Medical Stores Department
Pharmaceutical Importer · Tanzania · Antibiotics Focus · $500.0K Total Trade · DGFT Verified
Medical Stores Department is a pharmaceutical importer based in Tanzania with a total trade value of $500.0K across 3 products in 3 therapeutic categories. Based on 10 verified import shipments from Indian Customs (DGFT) records, Medical Stores Department is the #1 buyer in 1 product including Captopril. Medical Stores Department sources from 22 verified Indian suppliers, with Lincoln Pharmaceuticals Limited accounting for 28.3% of imports.
Medical Stores Department — Import Portfolio & Supplier Network
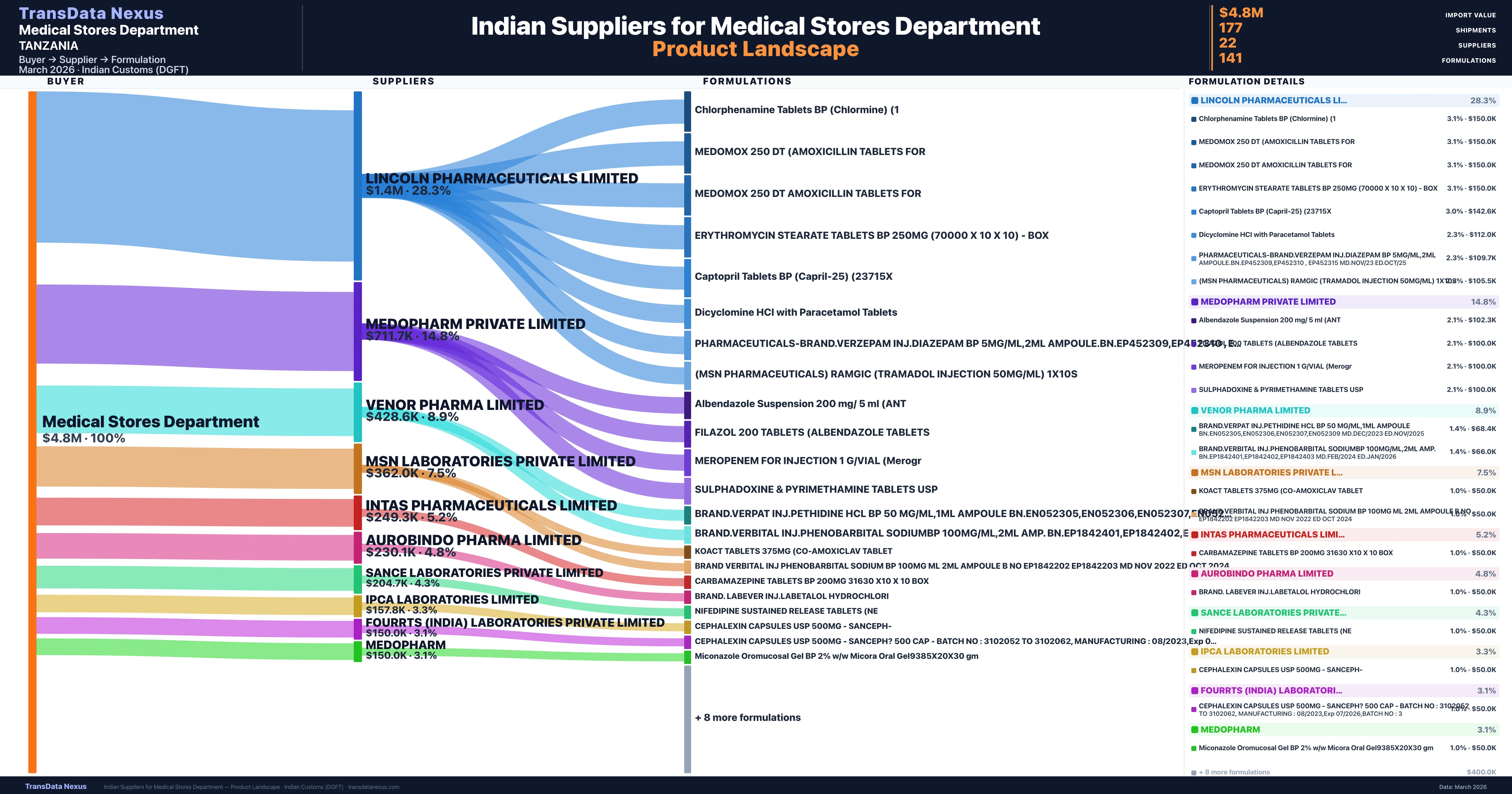
Who Are the Verified Indian Suppliers to Medical Stores Department?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Lincoln Pharmaceuticals Limited | $1.4M | 35 | 28.3% |
| Medopharm Private Limited | $711.7K | 15 | 14.8% |
| Venor Pharma Limited | $428.6K | 24 | 8.9% |
| Msn Laboratories Private Limited | $362.0K | 20 | 7.5% |
| Intas Pharmaceuticals Limited | $249.3K | 7 | 5.2% |
| Aurobindo Pharma Limited | $230.1K | 7 | 4.8% |
| Sance Laboratories Private Limited | $204.7K | 5 | 4.3% |
| Ipca Laboratories Limited | $157.8K | 7 | 3.3% |
| Fourrts (india) Laboratories Private Limited | $150.0K | 3 | 3.1% |
| Medopharm | $150.0K | 3 | 3.1% |
| Medo Pharm Private Limited | $150.0K | 3 | 3.1% |
| Intermed Laboratories Private Limited | $139.7K | 8 | 2.9% |
| Arechar Healthcare Llp | $90.3K | 5 | 1.9% |
| The Madras Pharmaceuticals | $67.5K | 4 | 1.4% |
| Mankind Pharma Limited | $57.7K | 3 | 1.2% |
| Brawn Laboratories Limited | $54.7K | 2 | 1.1% |
| Verve Human Care Laboratories | $53.0K | 4 | 1.1% |
| Naprod Life Sciences Private Limited | $43.3K | 2 | 0.9% |
| Sanpras Healthcare Private Limited | $41.3K | 6 | 0.9% |
| Premier Medical Agency | $36.1K | 10 | 0.8% |
| Baxter Pharmaceuticals India Private Limited | $35.9K | 1 | 0.7% |
| Fdc Limited | $30.3K | 3 | 0.6% |
Medical Stores Department sources from 22 verified Indian suppliers across 141 distinct formulations. The supply base is diversified across 22 suppliers, reducing single-source dependency risk.
What Formulations Does Medical Stores Department Import?
