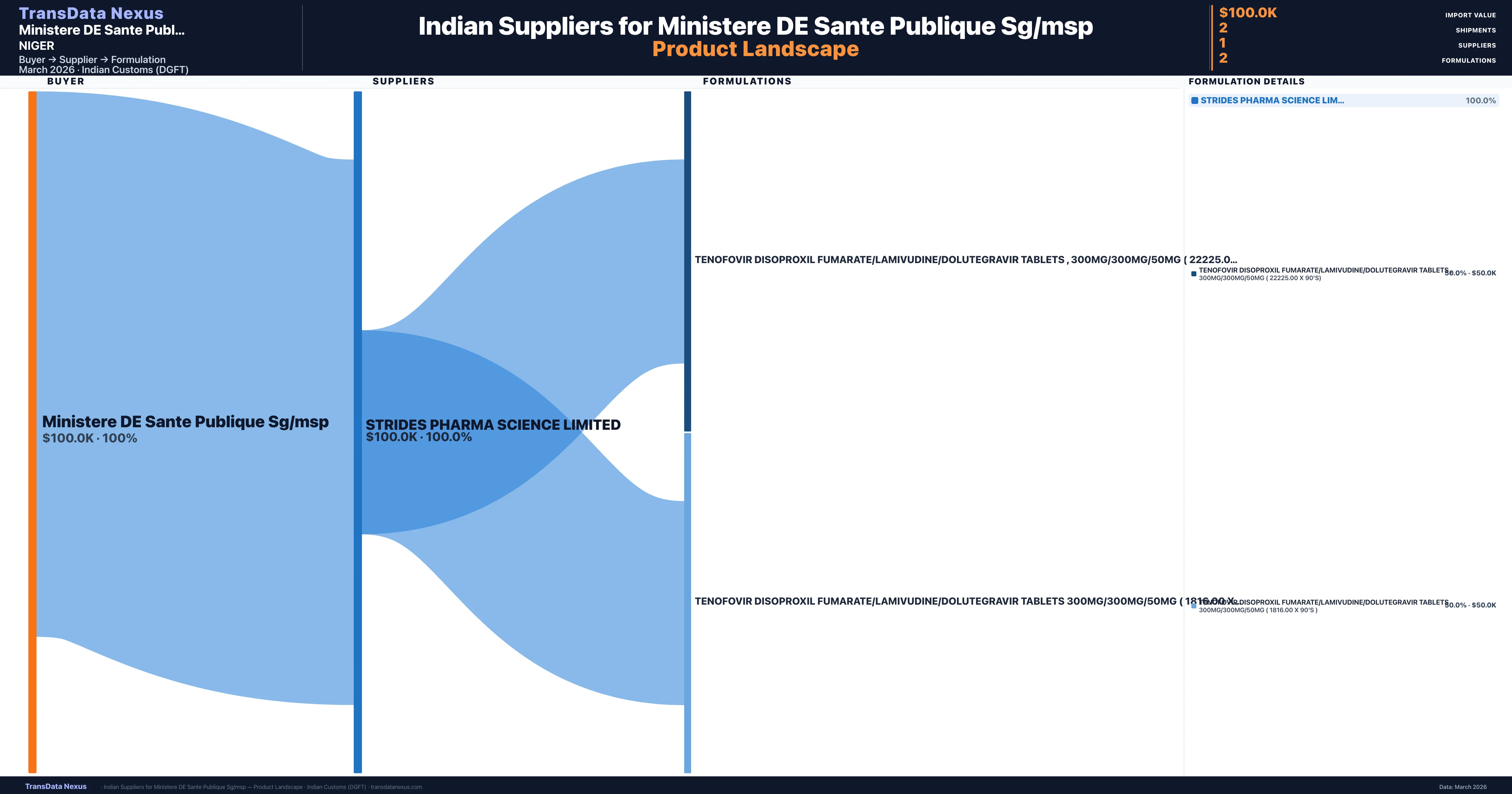
Ministere DE Sante Publique Sg/msp is a pharmaceutical importer based in Niger with a total trade value of $200.0K across 2 products in 1 therapeutic categories. Based on 4 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Ministere DE Sante Publique Sg/msp sources from 1 verified Indian supplier, with Strides Pharma Science Limited accounting for 100.0% of imports.
