Mermisi Llc
Pharmaceutical Importer · Georgia · Oncology Focus · $2.3M Total Trade · DGFT Verified
Mermisi Llc is a pharmaceutical importer based in Georgia with a total trade value of $2.3M across 5 products in 3 therapeutic categories. Based on 63 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Mermisi Llc sources from 6 verified Indian suppliers, with Getwell Pharma India Private Limited accounting for 66.1% of imports.
Mermisi Llc — Import Portfolio & Supplier Network
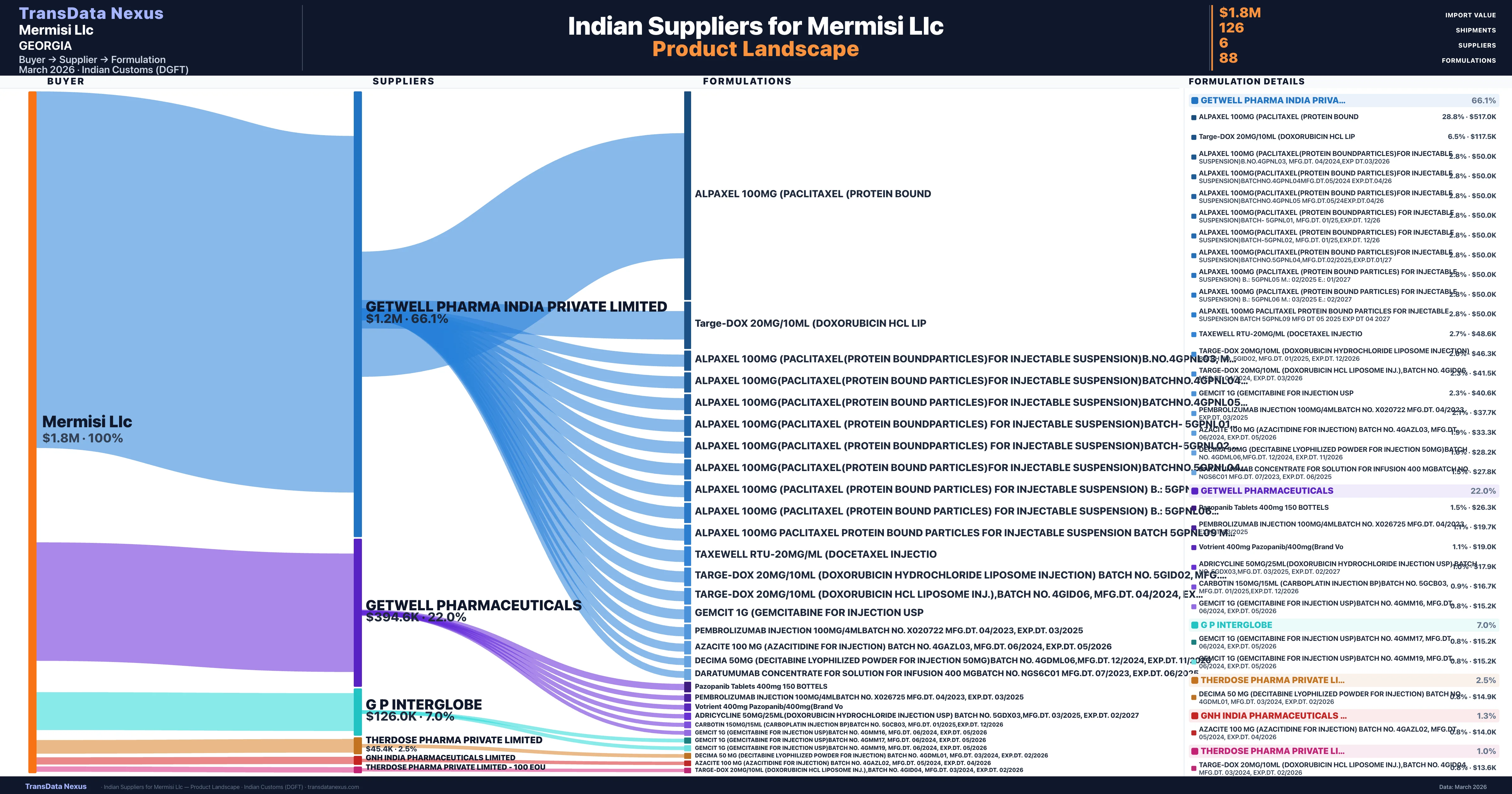
Who Are the Verified Indian Suppliers to Mermisi Llc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Getwell Pharma India Private Limited | $1.2M | 55 | 66.1% |
| Getwell Pharmaceuticals | $394.6K | 20 | 22.0% |
| G P Interglobe | $126.0K | 19 | 7.0% |
| Therdose Pharma Private Limited | $45.4K | 4 | 2.5% |
| Gnh India Pharmaceuticals Limited | $24.1K | 4 | 1.3% |
| Therdose Pharma Private Limited - 100 Eou | $17.8K | 24 | 1.0% |
Mermisi Llc sources from 6 verified Indian suppliers across 88 distinct formulations. The supply base is diversified across 6 suppliers, reducing single-source dependency risk.