| Formulation | Value | Ships |
|---|---|---|
| Chlorphenamine tablets BP (chlormine) | $150.0K | 3 |
| Medomox 250 dt (amoxicillin tablets | $150.0K | 3 |
| Medomox 250 dt amoxicillin tablets | $150.0K | 3 |
| Erythromycin stearate tablets BP 250MG (70000 x 10 x 10) - | $150.0K | 3 |
| Captopril tablets BP (capril-25) | $142.6K | 3 |
| Dicyclomine hci with paracetamol | $112.0K | 3 |
| Pharmaceuticals-brand.verzepam INJ.diazepam BP 5MG/ML,2ML ampoule.bn.ep452309,ep452310 , ep452315 md.nov/23 | $109.7K | 3 |
| (msn pharmaceuticals) ramgic (tramadol injection 50MG/ML) 1x10s | $105.5K | 3 |
| Albendazole suspension 200 MG/ 5 ML | $102.3K | 3 |
| Filazol 200 tablets (albendazole | $100.0K | 2 |
| Meropenem for injection 1 g/vial | $100.0K | 2 |
| Sulphadoxine & pyrimethamine tablets | $100.0K | 2 |
| Brand.verpat INJ.pethidine hcl BP 50 MG/ML,1ML ampoule bn.en052305,en052306,en052307,en052309 md.dec/2023 | $68.4K | 3 |
| Brand.verbital INJ.phenobarbital sodiumbp 100MG/ML,2ML amp. bn.ep1842401,ep1842402,ep1842403 md.feb/2024 | $66.0K | 3 |
| Koact tablets 375MG (co-amoxiclav | $50.0K | 1 |
Medical Stores Department imports 141 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Medical Stores Department Import?
Top Products by Import Value
Medical Stores Department Therapeutic Categories — 3 Specializations
Medical Stores Department imports across 3 therapeutic categories, with Antibiotics (50.0%), Cardiovascular (30.0%), Antimalarial & Antiparasitic (20.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antibiotics
1 products · 50.0% · $250.0K
Cardiovascular
1 products · 30.0% · $150.0K
Antimalarial & Antiparasitic
1 products · 20.0% · $100.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Erythromycin | Antibiotics | $250.0K | 5 | 0.3% | 5 |
| 2 | Captopril | Cardiovascular | $150.0K | 3 | 1.2% | 1 |
| 3 | Pyrimethamine | Antimalarial & Antiparasitic | $100.0K | 2 | 0.2% | 7 |
Medical Stores Department imports 3 pharmaceutical products across 3 categories into Tanzania totaling $500.0K. The company is the #1 buyer for 1 product: Captopril.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Medical Stores Department.