Ministere DE Sante Publique Sg/msp — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Ministere DE Sante Publique Sg/msp?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Strides Pharma Science Limited | $100.0K | 2 | 100.0% |
Ministere DE Sante Publique Sg/msp sources from 1 verified Indian supplier across 2 distinct formulations. The sourcing is highly concentrated — Strides Pharma Science Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Ministere DE Sante Publique Sg/msp Import?
| Formulation | Value | Ships |
|---|---|---|
| Tenofovir disoproxil fumarate/lamivudine/dolutegravir tablets , 300MG/300MG/50MG ( 22225.00 x 90's) | $50.0K | 1 |
| Tenofovir disoproxil fumarate/lamivudine/dolutegravir tablets 300MG/300MG/50MG ( 1816.00 x 90's ) | $50.0K | 1 |
Ministere DE Sante Publique Sg/msp imports 2 distinct pharmaceutical formulations. Showing top 2 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Ministere DE Sante Publique Sg/msp Import?
Top Products by Import Value
Ministere DE Sante Publique Sg/msp Therapeutic Categories — 1 Specializations
Ministere DE Sante Publique Sg/msp imports across 1 therapeutic categories, with Antiviral & HIV Medications (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antiviral & HIV Medications
2 products · 100.0% · $200.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Dolutegravir | Antiviral & HIV Medications | $100.0K | 2 | 0.0% | 4 |
| 2 | Tenofovir | Antiviral & HIV Medications | $100.0K | 2 | 0.0% | 4 |
Ministere DE Sante Publique Sg/msp imports 2 pharmaceutical products across 1 categories into Niger totaling $200.0K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Ministere DE Sante Publique Sg/msp.
Request DemoMinistere DE Sante Publique Sg/msp — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
The "Ministère de la Santé Publique Sg/msp" refers to the Ministry of Public Health of Niger, officially known as the Ministère de la Santé Publique, de la Population et des Affaires Sociales. This governmental body is responsible for formulating and implementing public health policies, overseeing healthcare services, and managing social affairs within Niger. Headquartered in Niamey, the capital city, the ministry operates under the leadership of the Minister of Public Health, currently Dr. Garba Hakimi.
As a government entity, the ministry does not function as a commercial pharmaceutical buyer or importer. Instead, it plays a pivotal role in the procurement and distribution of medical supplies and pharmaceuticals necessary for the country's public health initiatives. This includes managing the importation of essential medications, ensuring their quality, and overseeing their equitable distribution across various healthcare facilities in Niger. The ministry's operations are integral to maintaining the health infrastructure and addressing the medical needs of the population.
2Distribution Network
The Ministry of Public Health's distribution network is structured to ensure the efficient delivery of medical supplies and pharmaceuticals throughout Niger. While specific details about warehouse locations and logistics capabilities are not publicly disclosed, the ministry coordinates with regional health directorates and local health centers to facilitate the distribution process. This decentralized approach aims to provide timely access to medical resources across the country's diverse geographic regions.
The ministry's logistics operations are designed to address the unique challenges posed by Niger's vast and varied terrain. By leveraging partnerships with local authorities and international organizations, the ministry strives to enhance the reach and effectiveness of its distribution network, ensuring that essential medical supplies are accessible to all segments of the population.
3Industry Role
In Niger's pharmaceutical supply chain, the Ministry of Public Health serves as the central authority responsible for the procurement, regulation, and distribution of medical supplies and pharmaceuticals. Unlike private entities, the ministry's role is not that of a wholesaler, distributor, or hospital supplier. Instead, it functions as the primary governmental body overseeing the importation and allocation of medical resources, ensuring that they meet the country's public health standards and are distributed equitably across healthcare facilities.