What Formulations Does Mermisi Llc Import?
| Formulation | Value | Ships |
|---|---|---|
| Alpaxel 100MG (paclitaxel (protein | $517.0K | 12 |
| Targe-dox 20MG/10ML (doxorubicin hcl | $117.5K | 4 |
| Alpaxel 100MG (paclitaxel(protein boundparticles)for injectable suspension)b.no.4gpnl03, mfg.dt. 04/2024,exp | $50.0K | 1 |
| Alpaxel 100MG(paclitaxel(protein bound particles)for injectable suspension)batchno.4gpnl04mfg.dt.05/2024 | $50.0K | 1 |
| Alpaxel 100MG(paclitaxel(protein bound particles)for injectable suspension)batchno.4gpnl05 | $50.0K | 1 |
| Alpaxel 100MG(paclitaxel (protein boundparticles) for injectable suspension)batch- 5gpnl01, mfg.dt. 01/25,exp.dt. | $50.0K | 1 |
| Alpaxel 100MG(paclitaxel (protein boundparticles) for injectable suspension)batch-5gpnl02, mfg.dt. 01/25,exp.dt. | $50.0K | 1 |
| Alpaxel 100MG(paclitaxel(protein bound particles)for injectable suspension)batchno.5gpnl04,mfg.dt.02/2025 | $50.0K | 1 |
| Alpaxel 100MG (paclitaxel (protein bound particles) for injectable suspension) b.: 5gpnl05 m.: 02/2025 e. | $50.0K | 1 |
| Alpaxel 100MG (paclitaxel (protein bound particles) for injectable suspension) b.: 5gpnl06 m.: 03/2025 e. | $50.0K | 1 |
| Alpaxel 100MG paclitaxel protein bound particles for injectable suspension batch 5gpnl09 mfg dt 05 2025 exp dt 04 | $50.0K | 1 |
| Taxewell rtu-20MG/ML (docetaxel | $48.6K | 2 |
| Targe-dox 20MG/10ML (doxorubicin hydrochloride liposome injection) batch no. 5gid02, mfg.dt. 01/2025, exp.dt. | $46.3K | 1 |
| Targe-dox 20MG/10ML (doxorubicin hcl liposome INJ.),batch no. 4gid06, mfg.dt. 04/2024, exp.dt. | $41.5K | 1 |
| Gemcit 1g (gemcitabine for injection | $40.6K | 4 |
Mermisi Llc imports 88 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Mermisi Llc Import?
Top Products by Import Value
Mermisi Llc Therapeutic Categories — 3 Specializations
Mermisi Llc imports across 3 therapeutic categories, with Oncology (49.1%), Nutritional Supplements (44.9%), Advanced Oncology (6.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Oncology
1 products · 49.1% · $1.1M
Nutritional Supplements
1 products · 44.9% · $1.1M
Advanced Oncology
3 products · 6.0% · $141.1K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Paclitaxel | Oncology | $1.1M | 23 | 1.4% | 6 |
| 2 | Protein | Nutritional Supplements | $1.1M | 21 | 7.1% | 2 |
| 3 | Pembrolizumab | Advanced Oncology | $59.9K | 6 | 3.1% | 3 |
| 4 | Daratumumab | Advanced Oncology | $54.0K | 8 | 4.0% | 6 |
| 5 | Pazopanib | Advanced Oncology | $27.1K | 5 | 0.3% | 9 |
Mermisi Llc imports 5 pharmaceutical products across 3 categories into Georgia totaling $2.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Mermisi Llc.