Request DemoMedical Stores Department — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
The Medical Stores Department (MSD) is an autonomous government agency under Tanzania's Ministry of Health, established by Act No. 13 of 1993. Its primary mandate is to develop, maintain, and manage an efficient and cost-effective system for the procurement, storage, and distribution of approved medicines and medical supplies to public health facilities across the country. (msd.go.tz)
Headquartered in Dar es Salaam, Tanzania, MSD operates through ten zonal stores located in Dar es Salaam, Mwanza, Iringa, Kilimanjaro, Mbeya, Tabora, Dodoma, Tanga, Mtwara, and Kagera. This extensive network ensures the timely and reliable delivery of health commodities to various regions, supporting the nation's public health infrastructure. (msd.go.tz)
2Distribution Network
MSD's distribution network is strategically designed to cover the entire Tanzanian territory. With ten zonal stores and additional sales points in Tanga and Muleba, the department ensures that health commodities are accessible to all public health facilities nationwide. This widespread presence facilitates efficient logistics and timely delivery of medical supplies, contributing to the overall effectiveness of Tanzania's healthcare system. (msd.go.tz)
3Industry Role
In Tanzania's pharmaceutical supply chain, MSD functions as the primary wholesaler and distributor of medical supplies to public health facilities. By managing the procurement, storage, and distribution of health commodities, MSD plays a pivotal role in ensuring the availability of essential medicines and medical supplies across the country. This centralized approach enhances the efficiency and reliability of the healthcare supply chain, directly impacting the quality of care provided to the Tanzanian population. (msd.go.tz)
Supplier Relationship Intelligence — Medical Stores Department
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
MSD's sourcing strategy reveals a significant concentration in its supplier base, with a total import value of $500K USD from India across 10 shipments. The top three products imported are Captopril ($150K), Erythromycin ($250K), and Pyrimethamine ($100K), accounting for 1.2%, 0.3%, and 0.2% of the total import value, respectively. This concentration indicates a strategic choice to source specific products from a limited number of suppliers, potentially streamlining procurement processes and ensuring consistent quality. However, such dependency may pose risks if any of these suppliers face operational challenges or regulatory issues.
The shipment data, comprising 35 shipments from Lincoln Pharmaceuticals Limited and 15 from Medopharm Private Limited, suggests a stable and ongoing relationship with these suppliers. The consistent volume and frequency of shipments indicate a reliable supply chain, which is crucial for maintaining the uninterrupted availability of essential medicines. Nonetheless, the limited number of suppliers could be a vulnerability if there are disruptions in their operations or if they are unable to meet MSD's demand.
2Supply Chain Resilience
MSD's supply chain resilience is closely tied to its sourcing strategy from Indian suppliers. The department's reliance on a select group of suppliers for key products suggests a streamlined procurement process but also highlights potential vulnerabilities. The absence of backup suppliers for these critical products could lead to supply disruptions if any primary supplier encounters issues. Additionally, the limited diversity in product formulations imported from India may restrict MSD's ability to adapt to changing market demands or regulatory requirements.
Shipping route exposure is another factor affecting supply chain resilience. While specific shipping routes are not detailed, reliance on international shipping introduces risks related to geopolitical tensions, logistical challenges, and potential delays. Ensuring that key suppliers comply with international regulatory standards, such as WHO Good Manufacturing Practices (GMP), is essential to maintain product quality and safety. Regular audits and quality assessments of suppliers can mitigate some of these risks.
3Strategic Implications
MSD's sourcing pattern, characterized by a concentrated supplier base and limited product diversification, positions the department to negotiate favorable terms with its suppliers, potentially securing better pricing and consistent product quality. This strategic focus can lead to cost efficiencies and streamlined operations. However, the lack of supplier diversification and product variety may limit MSD's ability to respond swiftly to changes in market demand or to introduce new products that could enhance patient care.
For Indian exporters, understanding MSD's sourcing preferences presents an opportunity to strengthen existing relationships by ensuring consistent product quality and reliability. However, the limited number of suppliers and products may also indicate a saturated market, making it challenging for new entrants to establish a foothold without offering unique value propositions or addressing specific unmet needs within MSD's procurement strategy.