The ministry's responsibilities encompass the establishment of procurement policies, negotiation with international suppliers, and the implementation of quality control measures. By centralizing these functions, the ministry aims to maintain a cohesive and efficient pharmaceutical supply chain that aligns with national health objectives and addresses the medical needs of the population.
Supplier Relationship Intelligence — Ministere DE Sante Publique Sg/msp
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The Ministry of Public Health's sourcing strategy, as evidenced by its importation of two specific pharmaceutical products—Dolutegravir and Tenofovir—each valued at $100,000 USD from a single supplier, STRIDES PHARMA SCIENCE LIMITED, indicates a high level of supplier concentration. This approach suggests a strategic decision to streamline procurement processes and potentially negotiate favorable terms with a trusted supplier.
However, such a concentrated sourcing strategy also introduces risks associated with dependency on a single supplier. Any disruptions in the supplier's operations, such as production delays, quality control issues, or geopolitical challenges, could adversely affect the ministry's ability to meet the country's pharmaceutical needs. Therefore, while the current strategy may offer operational efficiencies, it is essential for the ministry to assess and mitigate potential risks to ensure a resilient and uninterrupted supply chain.
2Supply Chain Resilience
The resilience of the Ministry of Public Health's supply chain, particularly concerning its reliance on a single Indian supplier for key pharmaceutical imports, is a critical consideration. The absence of publicly available information regarding backup suppliers or alternative sourcing strategies suggests a potential vulnerability in the supply chain. This lack of diversification could expose the ministry to significant risks if the primary supplier encounters operational challenges.
To enhance supply chain resilience, it would be prudent for the ministry to explore the establishment of relationships with additional suppliers, both within India and internationally. Diversifying the supplier base can provide a buffer against potential disruptions and ensure a more stable and reliable supply of essential pharmaceuticals. Additionally, evaluating the regulatory compliance and quality assurance practices of potential suppliers is crucial to maintain the standards of medical products distributed within Niger.
3Strategic Implications
The Ministry of Public Health's current sourcing pattern, characterized by a concentrated procurement strategy from a single supplier, has several strategic implications. For the ministry, this approach may offer streamlined procurement processes and the potential for favorable pricing agreements. However, the associated risks of supplier dependency necessitate careful risk management and contingency planning.
For Indian pharmaceutical exporters, the ministry's sourcing pattern presents both opportunities and challenges. While the existing relationship with STRIDES PHARMA SCIENCE LIMITED indicates a preference for established suppliers, there may be opportunities to engage with the ministry by demonstrating compliance with Niger's regulatory standards and offering competitive pricing. However, any new entrants would need to navigate the complexities of the ministry's procurement processes and establish trust to become viable suppliers.
Importing Pharmaceuticals into Niger — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Niger
1Regulatory Authority & Framework
In Niger, the regulation of pharmaceutical imports is overseen by the Ministry of Public Health, which is responsible for setting policies and standards for the importation, distribution, and use of medical products. The ministry's role includes ensuring that imported pharmaceuticals meet the country's health and safety standards, thereby protecting public health. Specific legislative frameworks governing pharmaceutical imports are not detailed in the available sources; however, the ministry's oversight encompasses the entire pharmaceutical supply chain, from importation to distribution.
The approval process for pharmaceutical imports typically involves the submission of documentation demonstrating the product's safety, efficacy, and quality. This may include certificates of analysis, manufacturing licenses, and compliance with international standards. The ministry evaluates these submissions to grant marketing authorization, ensuring that only products meeting the required criteria are permitted for sale and distribution within Niger.
2Import Licensing & GMP
Import licensing requirements in Niger are managed by the Ministry of Public Health, which stipulates that all pharmaceutical imports must be accompanied by appropriate documentation. This documentation includes certificates of analysis, manufacturing licenses, and evidence of compliance with Good Manufacturing Practices (GMP). While specific details about the recognition of GMP certificates from other jurisdictions, such as EU GMP, WHO GMP, or PIC/S, are not explicitly stated in the available sources, it is common for regulatory authorities to accept such certifications, provided they meet the ministry's standards.