Request DemoMermisi Llc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Mermisi LLC, established in 1993, is a privately held pharmaceutical company headquartered in Tbilisi, Georgia. Specializing in the registration, import, and distribution of pharmaceutical products, Mermisi has been a leader in the Georgian pharmaceutical sector for over three decades. The company focuses on introducing innovative medical products, including targeted anticancer medicines, biosimilars, and advanced biologic and nanotechnologically advanced drugs, thereby contributing significantly to public health and welfare in Georgia. (mermisi.ge)
With a team of 11-50 employees, Mermisi LLC has developed strong partnerships with global pharmaceutical manufacturers, facilitating the launch and scaling of oncology products in Georgia. Their comprehensive services encompass regulatory approval, reimbursement, distribution, promotion, and long-term market growth, positioning them as a preferred partner for market entry and commercial success in Georgia's specialty pharmaceutical sector. (mermisi.ge)
2Distribution Network
Mermisi LLC operates a centralized distribution network from its headquarters in Tbilisi, Georgia. While specific details about warehouse locations and logistics capabilities are not publicly disclosed, the company's extensive experience and partnerships suggest a well-established infrastructure capable of efficiently managing the import and distribution of pharmaceutical products across Georgia. Their focus on oncology and specialty pharmaceuticals indicates a strategic approach to logistics, ensuring timely and reliable delivery to healthcare providers nationwide.
3Industry Role
In Georgia's pharmaceutical supply chain, Mermisi LLC functions primarily as a pharmaceutical importer and distributor. The company plays a pivotal role in introducing and supplying innovative medical products, particularly in the oncology and specialty sectors, to the Georgian market. By partnering with global manufacturers, Mermisi ensures the availability of advanced therapies, thereby enhancing the quality of healthcare services in the country. (mermisi.ge)
Supplier Relationship Intelligence — Mermisi Llc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Mermisi LLC's sourcing strategy reveals a significant concentration on a select group of suppliers. The company's total import value from India amounts to $2.3 million USD, with 63 shipments covering five products across three therapeutic categories. The top five products imported include Paclitaxel and Protein, each valued at $1.1 million USD, accounting for 1.4% and 7.1% of the total share, respectively. This concentration indicates a strategic choice to focus on high-value, specialized products, potentially leveraging supplier expertise and favorable pricing.
The shipment data, with 55 shipments from GETWELL PHARMA INDIA PRIVATE LIMITED and 20 from GETWELL PHARMACEUTICALS, suggests a stable and ongoing relationship with these suppliers. However, the reliance on a limited number of suppliers may pose risks related to supply chain disruptions or changes in supplier dynamics. Diversifying the supplier base could mitigate such risks and enhance supply chain resilience.
2Supply Chain Resilience
Mermisi LLC's supply chain resilience appears robust, given the company's longstanding partnerships with key Indian suppliers. The diversity in imported formulations, with 88 unique products, indicates a broad sourcing strategy within the oncology and specialty pharmaceutical sectors. While the primary suppliers are GETWELL PHARMA INDIA PRIVATE LIMITED and GETWELL PHARMACEUTICALS, the presence of additional suppliers like G P INTERGLOBE and THERDOSE PHARMA PRIVATE LIMITED suggests a level of diversification.
However, the concentration on a few suppliers may expose Mermisi LLC to risks such as supply chain disruptions or changes in supplier dynamics. To enhance resilience, the company could consider expanding its supplier base and exploring alternative sourcing options. Ensuring that all suppliers comply with Good Manufacturing Practice (GMP) standards is crucial for maintaining product quality and regulatory compliance.
3Strategic Implications
Mermisi LLC's focused sourcing strategy positions the company as a key player in the oncology and specialty pharmaceutical markets in Georgia. By importing high-value products like Paclitaxel and Protein, Mermisi demonstrates a commitment to providing advanced therapies to the Georgian healthcare system. This strategic focus allows the company to build strong relationships with suppliers and establish a reputation for quality and reliability.
For Indian exporters, Mermisi LLC represents a valuable partner for distributing specialized pharmaceutical products in Georgia. Understanding Mermisi's product preferences and regulatory requirements can facilitate smoother market entry and collaboration. Expanding the supplier base and offering a diverse range of formulations could further strengthen Mermisi's market position and provide additional opportunities for Indian exporters.