Importing Pharmaceuticals into Tanzania — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Tanzania
1Regulatory Authority & Framework
The Tanzania Medicines and Medical Devices Authority (TMDA) is the primary regulatory body overseeing the importation and exportation of medicines, medical devices, and related products in Tanzania. Established under the Tanzania Medicines and Medical Devices Act Cap. 219, TMDA is responsible for ensuring that all pharmaceutical products imported into the country are safe, effective, and of high quality. (tmda.go.tz)
Key legislation governing pharmaceutical imports includes the Tanzania Medicines and Medical Devices Act Cap. 219, which outlines the regulatory framework for the importation, registration, and distribution of medical products. This Act mandates that all pharmaceutical products must be registered with TMDA before importation and sets forth the conditions for import permits, including requirements related to product shelf-life and storage conditions. (tmda.go.tz)
The marketing authorization pathway for Indian generics involves several steps:
1. Product Registration: Indian manufacturers must submit an application to TMDA, providing comprehensive documentation on the product's quality, safety, and efficacy.
2. Import Permit Application: Once registered, importers must apply for an import permit through TMDA's online trader portal, ensuring compliance with all regulatory requirements.
3. Inspection and Approval: TMDA conducts inspections and evaluations to verify compliance with Tanzanian standards before granting approval for importation.
2Import Licensing & GMP
Import licensing requirements in Tanzania stipulate that all pharmaceutical products must be registered with TMDA prior to importation. Importers must ensure that their premises are duly registered with TMDA and adhere to the conditions prescribed under the Guidelines for Importation and Exportation of Pharmaceutical Products. Applications for import permits should be submitted at least six weeks prior to loading the consignment to avoid storage costs at ports of entry and potential disposal costs for unpermitted products. (tmda.go.tz)
TMDA recognizes Good Manufacturing Practice (GMP) certificates from various international standards, including WHO GMP, EU GMP, and PIC/S. Indian exporters seeking to supply MSD must ensure that their manufacturing facilities hold valid GMP certifications from recognized authorities to meet TMDA's quality standards. Additionally, importers must obtain wholesale distribution authorization from TMDA, demonstrating their capability to handle and distribute pharmaceutical products in compliance with Tanzanian regulations.
3Quality & Labeling
Batch testing and stability requirements are critical components of TMDA's regulatory framework. Pharmaceutical products imported into Tanzania must undergo batch testing to confirm their quality, safety, and efficacy. Products must also meet stability requirements to ensure they remain effective throughout their shelf-life. The remaining shelf-life of a product at the time of importation must be at least 60% for products with a shelf-life greater than 24 months and at least 80% for products with a shelf-life of 24 months or less. (tmda.go.tz)
Labeling requirements include the use of the English language for all labeling and packaging materials. Serialization mandates may be implemented to enhance traceability and prevent counterfeit products, although specific requirements should be confirmed with TMDA. Compliance with these standards is essential for the successful importation and distribution of pharmaceutical products in Tanzania.
4Recent Regulatory Changes
Between 2024 and 2026, Tanzania's regulatory framework for pharmaceutical imports has undergone several amendments:
1. Amendment of Act No. 13 of 1993: The Written Laws (Miscellaneous Amendments) (Act No. 4) of 2021 granted MSD corporate status, including perpetual succession and the ability to perform all actions legally allowed for a corporate body. (msd.go.tz)
2. Investment Authorization: The Written Laws (Miscellaneous Amendments) (No. 2) Act, 2024 authorized MSD to invest, subject to directives issued by the Treasury Registrar. (msd.go.tz)
3. Import Permit Application Timeline: Regulations now require import permit applications to be submitted at least six weeks prior to loading the consignment to avoid storage costs at ports of entry and potential disposal costs for unpermitted products. (tmda.go.tz)
Medical Stores Department — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
MSD's product category focus on antibiotics, cardiovascular drugs, and antimalarial & antiparasitic agents aligns with the prevalent health challenges in Tanzania. The substantial import value of Captopril ($150K) and Ery
Frequently Asked Questions — Medical Stores Department
What products does Medical Stores Department import from India?
Medical Stores Department imports 3 pharmaceutical products across 3 categories. Top imports: Erythromycin ($250.0K), Captopril ($150.0K), Pyrimethamine ($100.0K).
Who supplies pharmaceuticals to Medical Stores Department from India?
Medical Stores Department sources from 22 verified Indian suppliers. The primary supplier is Lincoln Pharmaceuticals Limited (28.3% of imports, $1.4M).
What is Medical Stores Department's total pharmaceutical import value?
Medical Stores Department's total pharmaceutical import value from India is $500.0K, based on 10 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Medical Stores Department focus on?
Medical Stores Department imports across 3 categories. The largest: Antibiotics (50.0%), Cardiovascular (30.0%), Antimalarial & Antiparasitic (20.0%).
Get Full Medical Stores Department Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Medical Stores Department identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Medical Stores Department's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 10 individual customs records matching Medical Stores Department.
- 5.Supplier Verification: Medical Stores Department sources from 22 verified Indian suppliers across 141 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.