Wholesale distribution authorization is also required for entities involved in the distribution of pharmaceuticals within Niger. This authorization ensures that distributors adhere to the regulatory standards set by the Ministry of Public Health, maintaining the integrity and safety of the pharmaceutical supply chain.
3Quality & Labeling
Pharmaceutical products imported into Niger are subject to quality control measures to ensure their safety and efficacy. Batch testing and stability studies are conducted to verify that products meet the required standards. Labeling requirements include the use of the French language, as it is the official language of Niger. Specific details regarding serialization mandates are not provided in the available sources; however, serialization is a common practice aimed at enhancing traceability and combating counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, there have been no publicly documented policy changes affecting Indian pharmaceutical imports into Niger. The Ministry of Public Health continues to enforce existing regulations governing the importation and distribution of medical products. However, it is advisable for stakeholders to monitor official communications from the ministry for any updates or changes to regulatory frameworks that may impact pharmaceutical imports.
Ministere DE Sante Publique Sg/msp — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
The Ministry of Public Health's focus on importing Dolutegravir and Tenofovir, both antiretroviral medications, indicates a strategic emphasis on addressing the HIV/AIDS epidemic in Niger. These medications are essential components of antiretroviral therapy regimens, which are critical for managing and treating HIV infections. The decision to import these specific products aligns with the ministry's objective to provide effective treatment options and improve health outcomes for individuals living with HIV/AIDS in the country.
The market demand for these medications is driven by the prevalence of HIV/AIDS in Niger and the need for accessible and effective treatment options. By focusing on these therapeutic areas, the ministry aims to enhance the quality of life for affected individuals and reduce the transmission of the virus within the population.
2Sourcing Profile
The Ministry of Public Health's sourcing strategy for antiretroviral medications involves procuring finished pharmaceutical formulations from a single supplier, STRIDES PHARMA SCIENCE LIMITED in India. This approach suggests a preference for established suppliers that can meet the ministry's quality standards and regulatory requirements. The choice to import finished formulations, rather than active pharmaceutical ingredients (APIs) or bulk drugs, indicates a focus on ensuring the availability of ready-to-use medications that can be directly distributed to healthcare facilities.
India's role in the ministry's overall procurement strategy is significant, given its capacity to produce high-quality generic pharmaceuticals at competitive prices. The country's adherence to international manufacturing standards and its established reputation in the global pharmaceutical market make it a preferred sourcing destination for the ministry.
3Market Positioning
Based on the importation of antiretroviral medications, the Ministry of Public Health serves the segment of the Niger market focused on public health initiatives, particularly in the treatment and
Frequently Asked Questions — Ministere DE Sante Publique Sg/msp
What products does Ministere DE Sante Publique Sg/msp import from India?
Ministere DE Sante Publique Sg/msp imports 2 pharmaceutical products across 1 categories. Top imports: Dolutegravir ($100.0K), Tenofovir ($100.0K).
Who supplies pharmaceuticals to Ministere DE Sante Publique Sg/msp from India?
Ministere DE Sante Publique Sg/msp sources from 1 verified Indian suppliers. The primary supplier is Strides Pharma Science Limited (100.0% of imports, $100.0K).
What is Ministere DE Sante Publique Sg/msp's total pharmaceutical import value?
Ministere DE Sante Publique Sg/msp's total pharmaceutical import value from India is $200.0K, based on 4 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Ministere DE Sante Publique Sg/msp focus on?
Ministere DE Sante Publique Sg/msp imports across 1 categories. The largest: Antiviral & HIV Medications (100.0%).
Get Full Ministere DE Sante Publique Sg/msp Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Ministere DE Sante Publique Sg/msp identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Ministere DE Sante Publique Sg/msp's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 4 individual customs records matching Ministere DE Sante Publique Sg/msp.
- 5.Supplier Verification: Ministere DE Sante Publique Sg/msp sources from 1 verified Indian suppliers across 2 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.