Importing Pharmaceuticals into Georgia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Georgia
1Regulatory Authority & Framework
In Georgia, the regulation of pharmaceutical products is overseen by the Agency of State Regulation of Medical Activities at the Ministry of Labor, Health and Social Protection. This agency is responsible for the registration, import, and distribution of pharmaceutical products, ensuring compliance with national and international standards. The legal framework governing pharmaceutical activities includes the Law of Georgia on Medicines and Pharmaceutical Activities, which outlines the procedures for drug registration, importation, and distribution. (matsne.gov.ge)
The registration process for pharmaceutical products in Georgia can be conducted through two regimes: national and recognition. The recognition regime allows for the simplified registration of drugs already approved by stringent regulatory authorities, such as the European Medicines Agency (EMA) or the U.S. Food and Drug Administration (FDA), without the need for re-testing. This facilitates faster market entry for products that have already undergone rigorous evaluation. (mkdlaw.ge)
2Import Licensing & GMP
Importing pharmaceutical products into Georgia requires obtaining a specific permit from the Regulation Agency. Additionally, warehouses must meet Good Distribution Practice (GDP) standards to ensure the quality and integrity of the products during storage and transportation. Compliance with GDP is mandatory for pharmaceutical distributors in Georgia, as it ensures that medicines are consistently stored, transported, and handled under suitable conditions. (legal.ge)
Good Manufacturing Practice (GMP) certification is also mandatory for pharmaceutical manufacturers in Georgia. This certification ensures that products are consistently produced and controlled according to quality standards, thereby safeguarding public health. For importers and distributors, recognizing GMP certificates from reputable authorities, such as the European Union (EU) GMP, World Health Organization (WHO) GMP, or Pharmaceutical Inspection Co-operation Scheme (PIC/S), is essential for maintaining product quality and regulatory compliance. (legal.ge)
3Quality & Labeling
Pharmaceutical products imported into Georgia must undergo batch testing to ensure they meet the required quality standards. Stability studies are also conducted to determine the shelf life and storage conditions of the products. Labeling requirements stipulate that all information, including the Patient Information Leaflet (PIL), must be provided in the Georgian language. This ensures that patients and healthcare providers can accurately understand the product's usage, dosage, and potential side effects. (legal.ge)
Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the market. These requirements involve assigning a unique serial number to each saleable unit of a pharmaceutical product, facilitating tracking throughout the supply chain. Compliance with these standards is crucial for maintaining the integrity of the pharmaceutical supply chain and ensuring patient safety.
4Recent Regulatory Changes
Between 2024 and 2026, Georgia has implemented several policy changes affecting pharmaceutical imports. Notably, the country has introduced a reference pricing system for a specific list of essential medicines, setting price caps to make medications more affordable. Selling above the reference price is illegal and results in fines. Additionally, compliance with Good Manufacturing Practice (GMP) and Good Distribution Practice (GDP) standards has been made mandatory for manufacturers, importers, and distributors since 2022, ensuring consistent product quality and safety. (legal.ge)
Mermisi Llc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Mermisi LLC's product strategy focuses on high-value, specialized pharmaceutical products, particularly in the oncology and specialty sectors. The company's top five imported products—Paclitaxel, Protein, Pembrolizumab, Daratumumab, and Pazopanib—align with this focus, addressing critical therapeutic areas with significant market demand. This strategic emphasis allows Mermisi to meet the needs of patients requiring advanced therapies and positions the company as a leader in providing innovative treatments in Georgia. (mermisi.ge)
The market demand for these products is driven by the increasing prevalence of cancer and other complex diseases in Georgia, necessitating access to cutting-edge treatments. By importing these specialized products, Mermisi LLC contributes to improving patient outcomes and advancing the standard of care in the country.
2Sourcing Profile
Mermisi LLC's sourcing strategy is centered on importing finished pharmaceutical formulations from India, focusing on generic drugs and specialized formulations. The company's preference for finished products allows for quicker market entry and ensures that the imported medicines are ready for distribution without the need for additional processing. India's robust pharmaceutical manufacturing sector, known for its compliance with international quality standards, makes it a strategic sourcing partner for Mermisi.
Frequently Asked Questions — Mermisi Llc
What products does Mermisi Llc import from India?
Mermisi Llc imports 5 pharmaceutical products across 3 categories. Top imports: Paclitaxel ($1.1M), Protein ($1.1M), Pembrolizumab ($59.9K), Daratumumab ($54.0K), Pazopanib ($27.1K).
Who supplies pharmaceuticals to Mermisi Llc from India?
Mermisi Llc sources from 6 verified Indian suppliers. The primary supplier is Getwell Pharma India Private Limited (66.1% of imports, $1.2M).
What is Mermisi Llc's total pharmaceutical import value?
Mermisi Llc's total pharmaceutical import value from India is $2.3M, based on 63 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Mermisi Llc focus on?
Mermisi Llc imports across 3 categories. The largest: Oncology (49.1%), Nutritional Supplements (44.9%), Advanced Oncology (6.0%).
Get Full Mermisi Llc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Mermisi Llc identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Mermisi Llc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 63 individual customs records matching Mermisi Llc.
- 5.Supplier Verification: Mermisi Llc sources from 6 verified Indian suppliers across 88 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.